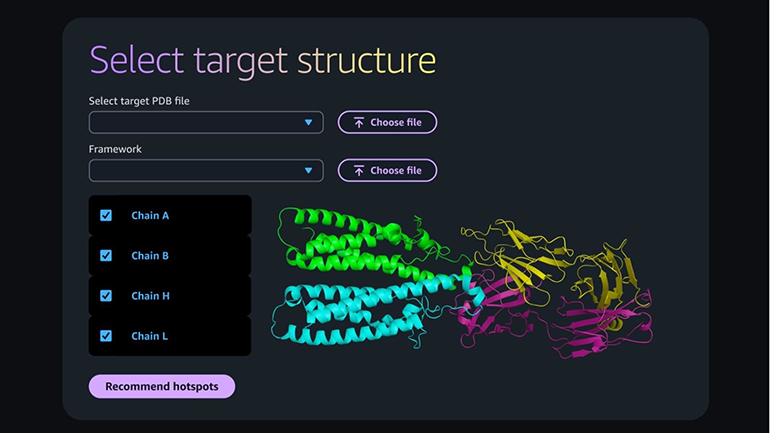
SEATTLE, WA – Amazon Web Services (AWS) has unveiled Amazon Bio Discovery, a groundbreaking application poised to dramatically accelerate drug discovery and development by integrating artificial intelligence (AI) with experimental wet-lab processes. The new platform, launched today, routes AI-designed antibody candidates directly to leading DNA synthesis and techbio firms, Twist Bioscience and Ginkgo Bioworks, for immediate synthesis and testing. Critically, the results from these experimental validations flow back into the same unified interface that generated the initial AI designs, creating a seamless, iterative loop designed to compress traditionally protracted research timelines from years to mere weeks.
This launch marks a significant escalation in AWS’s commitment to the life sciences sector, leveraging its robust cloud infrastructure and advanced AI capabilities to tackle some of the most persistent bottlenecks in pharmaceutical innovation. Early adopters of Amazon Bio Discovery include prominent institutions such as Bayer, the Broad Institute, Memorial Sloan Kettering Cancer Center (MSK), and Voyager Therapeutics, signaling broad industry confidence in the platform’s potential.
Revolutionizing Antibody Design with Integrated AI and Wet-Lab Workflows
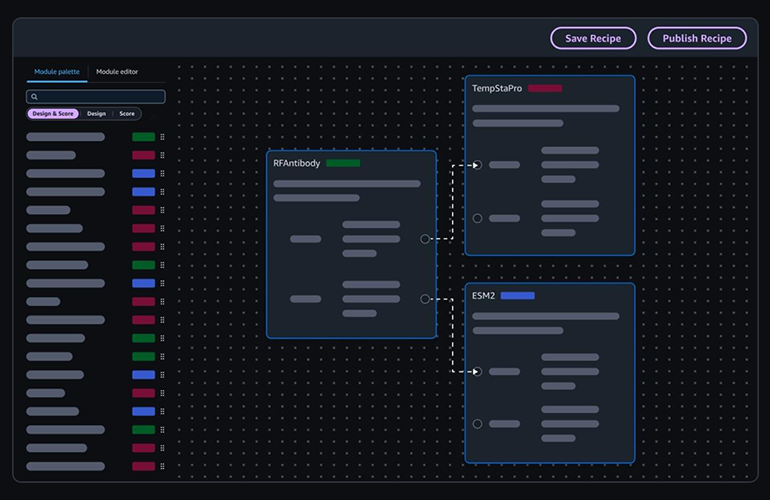
At its core, Amazon Bio Discovery combines a sophisticated catalog of biological foundation models (bioFMs) with an integrated wet-lab handoff mechanism. BioFMs are large AI models, meticulously trained on vast biological datasets, capable of generating and evaluating potential drug molecules with unprecedented speed and precision. These models are complemented by a natural-language agent, allowing researchers to design experiments, fine-tune models on their proprietary lab data, and interact with the platform using intuitive language rather than complex coding. This democratizes access to advanced AI tools, empowering a wider range of scientists to leverage computational power in their research.
The impact of this integration was vividly demonstrated in an early project with Memorial Sloan Kettering. Researchers, working under the leadership of Nai-Kong Cheung, M.D., Ph.D., Enid A. Haupt Chair in Pediatric Oncology at Memorial Sloan Kettering Cancer Center, utilized Amazon Bio Discovery to orchestrate multiple AI models. This collaborative effort resulted in the generation of nearly 300,000 novel antibody molecules. From this vast pool, the top 100,000 candidates were swiftly dispatched to Twist Bioscience for experimental validation. AWS reports that this streamlined process condensed an endeavor that historically could take up to a year into a matter of weeks.
