A groundbreaking discovery by a research team spearheaded by Universitat Autònoma de Barcelona (UAB) scientist David Reverter has illuminated the intricate molecular mechanism governing bacterial cell division, a fundamental process for all life. The study, published in the prestigious journal Nature Communications, pinpoints the MraZ protein as the pivotal regulator, revealing its precise method of binding to the dcw gene cluster to orchestrate this vital biological event. This seminal work not only clarifies a long-standing question in microbiology but also offers profound implications for understanding bacterial growth, development, and the potential for novel antimicrobial strategies.
The Genesis of Bacterial Proliferation: Understanding the dcw Operon
Cell division, the process by which a single cell gives rise to two or more daughter cells, is an indispensable cornerstone of existence for all living organisms. In the microbial world, this mechanism is a tightly regulated symphony of protein interactions and genetic instructions. For the vast majority of bacteria, the genetic blueprints for cell division and the construction of their protective cell walls are consolidated within a specialized genetic unit known as the dcw operon. This operon, a cluster of genes transcribed as a single messenger RNA molecule, contains the essential information required to synthesize the proteins that drive bacterial reproduction and maintain cellular integrity.
The activation of genes within the dcw operon is a carefully orchestrated event, initiated by specialized proteins called transcription factors. These molecular architects bind to specific DNA sequences upstream of the genes they regulate, known as promoter regions. These promoters act as signal flags, indicating the precise starting point for the transcription machinery to begin copying the genetic information into messenger RNA, the precursor for protein synthesis.
Central to this regulatory cascade is the MraZ protein. Intriguingly, MraZ itself is the very first gene encoded within the dcw operon in nearly all bacterial species. This autoregulatory feedback loop means that MraZ plays a dual role: it is both a product of the operon it controls and its primary activator. Upon its synthesis, MraZ binds to the dcw operon’s promoter, effectively switching on the downstream genes. This initiates the production of the suite of proteins necessary for bacteria to divide and proliferate, positioning MraZ as the master switch for cell division across a broad spectrum of bacterial life.
Visualizing the Molecular Ballet: Advanced Techniques Reveal MraZ’s Mechanism
The UAB research group, under the leadership of Professor David Reverter from the Department of Biochemistry and Molecular Biology and a researcher at the Institute of Biotechnology and Biomedicine of the UAB (IBB-UAB), has achieved a significant breakthrough by elucidating the precise molecular dance between MraZ and the dcw operon. Employing state-of-the-art structural biology techniques, including X-ray crystallography and cryo-electron microscopy (cryo-EM), the team was able to generate high-resolution images of this critical interaction.
These advanced imaging modalities were instrumental in revealing how the MraZ transcription factor engages with the promoter region of the dcw operon in the bacterium Mycoplasma genitalium. This microorganism, chosen for its exceptionally compact genome, serves as a valuable model system for dissecting fundamental cellular processes. The relative simplicity of M. genitalium‘s genetic makeup allows researchers to isolate and study key molecular mechanisms with greater clarity.
