Researchers from the Department of Medicine at the School of Clinical Medicine, LKS Faculty of Medicine, University of Hong Kong (HKUMed), have announced a landmark discovery in the field of osteology and regenerative medicine. The team has successfully identified a specific protein, Piezo1, which functions as the body’s internal "exercise sensor," playing a fundamental role in how physical activity maintains bone density and strength. This breakthrough, published in the prestigious international journal Signal Transduction and Targeted Therapy, offers a potential paradigm shift in the treatment of osteoporosis, particularly for aging populations and individuals with limited mobility who are unable to engage in traditional physical exercise.
The study provides a molecular explanation for the long-observed link between mechanical loading—such as walking, running, or weight-bearing activities—and bone health. By uncovering the signaling pathways controlled by Piezo1, the research team has opened the door to the development of "exercise mimetics." These are pharmacological interventions designed to replicate the biological benefits of physical movement at the cellular level, offering a lifeline to bedridden patients, those with chronic physical disabilities, and the elderly who face a high risk of debilitating fractures.
The Global and Local Burden of Osteoporosis
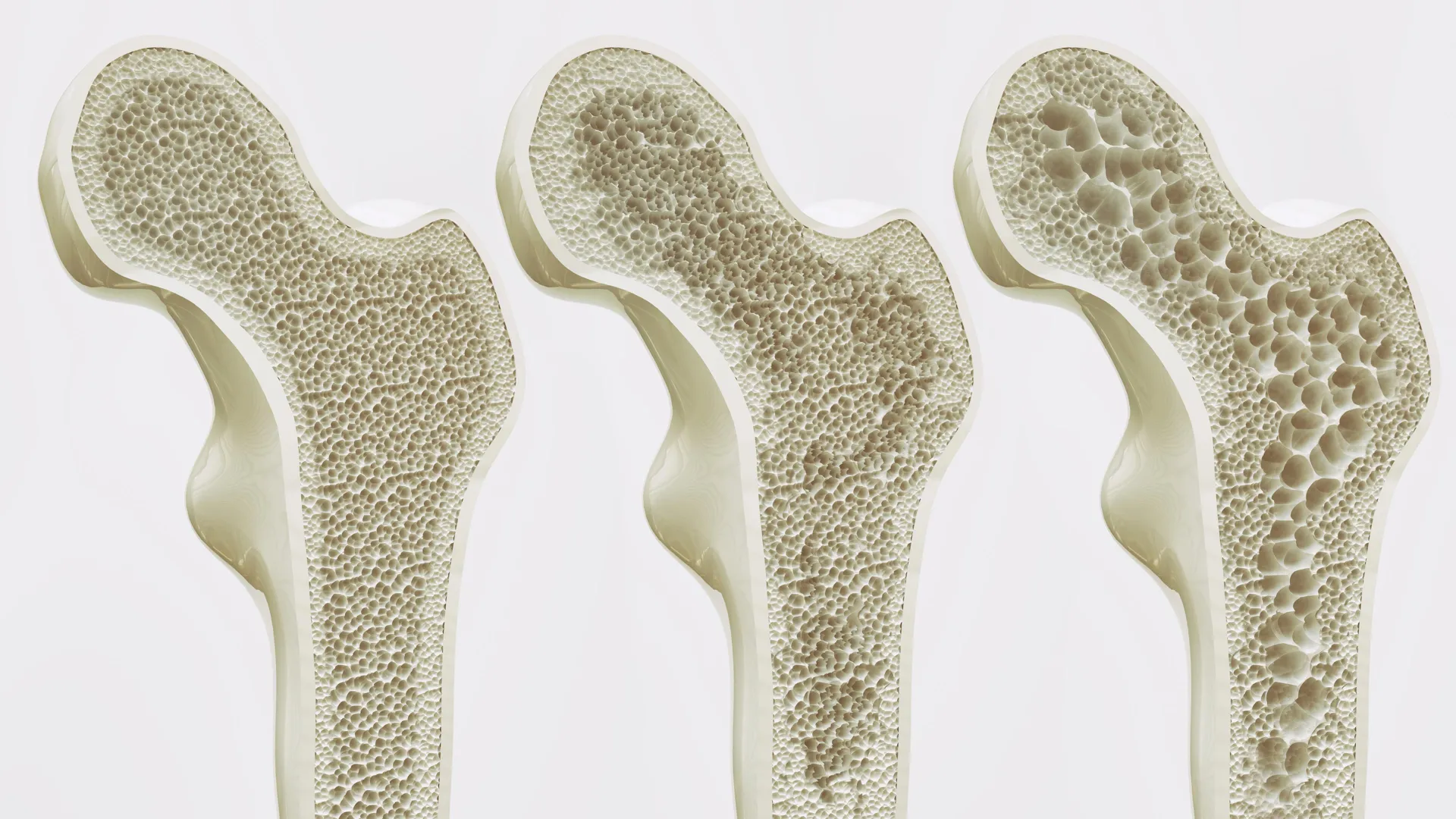
Osteoporosis is often characterized by the medical community as a "silent epidemic" because bone loss typically occurs without symptoms until a fracture happens. The scale of the problem is vast; data from the World Health Organization (WHO) indicates that approximately one in three women and one in five men over the age of 50 will suffer an osteoporotic fracture during their lifetime. These injuries are not merely physical setbacks; they are frequently precursors to a permanent loss of independence, chronic pain, and increased mortality rates.
