In a landmark achievement for regenerative medicine and hematology, a multidisciplinary team of scientists at the University of Basel and University Hospital Basel has successfully engineered a functional, three-dimensional model of human bone marrow using exclusively human cells. This breakthrough, recently detailed in the prestigious journal Cell Stem Cell, represents the first time that the intricate "blood factory" of the human body—complete with bone cells, neural networks, and vascular structures—has been replicated in a laboratory environment without the use of animal-derived components. Led by Professor Ivan Martin and Dr. Andrés García-García of the Department of Biomedicine, the research offers a sophisticated new platform for studying blood-related pathologies, testing pharmaceutical efficacy, and moving toward a future of personalized oncology.
The Vital Complexity of the Human Bone Marrow
The bone marrow is one of the most complex and dynamic tissues in the human body. Functioning as a centralized "blood factory," it is responsible for the continuous production of red blood cells, white blood cells, and platelets through a process known as hematopoiesis. Under normal physiological conditions, this tissue operates with remarkable efficiency, producing billions of new cells every day to replace those that have reached the end of their lifespan or have been lost to injury.
However, the marrow is also the site of devastating malignancies, such as leukemia and multiple myeloma. When the delicate balance of cell production is disrupted, the results are often fatal unless treated with aggressive therapies like chemotherapy or stem cell transplants. Historically, understanding the mechanisms behind these diseases has been hindered by the sheer complexity of the marrow’s microenvironment. It is not merely a collection of blood-forming cells; rather, it is a highly specialized architecture composed of mineralized bone, intricate blood vessels (vasculature), a network of nerves, and a variety of supporting stromal cells.
For decades, the scientific community has struggled to replicate this environment. While two-dimensional cell cultures provided basic insights, they failed to capture the spatial interactions and physical pressures that govern how blood cells mature and how cancer cells resist treatment. Animal models, primarily mice, have served as the gold standard for bone marrow research, but they possess inherent biological differences that often prevent findings from translating successfully to human clinical trials. The development of an all-human 3D model addresses these long-standing limitations.
Unlocking the Secrets of the Endosteal Niche
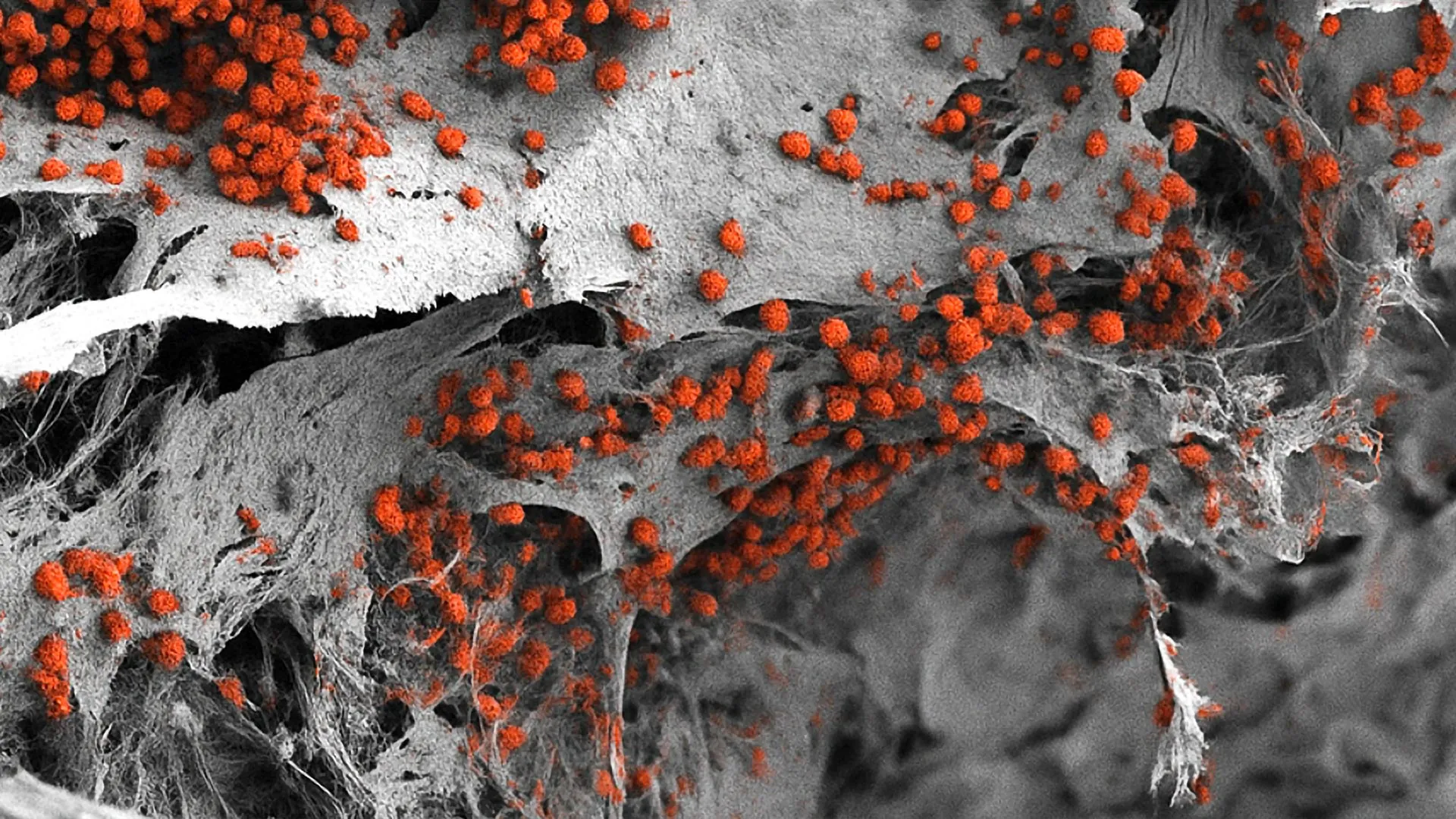
At the heart of the Basel team’s research is a specific microenvironment within the bone marrow known as the "endosteal niche." This region, located near the inner surface of the bone, is of particular interest to oncologists and hematologists. The endosteal niche acts as a sanctuary for hematopoietic stem cells—the "mother cells" of all blood types. Crucially, research has shown that blood cancer cells can hijack this niche, using its protective environment to hide from chemotherapy and remain dormant before causing a relapse.
