In a significant breakthrough for regenerative medicine and oncology, researchers at the University of Basel and University Hospital Basel have successfully engineered a complex, three-dimensional model of human bone marrow using exclusively human cells. This achievement, detailed in the journal Cell Stem Cell, represents the first time scientists have been able to replicate the intricate "blood factory" of the human body in a laboratory setting with such high fidelity. By simulating the specialized tissue that houses bone cells, nerves, and blood vessels, the research team has provided a powerful new tool for studying hematological diseases, testing new pharmaceuticals, and advancing the ethical goal of reducing reliance on animal experimentation.
The bone marrow is an extraordinarily complex organ, acting as the primary site for hematopoiesis—the process through which the body produces billions of new blood cells every day. Despite its vital role, the bone marrow often remains a "black box" in medical science because of its location within the hard interior of bones, making it difficult to observe in real-time within a living human subject. When this system malfunctions, as seen in various forms of leukemia and other blood cancers, the consequences are often fatal. For decades, the scientific community has sought a way to mirror this environment outside the human body to better understand how healthy blood production shifts into malignancy.
The Architecture of the Endosteal Niche
To understand the magnitude of this breakthrough, one must look at the specific microenvironments, or "niches," within the bone marrow. The research team, led by Professor Ivan Martin and Dr. Andrés García-García from the Department of Biomedicine, focused their efforts on the "endosteal niche." This specific zone, located near the inner surface of the bone, is considered the command center for blood stem cell regulation.
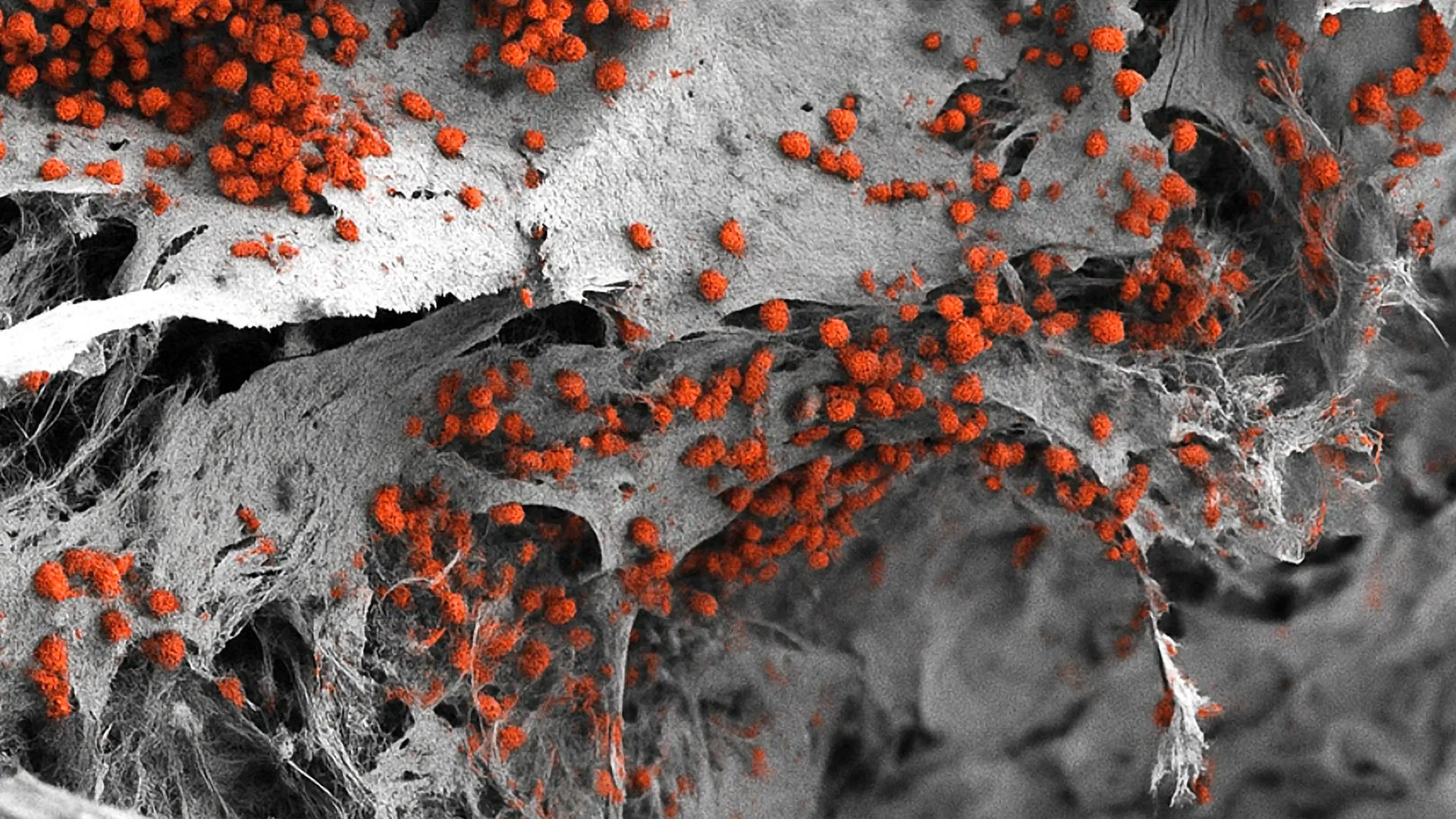
The endosteal niche is not merely a collection of cells; it is a highly coordinated ecosystem. It includes osteoblasts (bone-forming cells), vascular structures (blood vessels), immune cells, and a network of nerves. Crucially, this niche is often where cancer cells, such as those found in acute myeloid leukemia (AML), seek refuge from chemotherapy. By "hiding" in the endosteal niche, these malignant cells can remain dormant and resist treatment, leading to high rates of relapse. Previous laboratory models failed to incorporate all these disparate elements simultaneously, often relying on two-dimensional cultures or simplified systems that lacked the structural integrity of real bone tissue.
