A groundbreaking study led by researchers at the LKS Faculty of Medicine, the University of Hong Kong (HKUMed), has identified a critical biological mechanism that explains how physical movement translates into bone strength. The research, which centers on a specific protein known as Piezo1, serves as a significant milestone in the field of regenerative medicine. By pinpointing the "exercise sensor" within the body, the team has opened a new frontier for the development of pharmacological interventions designed to mimic the benefits of physical activity. This discovery is particularly vital for populations who are physically unable to exercise, such as the elderly, bedridden patients, and those suffering from chronic debilitating conditions.
The study, conducted by the Department of Medicine at the School of Clinical Medicine, was recently published in the prestigious international journal Signal Transduction and Targeted Therapy. The findings provide a molecular blueprint for how mechanical forces—generated through walking, running, or resistance training—are detected by bone marrow cells and converted into signals that promote bone formation while simultaneously inhibiting the accumulation of fat.
The Global Burden of Osteoporosis and Age-Related Bone Loss
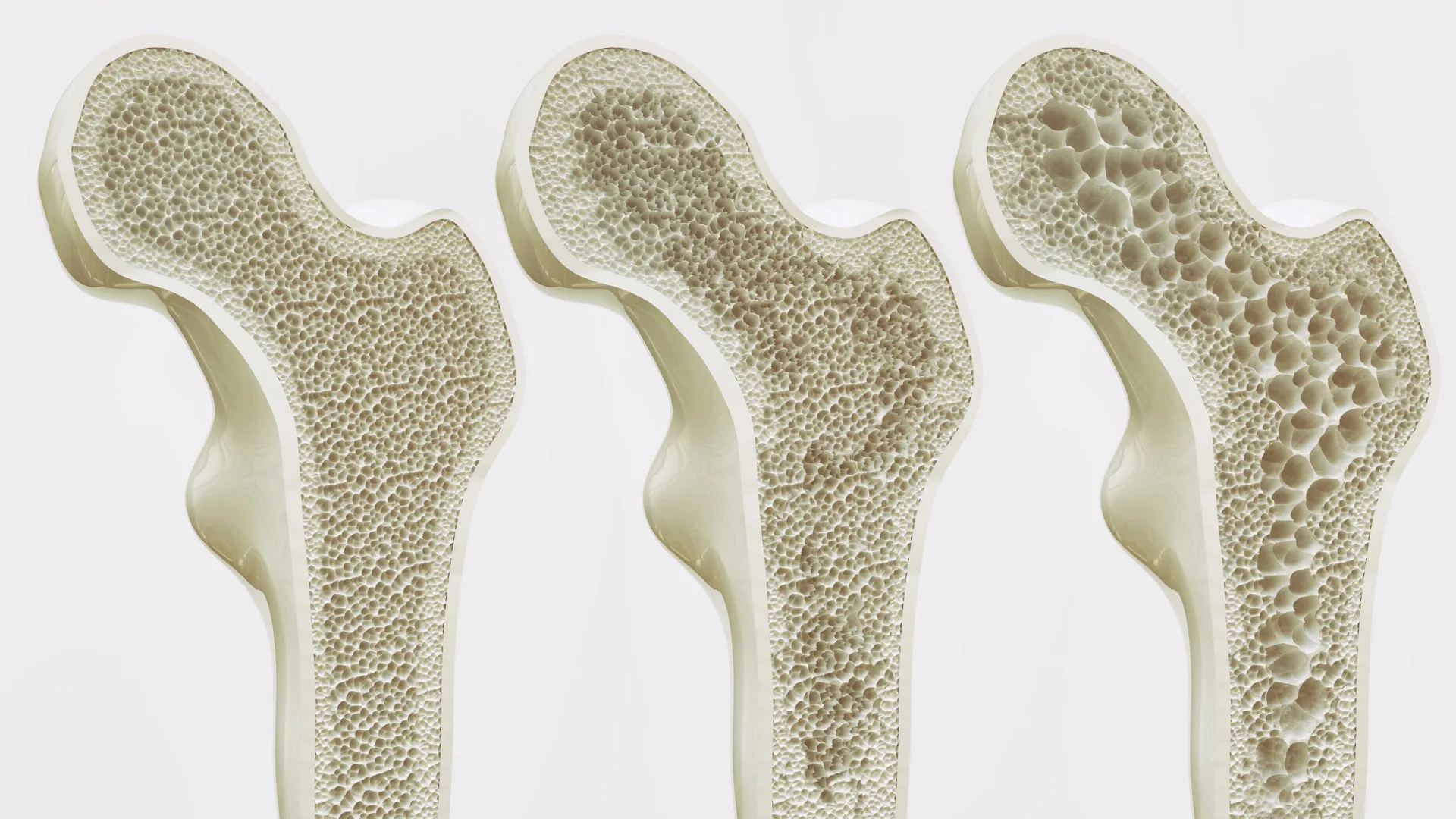
Osteoporosis is often described as a "silent epidemic" because bone loss occurs without symptoms until a fracture happens. According to the World Health Organization (WHO), the condition affects hundreds of millions of people worldwide. Statistics indicate that approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture in their lifetime. These injuries are not merely structural; they are life-altering events that frequently lead to chronic pain, permanent disability, and a significant decrease in life expectancy.
In Hong Kong, the challenge is amplified by a rapidly aging demographic. Current data suggests that osteoporosis affects approximately 45% of women and 13% of men aged 65 and older in the territory. As the population continues to age, the strain on the public healthcare system is expected to grow exponentially. Hip fractures, in particular, require intensive surgical intervention and long-term rehabilitation, often resulting in a loss of independence for the patient.
Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor in the Department of Medicine at HKUMed, emphasized the urgency of the research. "Osteoporosis and age-related bone loss often leave elderly and bedridden patients vulnerable to fractures. While physical activity is the most effective natural defense against bone loss, many patients simply cannot perform the necessary movements. Our goal was to understand the molecular level of this process so we can replicate it for those who need it most."
The Cellular Mechanics: Bone versus Fat
To understand the HKUMed discovery, one must look at the internal environment of the bone marrow. Within the marrow reside mesenchymal stem cells (MSCs), which are versatile progenitor cells capable of transforming into different types of tissue. In a healthy, active individual, these stem cells are regularly nudged by mechanical forces to become osteoblasts, the cells responsible for building new bone.
However, aging and physical inactivity disrupt this delicate balance. As the body ages or remains sedentary, the "lineage commitment" of these stem cells shifts. Instead of becoming bone-forming cells, the MSCs increasingly differentiate into adipocytes, or fat cells. This shift results in a dual tragedy for bone health: the density of the bone decreases while the marrow becomes clogged with fat. This fatty infiltration further weakens the skeletal structure and creates an inflammatory environment that inhibits the repair of existing bone.
Identifying Piezo1: The Molecular Exercise Sensor
The HKUMed research team, in collaboration with international experts, utilized advanced mouse models and human stem cell cultures to investigate how cells "feel" exercise. They identified a protein called Piezo1, located on the surface of mesenchymal stem cells in the bone marrow. Piezo1 belongs to a family of ion channels that are activated by mechanical pressure.
In the study’s experimental phase, researchers observed that when mice engaged in physical activity, the Piezo1 protein on their stem cells was triggered. This activation sent a cascade of signals through the cell, instructing it to prioritize bone production. Furthermore, the activation of Piezo1 suppressed the genetic pathways that lead to fat formation.
To confirm the protein’s necessity, the team conducted "loss-of-function" experiments. When Piezo1 was genetically removed or inhibited, the stem cells defaulted to fat production, even when the mice were active. The absence of Piezo1 led to accelerated bone loss, mirroring the effects of rapid aging or prolonged bed rest.
