In a landmark development for regenerative medicine and oncology, scientists at the University of Basel and University Hospital Basel have successfully engineered a comprehensive, three-dimensional model of human bone marrow using exclusively human cells. This achievement, recently detailed in the journal Cell Stem Cell, marks the first time that the intricate network of bone cells, blood vessels, nerves, and immune cells has been recreated in a laboratory setting without the use of animal-derived components. The breakthrough provides a sophisticated platform for studying blood formation, testing new pharmacological compounds, and developing personalized treatment protocols for complex blood cancers.
The Biological Complexity of the Bone Marrow Niche
The bone marrow is often described as the body’s "blood factory," a highly specialized tissue responsible for the continuous production of red blood cells, white blood cells, and platelets. Under normal physiological conditions, this process, known as hematopoiesis, occurs seamlessly and goes largely unnoticed by the individual. However, when the regulatory mechanisms of the bone marrow fail, the consequences are often life-threatening, manifesting as leukemias, lymphomas, or various forms of anemia.
Understanding why these processes fail requires a deep dive into the bone marrow "niches"—specialized microenvironments that dictate the behavior of stem cells. Among these, the endosteal niche is of particular interest to oncologists. Located near the inner surface of the bone, this niche is composed of a dense matrix of minerals, blood vessels, and nervous tissue. It is here that blood cancer cells often seek refuge, entering a dormant state that allows them to resist conventional chemotherapy and radiation treatments. Until the Basel team’s intervention, recreating the structural and functional complexity of the endosteal niche in a laboratory setting had remained an elusive goal for bioengineers.
A New Era of Bioengineering: The Human-Centric Approach
For decades, the gold standard for bone marrow research has been animal models, particularly mice. While mouse studies have yielded invaluable insights into mammalian biology, they possess inherent limitations. The physiological differences between murine and human hematopoietic systems—including different cytokine signaling pathways and bone densities—mean that results from animal studies do not always translate accurately to human clinical trials. Furthermore, traditional "in vitro" (test tube) systems were historically limited to two-dimensional cell cultures, which fail to capture the 3D spatial relationships and mechanical stresses present in living tissue.
The research team, led by Professor Ivan Martin and Dr. Andrés García García from the Department of Biomedicine, sought to bridge this gap by utilizing induced pluripotent stem cells (iPSCs). These are adult cells that have been molecularly reprogrammed to an embryonic-like state, giving them the capacity to differentiate into any cell type in the body. By guiding these stem cells through a series of controlled developmental stages, the researchers were able to generate the diverse array of cell types required to populate a functional bone marrow environment.
Chronology of Development and Technical Specifications
The development of this 3D model followed a rigorous, multi-stage engineering process designed to mimic natural bone development (ossification).
- Scaffold Fabrication: The process began with the creation of an artificial bone framework made of hydroxyapatite. This mineral is the primary inorganic constituent of human bone and teeth, providing the necessary structural integrity and chemical cues for cell attachment.
- Cellular Integration: Human iPSCs were introduced into the hydroxyapatite scaffold. Using specific growth factors and signaling molecules, the researchers directed these cells to differentiate into mesenchymal lineages (which form bone and fat) and endothelial lineages (which form blood vessels).
- Maturation Phase: The construct was placed in a bioreactor system that provided a continuous flow of nutrients and oxygen, simulating the circulatory environment of the human body. Over several weeks, the cells organized themselves into a complex tissue structure.
- Functional Validation: Once the niche was established, the team introduced hematopoietic stem cells. The success of the model was confirmed when these cells began to proliferate and differentiate into mature blood cells, maintaining this function for several weeks in the laboratory.
The resulting model is notably larger and more robust than previous iterations in the field. Measuring approximately eight millimeters in diameter and four millimeters in thickness, the tissue provides a sufficient volume for detailed analysis and multi-point sampling, a critical requirement for high-quality scientific data.