Technological Innovation: From Synthetic Scaffolds to Pluripotent Stem Cells
The construction of the model began with a sophisticated bioengineering approach. The researchers utilized a scaffold made of hydroxyapatite, a naturally occurring mineral form of calcium apatite that constitutes the bulk of human bone and tooth enamel. This mineral framework provided the necessary physical environment for the cells to attach and grow in three dimensions.
However, the true innovation lay in the cellular components. Rather than using donor tissue, which is often scarce and difficult to standardize, the team utilized human induced pluripotent stem cells (iPSCs). These are adult cells that have been molecularly reprogrammed to an embryonic-like state, giving them the ability to differentiate into any cell type in the human body. By applying specific chemical signals and growth factors, the researchers guided these stem cells to develop into the various specialized components of the bone marrow, including the stromal cells, vascular cells, and bone-forming cells required to populate the hydroxyapatite scaffold.
The resulting structure is a functional 3D system measuring approximately eight millimeters in diameter and four millimeters in thickness. While small to the human eye, this scale is significantly larger and more complex than previous "organ-on-a-chip" attempts. Most importantly, the model proved capable of maintaining the formation of human blood cells for several weeks, demonstrating that it is not just a structural replica but a functional biological unit.
A Chronology of Bone Marrow Research
The journey toward this human-only model has been decades in the making. The timeline of this field reflects a gradual shift from animal-centric models to human-centric bioengineering:
- 1950s–1980s: Early bone marrow research relied heavily on murine (mouse) models. These studies established the fundamental principles of hematopoiesis but often failed to translate to human clinical trials due to physiological differences between species.
- 1990s: The emergence of two-dimensional cell cultures allowed for the study of specific cell interactions, though these lacked the "niche" environment necessary to simulate complex diseases like cancer.