Dr. Cheung underscored the urgency driving this innovation, stating, “Patients come here with a clock. We need results sooner.” This sentiment encapsulates the critical need for accelerated timelines in therapeutic development, particularly in areas like oncology where timely intervention can be life-saving. This specific antibody design project is a direct outgrowth of a broader collaboration between AWS and MSK, initially announced in February 2025, which aims to accelerate AI-driven cancer innovation across various research fronts.
The Strategic Imperative: Addressing the Drug Discovery Bottleneck
Traditional drug discovery is notoriously slow, expensive, and fraught with high failure rates. The journey from initial target identification to an approved drug can span 10-15 years and cost billions of dollars, with success rates often below 10% for compounds entering clinical trials. A significant portion of this time and cost is consumed in the preclinical phase, particularly in the iterative design, synthesis, and testing of molecular candidates. Antibodies, in particular, are complex biological molecules whose design and optimization often require extensive empirical testing. By automating and integrating these steps, Amazon Bio Discovery aims to fundamentally alter this paradigm.
The platform’s ability to generate hundreds of thousands of candidates and rapidly push the most promising ones into physical synthesis and testing represents a paradigm shift. This high-throughput, AI-driven approach minimizes manual intervention, reduces human error, and allows scientists to explore a far wider chemical and biological space than previously possible. The immediate feedback loop from wet-lab results back to the AI models enables continuous learning and refinement, fostering a virtuous cycle of accelerated discovery.
Broader AWS Initiatives in Life Sciences: A Multi-faceted Approach
Beyond Amazon Bio Discovery, AWS utilized recent symposium materials shared with media to outline several other strategic collaborations aimed at transforming various aspects of life sciences. These initiatives underscore AWS’s ambition to be a foundational technology provider across the entire biopharmaceutical value chain:
- AI-Powered Real-World Data Platform for Alzheimer’s Disease: AWS is collaborating with Labcorp and data-linkage firm Datavant to develop an AI-powered real-world data (RWD) platform specifically for Alzheimer’s disease research. This platform draws upon Labcorp’s extensive blood-based biomarker testing data, which the company claims is the broadest such portfolio for Alzheimer’s and dementia. The proprietary lab data serves as a key differentiator, with AWS providing Bedrock agents and SageMaker analytics for sophisticated data processing and analysis. Datavant contributes privacy-preserving data linkage capabilities, connecting lab results to medical claims data, while Labcorp supplies the crucial longitudinal diagnostic records. Early use cases for this platform are focused on identifying patient cohorts for clinical trial recruitment, a persistent and costly bottleneck in Alzheimer’s drug development, which often struggles to find suitable participants. The Alzheimer’s version of this platform is slated to complete initial validation this spring, with ambitious plans for expansion into inflammatory diseases, cardiometabolic conditions, women’s health, and oncology in 2026. Labcorp further highlighted its significant role in the industry, stating it supported over 85% of FDA drug approvals in 2025.
- AI-Enabled Clinical Trial Site Selection: In partnership with Merck and Boston Consulting Group (BCG), AWS is working on an AI-enabled solution to optimize clinical trial site selection. This initiative aims to improve the efficiency and speed of clinical trials by using AI to identify the most suitable sites, thereby reducing recruitment times and operational costs, which are major pain points in drug development.
- Verily Workbench General Availability: AWS is also collaborating with Verily to make Verily Workbench generally available for AWS users. Verily Workbench is a secure, cloud-based platform designed to facilitate large-scale biomedical research, enabling researchers to access, analyze, and collaborate on complex datasets. Its availability on AWS will broaden its reach and utility within the scientific community.
These diverse initiatives demonstrate AWS’s comprehensive strategy to embed its cloud and AI capabilities deeply within the life sciences ecosystem, addressing challenges from early discovery to clinical development and real-world evidence generation.
The Ascent of AI Agents in Drug Discovery: An Industry-Wide Trend
The emergence of "agents" as a preferred architectural framework for AI in drug discovery is a burgeoning trend across the industry, and Amazon Bio Discovery’s use of a natural-language agent positions it squarely within this cutting-edge movement. AI agents are designed to autonomously perform tasks, make decisions, and interact with other systems or humans to achieve specific goals, often mimicking human-like reasoning and problem-solving.
This shift towards agent-based AI is evident in the strategic moves of several key players:
- Isomorphic Labs: Google DeepMind’s spinout, Isomorphic Labs, has secured high-value drug-design partnerships with pharmaceutical giants such as Eli Lilly, Novartis, and Johnson & Johnson. These deals, announced in 2024, have a potential aggregate value exceeding $3 billion, underscoring the immense commercial potential seen in advanced AI for drug discovery. Isomorphic Labs focuses on leveraging DeepMind’s foundational AI research to predict protein structures and interactions, a critical step in rational drug design.