Recreating the endosteal niche required the integration of multiple cell types that rarely interact in traditional lab settings. The researchers began their process with a synthetic scaffold made of hydroxyapatite. Hydroxyapatite is a naturally occurring mineral form of calcium apatite that makes up the primary inorganic constituent of human bone and tooth enamel. By using this material, the team provided the structural and chemical cues necessary for bone cells to thrive.
The true innovation, however, lay in the cellular source. The team utilized human induced pluripotent stem cells (iPSCs). These are adult cells that have been molecularly reprogrammed to an embryonic-like state, giving them the potential to differentiate into any cell type in the body. By carefully controlling the signals provided to these iPSCs, the researchers guided them to develop into the various components of the niche: osteoblasts (bone-forming cells), endothelial cells (which form blood vessels), and mesenchymal cells (which provide structural support and neural signaling).
Chronology of Development and Technical Specifications
The development of this model was the culmination of years of iterative experimentation within the Department of Biomedicine at the University of Basel. The project evolved through several critical phases:
- Scaffold Optimization (Early Phase): The team evaluated various porous materials to host the cell cultures, eventually settling on hydroxyapatite due to its bio-compatibility and its ability to mimic the rigidity of the human skeletal system.
- Cellular Differentiation Protocols (Mid Phase): Significant time was invested in perfecting the "cocktail" of growth factors and signaling molecules required to transform iPSCs into a multi-lineage tissue. Unlike previous models that added different cell types separately, the Basel model allowed these cells to develop and organize themselves somewhat autonomously within the 3D scaffold.
- Vascularization and Innervation (Final Phase): The most recent and complex step involved ensuring that the model included functional blood vessel-like structures and neural elements. This was essential for mimicking the way the marrow communicates with the rest of the body.
The resulting tissue "organoid" is significantly larger and more robust than previous attempts at bone marrow modeling. Measuring approximately eight millimeters in diameter and four millimeters in thickness, the model is visible to the naked eye and provides a substantial volume of tissue for analysis. In laboratory tests, the system demonstrated its functionality by maintaining the production and maturation of human blood cells for several weeks, a feat that traditional cultures cannot achieve.
