In an era increasingly shaped by the transformative power of artificial intelligence, a Los Angeles-based telehealth startup, MEDVi, has captured significant attention for its rapid ascent, seemingly validating OpenAI CEO Sam Altman’s 2024 prediction that a single entrepreneur leveraging AI could build a business worth over a billion dollars. However, this impressive growth narrative, recently highlighted by The New York Times, is now overshadowed by a growing storm of regulatory warnings, allegations of deceptive marketing practices, and multiple class-action lawsuits, raising serious questions about the ethics and legality of its operations in the sensitive healthcare sector.
The Allure of AI-Powered Entrepreneurship Meets Healthcare Reality
Matthew Gallagher, the entrepreneur behind MEDVi, has boldly proclaimed his firm as "the fastest growing company in history," a claim that resonated with the burgeoning enthusiasm for AI-driven ventures. The company, which primarily markets compounded GLP-1s (glucagon-like peptide-1 receptor agonists) and other pharmaceuticals, presented itself as a prime example of Altman’s vision. On April 2, 2026, The New York Times published a profile showcasing MEDVi’s projected $1.8 billion in sales for 2026, achieved with a lean operation consisting of Gallagher, his brother Elliot as the only two full-time employees, supplemented by contract engineers, account managers, and outside agencies. The Times article, which reportedly gained access to MEDVi’s financials for revenue and profit verification, detailed Gallagher’s reliance on a suite of AI services, including ChatGPT, Claude, Grok, MidJourney, and Runway, to build the company from the ground up. Critical operational functions such as connecting patients with doctors, pharmacy services, shipping, and compliance were outsourced to telehealth infrastructure platforms like CareValidate and OpenLoop Health. This model epitomized the promise of AI: hyper-efficiency, minimal overhead, and unprecedented scalability.
A Rapid Ascent and the Unseen Regulatory Cloud

While MEDVi was basking in the glow of mainstream media recognition, a critical piece of information was notably absent from The New York Times profile: a stern warning letter from the U.S. Food and Drug Administration (FDA) issued six weeks prior, on February 20, 2026. This letter, available in public records, directly addressed MEDVi LLC, operating under the name MEDVi, for misbranding the very compounded drugs that were driving its revenue. The FDA specifically cited that MEDVi’s website at medvi.io (reviewed in December 2025) falsely suggested the company was the actual compounder of the semaglutide and tirzepatide it sold. Furthermore, the agency found claims such as "Same active ingredient as Wegovy® and Ozempic®" and "Same active ingredient as Mounjaro® and Zepbound®" misleading, as they falsely implied FDA approval or evaluation for the compounded versions. The FDA unequivocally warned that failure to correct these violations could lead to severe enforcement actions, including product seizure or injunctions, adding that the cited violations were not an exhaustive list of potential issues. The current consumer-facing site for MEDVi is home.medvi.org, indicating a potential shift in online presence since the FDA’s initial review.
The FDA’s warning letter highlights a critical distinction in the pharmaceutical landscape. While compounding pharmacies are permitted to create customized medications for individual patients, especially during drug shortages, they are strictly prohibited from misrepresenting these products as FDA-approved or equivalent to proprietary brand-name drugs. The immense popularity and supply chain challenges of GLP-1s like Ozempic, Wegovy, Mounjaro, and Zepbound, which have become blockbuster medications for weight loss and type 2 diabetes management, have created a fertile ground for compounded alternatives. The global GLP-1 market is projected to reach hundreds of billions of dollars in the coming years, fueling a surge in telehealth companies offering these drugs. However, the FDA has maintained a vigilant stance, emphasizing that compounded versions have not undergone the rigorous safety and efficacy trials required for FDA approval and may carry different risks.