- Insilico Medicine: A pioneer in AI-driven drug discovery, Insilico Medicine’s Pharma.AI platform developed Rentosertib, which reached the preclinical candidate stage in just 18 months. This compound has since completed a Phase IIa trial for idiopathic pulmonary fibrosis, with promising results published in Nature Medicine in 2025. Insilico’s success demonstrates the capability of AI to drastically shorten discovery timelines and bring novel therapies closer to patients.
- NVIDIA’s BioNeMo: NVIDIA, a leader in AI hardware and software, is also pitching a similar "co-scientist" architecture through its BioNeMo platform. BioNeMo provides a generative AI framework for drug discovery, allowing researchers to train, customize, and deploy AI models for tasks like protein structure prediction, molecular generation, and simulation, effectively acting as an intelligent assistant to scientists.
- Anthropic’s Acquisition of Coefficient Bio: In a notable development in early April, AI safety-focused company Anthropic reportedly acquired Coefficient Bio for $400 million in stock. Coefficient Bio, an eight-month-old drug design startup, was staffed by computational biologists from prestigious institutions like Evozyne, Genentech, and Prescient Design. This acquisition signals a deeper push by general AI companies into the specialized domain of drug discovery, recognizing the strategic importance and potential for impact.
These examples highlight a collective industry recognition that AI agents and large biological models are not just incremental improvements but rather transformative technologies capable of fundamentally redefining the drug discovery landscape. They promise to automate complex, data-intensive tasks, generate novel hypotheses, and significantly accelerate the identification and optimization of therapeutic candidates.
Technological Underpinnings: The Power of Cloud and Generative AI
The efficacy of Amazon Bio Discovery hinges on several core technological pillars. At its foundation is the AWS cloud infrastructure, providing unparalleled scalability, security, and computational power. Drug discovery workflows often require processing petabytes of data and running millions of simulations, tasks that are infeasible without elastic cloud resources. AWS’s offerings like Amazon Bedrock, which provides access to foundation models, and Amazon SageMaker, a fully managed service for building, training, and deploying machine learning models, are critical enablers.
The biological foundation models (bioFMs) are particularly significant. Analogous to large language models (LLMs) that learn patterns in human language, bioFMs learn the intricate rules governing biological systems, from protein folding and molecular interactions to genetic pathways. Trained on vast datasets of genomic, proteomic, and chemical information, these models can generate novel molecular structures, predict their properties, and even simulate their behavior in silico. This generative AI capability allows researchers to move beyond screening existing libraries to designing entirely new molecules tailored for specific therapeutic targets.
The natural-language agent further enhances accessibility and efficiency. Researchers can simply describe their experimental goals or desired molecular properties, and the agent translates these into computational tasks, orchestrating the bioFMs, data analysis, and even the physical handoff to partner labs. This intuitive interface bridges the gap between biological expertise and computational complexity.
Implications and Future Outlook
The launch of Amazon Bio Discovery and similar platforms signals a pivotal moment in the history of medicine. The implications are far-reaching:
- Accelerated Drug Development: The most immediate impact is the potential to drastically reduce the time it takes to bring new drugs to market. By compressing preclinical stages from years to weeks, patients could gain access to life-saving therapies much faster.
- Reduced Costs: While AI platforms involve significant initial investment, the long-term potential for cost reduction is substantial. Faster development cycles, higher success rates in early stages, and optimized experimental design can lead to more efficient R&D spending.
- Enhanced Drug Design: Generative AI allows for the design of novel molecules with optimized properties, potentially leading to more effective and safer drugs that were previously unimaginable through traditional methods.
- Democratization of Advanced Research: By simplifying access to sophisticated AI tools through user-friendly interfaces, Amazon Bio Discovery can empower smaller biotech firms and academic institutions to compete with larger pharmaceutical companies, fostering broader innovation.
- Ethical Considerations: As AI becomes more embedded in drug discovery, discussions around data privacy, algorithmic bias, intellectual property, and the validation of AI-generated insights will become increasingly important. Robust regulatory frameworks and ethical guidelines will be crucial to ensure responsible innovation.
- Competitive Landscape: AWS’s move intensifies the competition among cloud providers (Microsoft Azure, Google Cloud) and specialized AI biotech companies to dominate the life sciences market. The race is on to provide the most comprehensive, integrated, and effective platforms for drug discovery and development.
In essence, Amazon Bio Discovery represents more than just a new application; it symbolizes a growing convergence of cloud computing, artificial intelligence, and biotechnology. By creating an end-to-end, AI-driven pipeline from digital design to physical testing, AWS is not merely offering tools but orchestrating an entirely new paradigm for how the next generation of medicines will be discovered and delivered. This could profoundly reshape the pharmaceutical industry, offering renewed hope for patients facing diseases that currently lack effective treatments.














Leave a Reply