Supporting Data and Comparative Analysis
The Basel study provided extensive data comparing the lab-grown tissue to actual human bone marrow biopsies. Molecular analysis confirmed that the gene expression profiles of the cells within the 3D model closely mirrored those found in vivo. Specifically, the expression of key proteins involved in cell-to-cell communication and the "homing" of stem cells to the niche were found to be highly accurate.
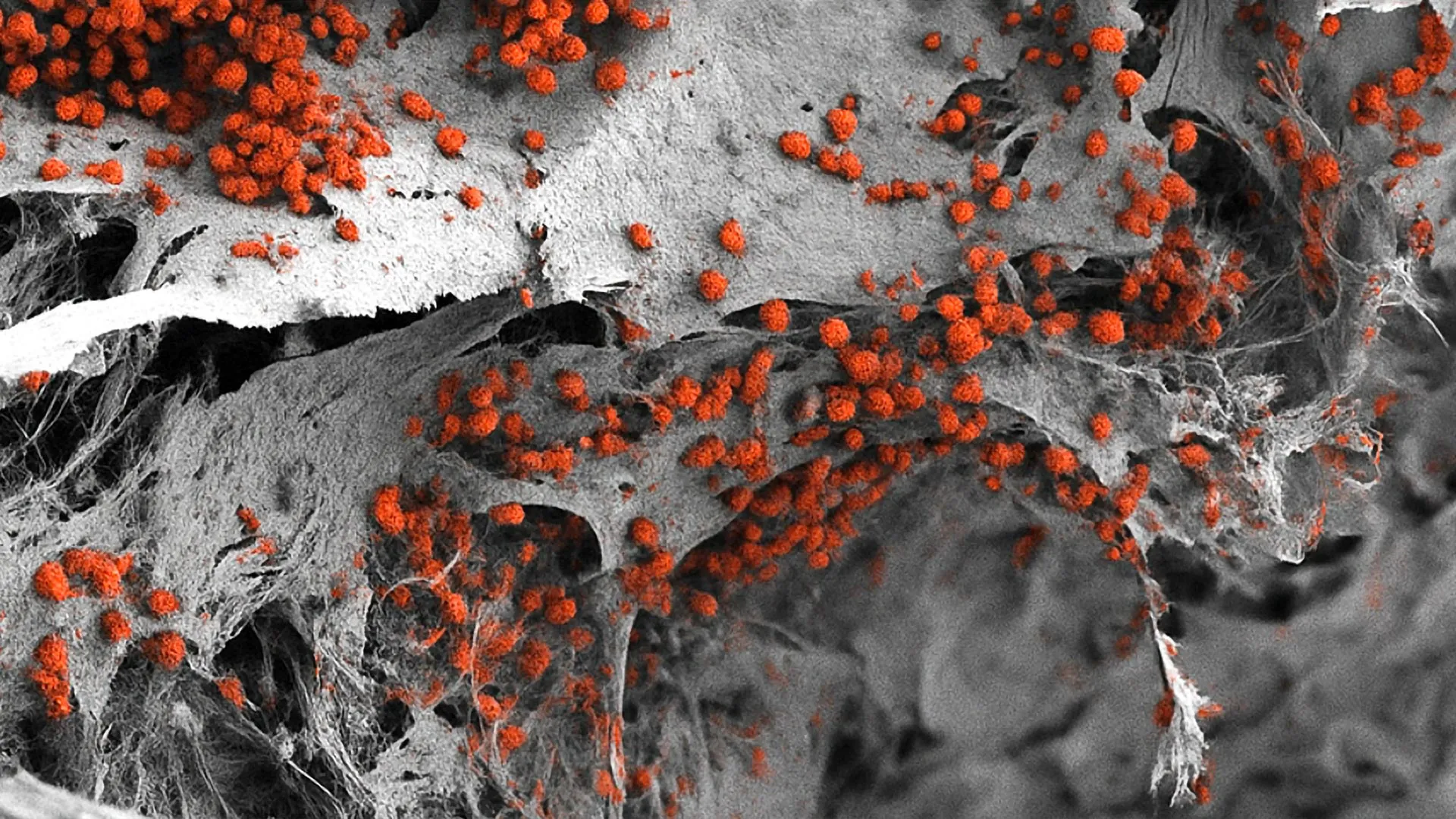
One of the most significant data points revealed by the study was the model’s ability to support the survival of human leukemia cells. In traditional 2D cultures, cancer cells often behave differently than they do in a patient’s body, leading to false positives or negatives during drug testing. In the 3D human model, the leukemia cells interacted with the bone and vascular cells in a manner that replicated the "chemo-resistance" seen in clinical settings. This confirms that the model is not just a structural replica, but a functional biological analog.