A Pattern of Deceptive Marketing: AI-Generated Personas and Unverified Claims
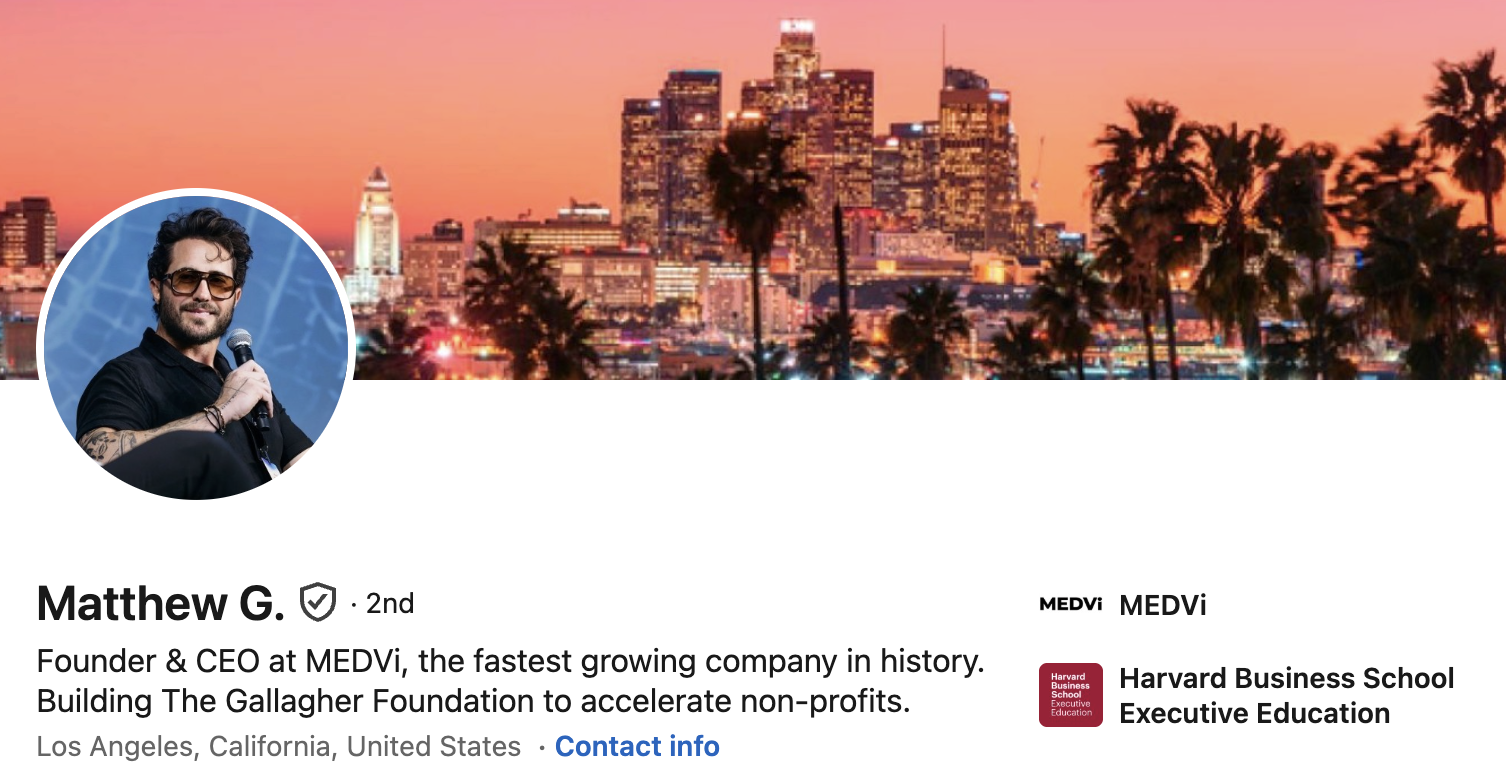
Beyond the FDA’s specific misbranding concerns, a review by Drug Discovery & Development uncovered a broader pattern of questionable marketing tactics employed by MEDVi, particularly involving the extensive use of AI-generated personas and potentially misleading claims. A search of Meta’s Ad Library for "medvi" revealed over 5,000 active advertisements, many of which ran under fabricated physician personas. For instance, a Facebook page for "Dr. Robert Whitworth," promoting MEDVi’s QUAD erectile dysfunction product through sponsored ads, bizarrely listed its category as "Entertainment website" and provided a non-existent address: "2015 Nutter Street, Cameron, MT, 64429." Other advertisements utilized names like "Professor Albust Dongledore" and "Dr. Richard Hörzgock," featuring AI-generated video testimonials that recycled identical scripts across multiple fabricated identities. In several instances, a doctor’s headshot would be displayed on the page, while the accompanying ad featured an entirely unrelated individual delivering a patient testimonial, further blurring the lines between authentic medical advice and marketing theatrics.