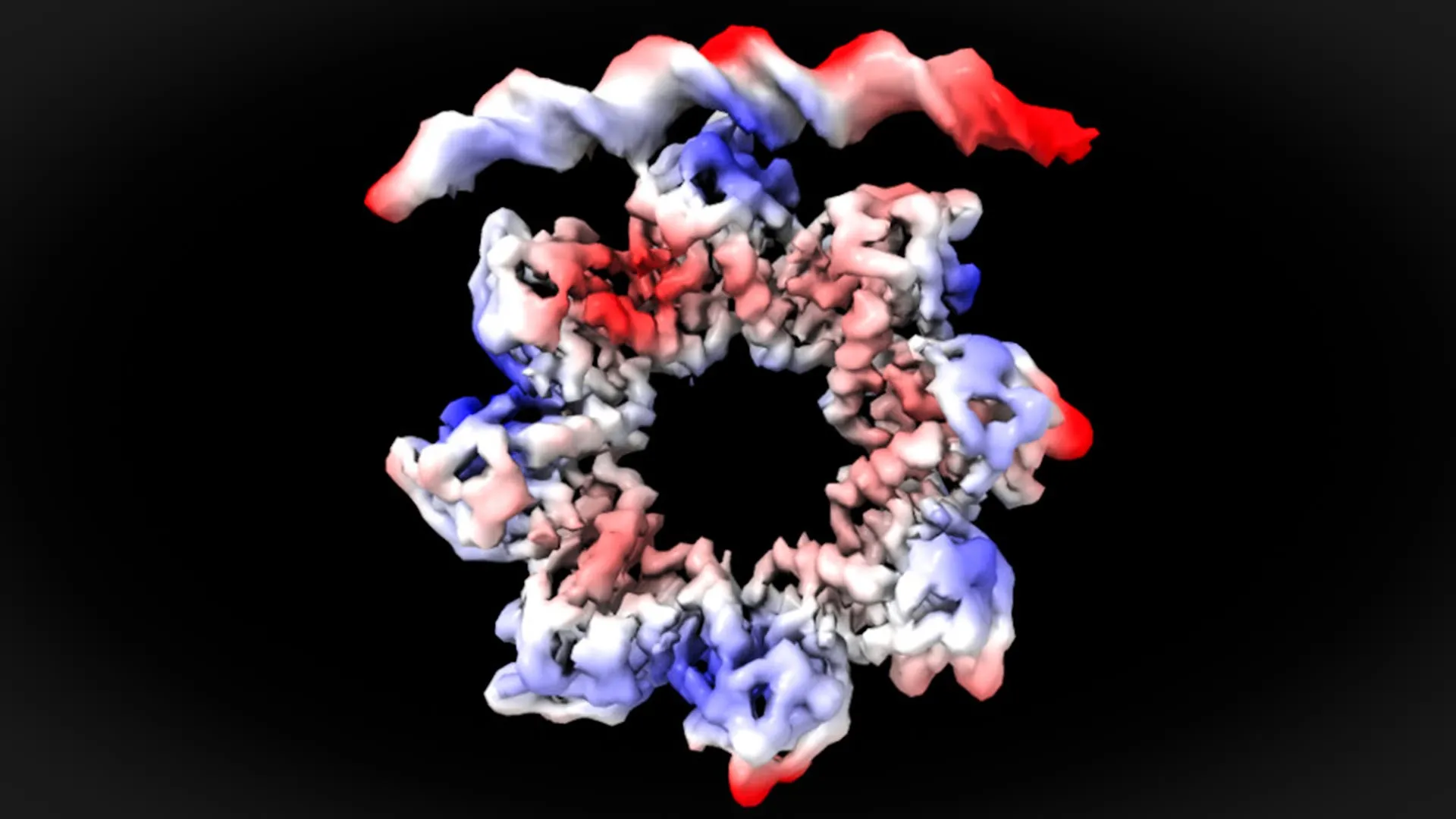
Atomic-Level Precision: The Deformable Donut of MraZ
The promoter region of the dcw operon is characterized by a specific DNA sequence comprising four repetitive segments, often referred to as "boxes." Each of these boxes is approximately six nucleotides long and plays a crucial role in dictating the binding affinity and specificity of transcription factors. It is within these repeated DNA sequences that MraZ exerts its regulatory control.
Through the application of cryo-electron microscopy, the researchers were able to achieve near-atomic resolution, offering an unprecedented glimpse into the interaction between the MraZ protein and the DNA bases within these four repetitive boxes. The findings revealed a surprising conformational change within the MraZ protein that is essential for its successful binding to the operon.
"This is a surprising observation," stated David Reverter. "The MraZ protein is typically an octamer, meaning it is composed of eight identical subunits arranged in a donut-like structure. However, its inherent curvature would seemingly preclude it from efficiently binding to the four distinct ‘boxes’ of the promoter. Yet, to regulate cell division, we observe how this donut-like structure breaks and deforms. This remarkable plasticity allows four of its subunits to precisely engage with the four promoter ‘boxes’." This dynamic structural adaptation underscores the sophisticated molecular engineering that underpins bacterial cell division.
A Paradigm Shift in Understanding Bacterial Reproduction
The ability to directly visualize the atomic-level interaction between MraZ and the promoter DNA that initiates cell division represents a significant leap forward in the field. Prior to this study, researchers relied heavily on indirect methods, such as biochemical assays and computational modeling, to infer the mechanics of this regulatory process. While these approaches provided valuable insights, they lacked the direct, visual confirmation now afforded by structural biology.