Implications for Drug Development and the 3R Principles
The pharmaceutical industry faces a significant challenge: approximately 90% of drugs that pass animal testing fail during human clinical trials, often due to unforeseen toxicity or lack of efficacy in the human biological context. The creation of a fully human bone marrow model offers a potential solution to this "translational gap."
By using human tissue early in the drug discovery phase, researchers can identify which compounds are truly effective against human cells before moving to expensive and time-consuming clinical trials. Dr. Andrés García García noted, however, that while the current model is a breakthrough for biological accuracy, its size presents a challenge for high-throughput screening. "To test hundreds or thousands of drug variations simultaneously, we will need to work on miniaturizing the platform," he explained.
Furthermore, this research aligns with the global "3R" mandate: to Replace, Reduce, and Refine the use of animals in scientific research. The University of Basel has been a vocal proponent of these principles, and the new bone marrow model represents a tangible step toward a future where human-based systems replace animal proxies in many areas of medical science.
Reactions from the Scientific Community
The publication in Cell Stem Cell has sparked significant interest among hematologists and bioengineers worldwide. Independent experts have praised the study for its "holistic" approach, noting that previous models often ignored the role of the nervous system or the specific mineral composition of the bone.
"The inclusion of vascular and neural elements within a synthetic hydroxyapatite scaffold is a masterclass in tissue engineering," said one independent reviewer. "It moves us away from the reductionist view of the bone marrow as just a bag of cells and toward a view of it as a complex organ system."
Hospital administrators and oncologists have also expressed optimism. The potential to create "patient-on-a-chip" systems—where a patient’s own cells are used to grow a model of their bone marrow—could revolutionize how we treat aggressive blood cancers. Doctors could theoretically test five different chemotherapy cocktails on the patient’s lab-grown marrow to see which one kills the cancer cells most effectively without destroying the healthy stem cells, all before the patient receives a single dose.
Future Outlook: Personalized Medicine and Scaling
While the current model is a monumental first step, the researchers at the University of Basel emphasize that there is still work to be done. The next phase of research will focus on several key areas:
- Miniaturization: Developing "micro-niche" versions of the model that can fit onto standard laboratory plates for automated drug testing.
- Disease Modeling: Creating specific versions of the marrow that replicate conditions like myelofibrosis (scarring of the bone marrow) or rare genetic blood disorders.
- Longevity: Extending the lifespan of the laboratory model from weeks to months to study long-term drug toxicity and the slow progression of chronic leukemias.
The ultimate vision is the integration of these models into routine clinical practice. As the costs of iPSC technology and 3D bio-printing continue to decrease, the possibility of providing every cancer patient with a personalized "avatar" of their own bone marrow becomes increasingly feasible.
In conclusion, the work of Professor Ivan Martin, Dr. Andrés García García, and their team represents a paradigm shift in how we study the human body. By recreating the complexity of the "blood factory" using only human components, they have not only provided a new window into the biology of cancer but have also laid the groundwork for a more ethical, accurate, and personalized era of medical research. As this technology matures, it promises to reduce the reliance on animal models and accelerate the discovery of life-saving treatments for patients worldwide.














Leave a Reply