This advertising strategy aligns with a wider trend of AI-fueled deceptive practices that have drawn the attention of regulators. In December 2025, a bipartisan coalition of 35 state attorneys general penned a letter to Meta, alleging the social media giant was permitting companies to advertise weight-loss products with deceptive, AI-generated "before-and-after" comparison photos. The letter unequivocally stated, "The use of artificial intelligence to fabricate images, spokespeople, and medical claims crosses a line and makes these ads particularly dangerous." This concern was echoed by a late 2025 Reuters investigation, which reported that Meta was "earning a fortune on a deluge of fraudulent ads," with internal documents reportedly projecting one-tenth of its 2024 revenue stemming from "ads for scams and banned goods." MEDVi’s own website fine print offers a revealing admission: a disclaimer at the bottom of the site notes, "Individuals appearing in advertisements may be actors or AI portraying doctors and are not licensed medical professionals." While this disclaimer exists, its placement and the overall presentation of the marketing materials raise questions about its effectiveness in mitigating consumer deception.
Earlier Red Flags: Futurism’s Investigation and Unaffiliated Doctors
The current scrutiny is not the first time MEDVi’s marketing practices have come under fire. In May 2025, a report by Futurism meticulously documented several alarming instances of AI-generated content on MEDVi’s website. This included AI-generated patient photos, deepfaked "before-and-after" weight loss images (traced to photos that had circulated online for years with faces swapped by AI), and even AI-generated Ozempic box imagery containing clear errors. Adding to the deceptive veneer, the website prominently displayed logos from prestigious publications like The New York Times, Bloomberg, and Forbes, implying editorial coverage that, at that point, did not exist. When Futurism contacted doctors listed on MEDVi’s site, at least one, osteopathic practitioner Tzvi Doron, explicitly denied any association with the company and requested his removal. In the subsequent Times profile, Gallagher acknowledged that MEDVi’s initial site indeed featured AI-generated photos and before-and-after weight loss images with swapped faces, a concession that retrospectively validated Futurism‘s findings.
Questionable Physician Affiliations and Board Certifications
Further investigation into MEDVi’s current operations reveals additional concerns regarding the transparency and legitimacy of its listed medical professionals. MEDVi’s current site lists two physicians: Dr. Ana Lisa Carr and Dr. Kelly Tenbrink. Both are licensed doctors who reportedly work together at Ringside Health, a concierge practice in Wellington, Florida, catering to the equestrian community. However, neither physician is identified on MEDVi’s site as being affiliated with Ringside Health, potentially obscuring their primary practice. On MEDVi’s site, Dr. Tenbrink is listed under "American Board of Emergency Medicine," while Dr. Carr is listed under St. George’s University, School of Medicine, her medical school. A check with the Florida Department of Health practitioner profiles for both physicians, however, states that neither "hold any certifications from specialty boards recognized by the Florida board." Furthermore, a search of the American Board of Emergency Medicine’s public directory, which lists 48,863 certified members, yielded no current affiliation for Dr. Tenbrink, casting doubt on the accuracy of her listed board certification on MEDVi’s platform. These discrepancies raise significant questions about the qualifications and transparency of the medical oversight provided by MEDVi.
A Gathering Legal Storm: Data Breaches and "Snake Oil" Allegations
MEDVi’s rapid expansion, from 250,000 customers by the end of 2025 to over 500,000 "MEDVi patients" advertised on its current homepage, has been described by Gallagher as "insane" and accelerating. The site promises "unlimited 24/7 support" and "doctor-led plans & coaching." The medical services are presented as being provided by OpenLoop Health clinicians, who "retain the decision to prescribe compounded GLP-1s to patients," with both Dr. Carr’s and Dr. Tenbrink’s practices connected to CareValidate, one of the telehealth platforms Gallagher cited as a partner.