The Role of Inflammatory Signaling
A critical aspect of the study involved identifying the secondary signals that occur when the exercise sensor is absent. The researchers found that the lack of Piezo1 activity triggered the release of specific inflammatory markers, namely Ccl2 and lipocalin-2. These proteins act as chemical messengers that further push stem cells toward fat production and actively interfere with the growth of healthy bone tissue.
By blocking these inflammatory signals in the laboratory, the researchers were able to partially restore bone health, suggesting that the Piezo1 pathway is a "master regulator" that controls multiple downstream effects. This multi-layered understanding of the pathway provides several potential targets for future drug development.
The Concept of "Exercise Mimetics"
The most significant implication of this research is the potential for "exercise mimetics"—a class of drugs that could provide the physiological benefits of a workout through a pill or injection.
Dr. Wang Baile, Research Assistant Professor at HKUMed and co-leader of the study, highlighted the transformative nature of this concept. "For a patient who has been bedridden due to a stroke or a severe fracture, the traditional advice to ‘exercise more’ is impossible to follow. By activating the Piezo1 pathway chemically, we can essentially trick the body into thinking it is moving. This would maintain bone mass and strength, preventing the downward spiral of frailty."
This approach represents a shift from current osteoporosis treatments, which largely focus on slowing down bone resorption (the breakdown of bone) rather than actively stimulating the biological pathways associated with physical movement.
International Collaboration and Methodology
The study was a highly collaborative effort, reflecting the global nature of medical research. Joining the HKUMed team was Professor Eric Honoré, a renowned leader at the Institute of Molecular and Cellular Pharmacology of the French National Centre for Scientific Research (CNRS). Professor Honoré, who also serves as a Visiting Professor at HKUMed, brought expertise in mechanobiology—the study of how physical forces affect cells.
The methodology involved a rigorous timeline of testing:
- In Vitro Analysis: Human mesenchymal stem cells were subjected to mechanical stretching in a lab setting to observe Piezo1 activation.
- In Vivo Mouse Models: Researchers compared bone density and marrow fat levels in active mice versus sedentary mice, and in mice with and without the Piezo1 protein.
- Signaling Mapping: The team mapped the entire pathway from the physical touch on the cell membrane to the genetic expression in the nucleus.
- Pharmacological Testing: Initial tests were conducted to see if chemical agonists (activators) could stimulate Piezo1 in the absence of physical force.
Future Outlook: Clinical Applications and Challenges
While the discovery is a major scientific breakthrough, the transition from lab results to clinical therapy involves several steps. The research team is now focusing on identifying the most effective and safe chemical compounds that can target Piezo1 without causing off-target effects in other organs, as Piezo1 is also present in the cardiovascular and nervous systems.
The potential applications extend beyond osteoporosis. Astronauts in microgravity environments suffer from rapid bone loss due to the lack of mechanical loading; Piezo1-based therapies could mitigate these effects during long-term space missions. Similarly, athletes recovering from major injuries could use these treatments to prevent bone atrophy during their immobilization period.
Professor Eric Honoré noted the broader impact: "This offers a promising strategy beyond traditional physical therapy. We are looking at a future where we can provide the biological benefits of exercise through targeted treatments, substantially reducing the risk of fractures in the most vulnerable groups."
Conclusion and Support
The study was supported by an array of prestigious funding bodies, including the Research Grants Council of Hong Kong, the National Key R&D Program of China, and the French National Research Agency. The multi-jurisdictional support underscores the importance of the findings in addressing a global health crisis.
As the HKUMed team moves toward the next phase of their research, the medical community remains optimistic. The identification of Piezo1 as the bone’s exercise sensor provides a definitive target for a new generation of medications. For the millions of people worldwide who face the daily fear of a life-changing fracture, this molecular "exercise in a pill" represents more than just a scientific curiosity—it represents a path back to strength, mobility, and independence.














Leave a Reply