Supporting Data and Comparative Analysis
Data published in the study highlights the superiority of this 3D human model over traditional 2D systems. Genetic analysis (RNA sequencing) of the cells within the model showed that they expressed markers nearly identical to those found in native human bone marrow. Specifically, the mesenchymal stromal cells in the model exhibited the same transcriptional signatures as cells harvested from healthy human donors, suggesting that the laboratory environment successfully "tricked" the cells into behaving as they would inside a living body.
Furthermore, when the researchers introduced hematopoietic stem cells into the model, they observed a high degree of "homing"—a process where stem cells find their way to their protective niches. In 2D cultures, stem cells often differentiate too quickly or lose their regenerative potential. In the Basel 3D model, the stem cells remained in a "quiescent" or dormant state, which is exactly how they behave in the human body to ensure a long-term supply of blood cells. This specific finding is critical for cancer research, as dormant cells are the most difficult to kill with standard treatments.
Official Responses and Scientific Implications
The scientific community has reacted with cautious optimism to the Basel report. Dr. Andrés García-García, a lead author of the study, emphasized the potential for drug discovery. "Our platform allows us to observe how drugs interact with the bone marrow environment in a way that was previously impossible without using animal subjects," García-García stated. However, he also noted a logistical challenge: "For high-throughput drug testing, where thousands of compounds are screened simultaneously, the current size of our model—eight millimeters—might be too large and resource-intensive. Our next step is to miniaturize the system to fit into standard lab plates while maintaining its biological complexity."
Professor Ivan Martin, who has long been a proponent of the "3R" principle (Replace, Reduce, Refine) in animal experimentation, highlighted the ethical and scientific benefits. "While mouse models have provided the foundation for our understanding of hematology, they are not humans. There are subtle but vital differences in how human blood cells interact with their environment. Our model brings us closer to the actual biology of the human organism," Martin explained.
The University of Basel has indicated that this research aligns with a broader institutional commitment to reducing the reliance on animal testing. By providing a reliable human-based alternative, the university hopes to lead a shift in how preclinical trials are conducted, potentially lowering the costs and increasing the success rates of new drug candidates.
Impact on Personalized Medicine and Oncology
The long-term vision for this technology extends into the realm of personalized medicine. Currently, patients with blood cancers often undergo a trial-and-error approach to treatment, where doctors prescribe standard chemotherapy regimens and wait to see if the patient responds. If the cancer returns, it is often because a small population of cells survived within the marrow’s niches.
With the Basel model, researchers envision a future where a small sample of a patient’s own cells is used to grow a personalized "marrow avatar" in the lab. This avatar would contain the patient’s specific genetic makeup and their specific cancer cells. Doctors could then test dozens of different drug combinations on this lab-grown tissue to see which one most effectively clears the cancer without damaging the healthy stem cells. This would allow for a bespoke treatment plan tailored to the individual, potentially saving lives and reducing the grueling side effects of ineffective therapies.
Broader Economic and Regulatory Context
Beyond the clinical applications, the development of a fully human bone marrow model has significant economic implications for the global pharmaceutical industry. The cost of bringing a new drug to market currently exceeds $2 billion, with a high percentage of drugs failing in human trials despite successful results in animal studies. This "translational gap" is a major bottleneck in medical innovation.
Regulatory bodies, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have recently begun to signal a willingness to accept data from "organ-on-a-chip" and complex 3D human models in lieu of some animal data. The Basel model stands as a prime candidate for this transition. By providing more accurate human data earlier in the development process, pharmaceutical companies could identify toxic or ineffective drugs years before they reach expensive clinical trials.
Conclusion and Future Directions
The creation of a functional, 3D human bone marrow model marks a turning point in hematological research. By successfully integrating bone, blood vessels, and nerves into a single human-cell-based system, the researchers at the University of Basel have provided a new lens through which we can view the "blood factory" of the human body.
While the model is currently a research tool, the path toward clinical and industrial application is clear. Future efforts will focus on scaling down the technology for mass drug screening and refining the "innervation" of the model to better understand how stress and the nervous system influence blood production. As the scientific community continues to move away from animal models in favor of more accurate human analogs, the Basel bone marrow model will likely serve as a foundational technology for the next generation of cancer treatments and regenerative therapies. This study does not just represent a technical success; it represents a fundamental shift toward a more ethical, accurate, and personalized era of medical science.















Leave a Reply