The implications of this discovery extend far beyond a single bacterial species. Professor Reverter posited that the regulatory system identified in this study is likely conserved across a vast majority of bacteria. "This mechanism is universal to most bacteria," he explained, "because all MraZ proteins exhibit remarkable similarity in their structure, maintaining the same octameric donut formation. Furthermore, the DNA sequences of the promoters controlling cell division operons are also highly conserved. This suggests that our findings have broad applicability and could unlock our understanding of cell division in countless bacterial species."
A Collaborative Endeavor: Forging International Scientific Partnerships
This pivotal research was a testament to robust international collaboration. The study was led by David Reverter’s dedicated team at the Institute of Biotechnology and Biomedicine and the Department of Biochemistry and Molecular Biology at UAB. Crucially, the research benefited from the specialized capabilities of the ALBA synchrotron, a cutting-edge light source facility, and the advanced cryo-electron microscopy services provided by the Institute of Genetics and Molecular and Cellular Biology in Strasbourg, France. The synergy between these institutions and their respective expertise was indispensable in achieving the high-resolution structural data that underpins this discovery.
Broader Implications: From Fundamental Biology to Antimicrobial Innovation
The precise understanding of how MraZ controls bacterial cell division opens up new avenues for research and potential applications.
Implications for Fundamental Microbiology:
- Elucidating Bacterial Development: This discovery provides a critical piece of the puzzle in understanding the complex developmental pathways of bacteria. Knowing how cell division is initiated allows scientists to better investigate how bacteria grow, form colonies, and adapt to diverse environments.
- Comparative Genomics and Evolution: The universality of the MraZ- dcw operon interaction suggests it is an ancient and highly conserved mechanism. Further research can explore the evolutionary trajectory of this system across different bacterial lineages, shedding light on bacterial diversification and adaptation.
- Understanding Biofilm Formation: Bacterial biofilms, communities of microorganisms encased in a self-produced matrix, are often associated with infections and medical device contamination. Cell division is a fundamental process within biofilms, and understanding its regulation could provide insights into how these structures are formed and maintained.
Implications for Human Health and Medicine:
- Novel Antimicrobial Targets: The MraZ protein, as a key regulator of bacterial proliferation, represents a compelling target for the development of new antimicrobial drugs. By inhibiting MraZ’s function, it may be possible to selectively halt bacterial growth without adversely affecting human cells. This is particularly significant in an era of increasing antibiotic resistance, where novel therapeutic strategies are urgently needed.
- Developing Resistance-Breaking Therapies: Current antibiotics often target bacterial cell wall synthesis or protein synthesis. By targeting cell division directly through a regulator like MraZ, researchers could develop entirely new classes of antibiotics that bypass existing resistance mechanisms.
- Diagnostic Tools: While speculative at this stage, a deeper understanding of MraZ regulation could potentially inform the development of novel diagnostic tools for identifying bacterial infections or monitoring treatment efficacy.
Challenges and Future Directions:
Despite the significant progress, several questions remain. Future research will likely focus on:
- Kinetic Studies: While the structure of MraZ binding has been elucidated, understanding the kinetics—the speed and dynamics of the binding and release process—will be crucial for fully appreciating its regulatory role.
- In Vivo Validation: While the M. genitalium model is powerful, validating these findings in a broader range of bacterial species, including those with pathogenic potential, will be essential.
- Drug Discovery Efforts: Translating this fundamental discovery into tangible therapeutic applications will require extensive drug discovery and development programs, including high-throughput screening for compounds that can modulate MraZ activity.
The work by David Reverter and his international collaborators represents a significant stride in our comprehension of bacterial life. By unraveling the molecular intricacies of MraZ-mediated cell division, this research not only advances fundamental biological knowledge but also illuminates promising pathways for addressing pressing global health challenges, particularly the escalating crisis of antibiotic resistance. The precise choreography of bacterial proliferation, once a molecular mystery, is now coming into sharp, atomic-level focus.















Leave a Reply