- 2006: Shinya Yamanaka’s discovery of induced pluripotent stem cells (iPSCs) provided the "raw material" needed for personalized tissue engineering.
- 2010s: Development of "bone-on-a-chip" technologies began, but these models usually lacked the full integration of nerves and varied vascular structures.
- 2024: The University of Basel team publishes their findings in Cell Stem Cell, marking the first successful integration of the endosteal niche’s full complexity using exclusively human-derived components.
Implications for the "3Rs" and Animal Welfare
One of the most profound impacts of this research is its potential to revolutionize the ethical landscape of medical science. The "3Rs" principle—Replacement, Reduction, and Refinement of animal testing—is a cornerstone of modern research ethics. Professor Ivan Martin emphasized that while mouse studies have provided a wealth of knowledge, they are not a perfect surrogate for human biology.
"Our model brings us closer to the biology of the human organism," Martin stated. He noted that the human-only system could serve as a vital complement to, and in many cases a replacement for, animal experiments. In Switzerland, where the University of Basel is located, there is significant public and institutional pressure to minimize animal testing. This model aligns with national initiatives to find high-tech alternatives that provide more accurate data for human patients while sparing animal lives.
Data and Supporting Analysis: Why Human Models Matter
The necessity for human-specific models is backed by rigorous data regarding the failure rates of new drugs. Historically, approximately 90% of drugs that pass animal testing fail in human clinical trials. A significant portion of these failures in the oncology sector is attributed to the "microenvironment effect"—where a drug effectively kills cancer cells in a petri dish or a mouse but fails in a human because the bone marrow niche protects the cancer cells.
By using the Basel model, pharmaceutical companies can test drug candidates in a simulated human environment before they ever reach a human volunteer. This could drastically reduce the cost of drug development, which currently averages over $2 billion per successful new drug, by identifying ineffective compounds much earlier in the pipeline.
Official Responses and Scientific Reception
The scientific community has responded with cautious optimism to the Basel study. Independent hematologists have noted that the inclusion of the nervous system within the model is a particularly impressive feat. Nerves are known to play a regulatory role in the release of stem cells into the bloodstream, a process that is often disrupted in patients with leukemia.
Dr. Andrés García-García highlighted that while the model is a massive leap forward, it is currently optimized for biological study rather than high-throughput drug screening. "For this specific purpose [drug testing], the size of our bone marrow model might be too large," he explained. The next phase of research will likely involve "scaling down" the model to a micro-scale format that would allow researchers to test hundreds of different drug combinations simultaneously on a single plate.
The Future of Personalized Medicine
The ultimate goal of this research is the realization of truly personalized medicine. In the future, a patient diagnosed with a blood disorder could have their own cells used to create a "personalized bone marrow model." Doctors could then use this model to test various chemotherapy cocktails or immunotherapy treatments to see which one most effectively targets that specific patient’s cancer within their unique bone marrow environment.
This "avatar" approach would eliminate the trial-and-error period that many cancer patients currently endure, potentially saving lives by identifying the most effective treatment immediately. Furthermore, the model could be used to study rare genetic blood disorders where animal models do not exist, providing hope for patients who have previously been overlooked by mainstream pharmaceutical research.
Broader Impact on Hematology and Beyond
The success of the University of Basel’s bone marrow model sets a precedent for the engineering of other complex tissues. If the bone marrow—one of the most dynamic and protected tissues in the body—can be replicated, it opens the door for similar advancements in replicating the blood-brain barrier, the liver’s metabolic niches, or the complex microenvironments of the lungs.
As the global medical community shifts toward more ethical and human-relevant research methodologies, the Basel model stands as a testament to the power of combining stem cell biology with advanced bioengineering. While further refinements are necessary to integrate the model into standard clinical practice, the foundation has been laid for a future where the "blood factory" of the human body is no longer a mystery, but a controllable and treatable biological system.














Leave a Reply