However, the backend infrastructure on which MEDVi relies is itself embroiled in significant legal and ethical challenges. OpenLoop Health, a Des Moines, Iowa-based telehealth infrastructure company, recently disclosed a substantial cyber breach. On January 7, 2026, a threat actor breached OpenLoop’s systems, claiming to have exfiltrated records from approximately 1.6 million patients, including sensitive personal and medical information such as names, contact details, dates of birth, and medical data. OpenLoop notified the Texas Attorney General in March 2026, confirming at least 68,160 affected individuals in that state alone, and now faces multiple class-action lawsuits stemming from this breach.
More critically, in November 2025, a North Carolina consumer filed a class-action complaint in the U.S. District Court for the District of Delaware against OpenLoop Health and compounding pharmacy Triad Rx. The lawsuit, first reported by Fierce Healthcare, alleges that these firms manufactured and sold what the complaint provocatively labels "modern-day snake oil": compounded oral tirzepatide tablets with "no demonstrated mechanism of absorption or efficacy." The named plaintiff purchased these pills through MEDVi, which the complaint alleges functioned as one of dozens of nearly identical consumer-facing telehealth storefronts operating within a network managed by OpenLoop.
The scientific basis for the "snake oil" allegation is compelling. Tirzepatide is a large peptide molecule that is highly susceptible to destruction by digestive enzymes before it can reach the bloodstream and exert its therapeutic effects. The only FDA-approved oral GLP-1 medication, Rybelsus (oral semaglutide), required the incorporation of a specialized absorption enhancer called SNAC to achieve even a modest 1% bioavailability, and even then, it must be taken under tightly controlled conditions (e.g., on an empty stomach with a small amount of water, avoiding food and other medications for a period). The complaint posits that without such a sophisticated delivery mechanism, compounded oral tirzepatide is effectively inert.
The lawsuit brings claims under the Racketeer Influenced and Corrupt Organizations Act (RICO), various state consumer protection statutes, and common law fraud, painting a picture of a coordinated scheme to market ineffective medications. It specifically alleges that MEDVi was just one of at least a dozen similar telehealth storefronts, each with a designated OpenLoop patient waiting room, all marketing the same product through the same backend infrastructure. The complaint even cites Dr. David Mansour, another physician who appeared in a January 2026 MEDVi press release but is no longer listed on the site, along with Drs. Carr and Tenbrink, alleging that their "Meet the incredible doctors we’ve partnered with" presentation was integral to the marketing scheme.
Beyond these significant legal challenges, MEDVi has faced other legal entanglements. The company was served in Siuksta v. MEDVi, LLC, a federal Telephone Consumer Protection Act (TCPA) lawsuit filed in May 2025, but failed to appear, leading to a voluntary dismissal after the court noted the default. Separately, docket listings indicate James v. Medvi LLC, No. 8:26-cv-00641, with a complaint filed on March 20, 2026, in the Central District of California, suggesting further legal battles on the horizon.
Implications for the Telehealth and AI Industries
The case of MEDVi serves as a stark reminder of the complexities and potential pitfalls at the intersection of rapidly advancing AI technology, the booming telehealth industry, and the highly regulated pharmaceutical sector. While AI offers unprecedented opportunities for efficiency and scale in business, its application in healthcare demands rigorous ethical oversight, transparency, and adherence to established regulatory frameworks. The alleged use of AI to generate deceptive marketing personas and unverified medical claims not only risks consumer harm but also erodes public trust in both telehealth services and AI technologies.
The lack of response from MEDVi, Matthew Gallagher, Dr. Carr, and Dr. Tenbrink to Drug Discovery & Development‘s requests for comment prior to publication further deepens the concerns surrounding the company’s operations. As regulatory bodies like the FDA and state attorneys general intensify their scrutiny of online medical marketing and compounded drugs, and as class-action lawsuits proceed, the future of MEDVi and its AI-driven model remains uncertain. This developing story will undoubtedly influence discussions around appropriate regulatory frameworks for AI in healthcare, the responsibilities of telehealth platforms, and the critical need for consumer protection in a digital health landscape that is evolving at an unprecedented pace.








Leave a Reply