In Hong Kong, the challenge is exacerbated by a rapidly aging demographic. Statistics reveal that osteoporosis affects approximately 45% of women and 13% of men aged 65 and older in the territory. As the population continues to age, the strain on the public healthcare system is expected to intensify. Hip fractures, the most severe consequence of weakened bones, often require surgery and long-term rehabilitation, placing a significant financial and resource burden on hospital authorities. The HKUMed study addresses this urgent public health need by seeking a preventative and therapeutic solution that transcends the requirement for physical exertion.
The Cellular Mechanics of Bone vs. Fat
To understand the significance of the Piezo1 discovery, it is necessary to examine the internal environment of the bone marrow. Within the marrow reside mesenchymal stem cells (MSCs), which are versatile progenitor cells capable of differentiating into various types of tissue. In a healthy, active individual, these stem cells are naturally inclined to become osteoblasts—cells responsible for bone formation. This process is driven by the mechanical stress placed on the skeleton during movement.
However, the aging process and sedentary lifestyles disrupt this delicate balance. As people age or become less active, MSCs undergo a "lineage switch." Instead of forming bone, they increasingly differentiate into adipocytes, or fat cells. This accumulation of marrow fat is a hallmark of osteoporosis. As fat cells crowd out bone-forming cells, the structural integrity of the bone diminishes, making it porous, brittle, and susceptible to breakage. Current pharmaceutical treatments, such as bisphosphonates or hormone-related therapies, focus primarily on slowing bone resorption, but they often struggle to reverse the underlying shift from bone formation to fat accumulation.
Piezo1: The Molecular Exercise Sensor
The research team, led by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology at HKUMed, utilized advanced mouse models and human stem cell cultures to pinpoint the mechanism behind this cellular shift. They identified Piezo1, a mechanosensitive ion channel protein located on the surface of mesenchymal stem cells.
Piezo1 acts as a transducer, converting mechanical pressure from physical activity into chemical signals within the cell. The study found that when Piezo1 is activated—either through physical movement or laboratory stimulation—it sends signals that actively suppress the formation of fat cells while simultaneously promoting the development of bone-forming osteoblasts.
In experimental models where Piezo1 was removed or inactivated, the researchers observed a rapid deterioration of bone quality. Without the protein to sense movement, the stem cells defaulted to fat production, even if the subjects were active. Furthermore, the absence of Piezo1 triggered the release of specific inflammatory markers, namely Ccl2 and lipocalin-2. These signals were found to create a pro-inflammatory environment that further inhibited bone growth and accelerated the aging of the bone marrow environment. By blocking these inflammatory signals in the lab, researchers were able to partially mitigate bone loss, confirming that Piezo1 serves as a master regulator of bone health.
International Collaboration and Research Chronology
The discovery is the result of a multi-year international collaboration. The project was co-led by Professor Xu Aimin and Dr. Wang Baile, Research Assistant Professor at HKUMed, in partnership with Professor Eric Honoré from the Institute of Molecular and Cellular Pharmacology at the French National Centre for Scientific Research (CNRS).
The timeline of the research reflects a rigorous progression from basic molecular biology to potential clinical application:
- Initial Phase: Researchers observed the correlation between sedentary behavior and marrow fat accumulation in aging populations.
- Discovery Phase: Screening of various mechanosensitive proteins led to the identification of Piezo1 as a primary actor in bone marrow MSCs.
- Validation Phase: Using CRISPR/Cas9 and other genetic tools, the team created models to observe the effects of Piezo1 deficiency.
- Mechanism Mapping: The team identified the Ccl2 and lipocalin-2 pathways as the specific inflammatory drivers of bone loss in the absence of Piezo1.
- Current Status: The findings have been published, and the team is now moving toward the identification of chemical compounds that can safely activate Piezo1 in humans.
Implications for Future Medical Treatment
The most profound implication of this study is the concept of "exercise in a pill." While the medical community has long advocated for "exercise as medicine," this discovery provides the molecular blueprint to make that a reality for those who cannot move.
Professor Xu Aimin emphasized that while current treatments are effective for many, they are often reactive rather than proactive. "Current treatments rely heavily on physical activity, which many patients simply cannot perform," Xu noted. "We have essentially decoded how the body converts movement into stronger bones. By activating the Piezo1 pathway, we can mimic the benefits of exercise, effectively tricking the body into thinking it is exercising."
This approach could revolutionize care for:
- Bedridden Patients: Individuals recovering from surgery or suffering from paralysis who cannot perform weight-bearing exercises to maintain bone mass.
- The Frail Elderly: Seniors for whom high-impact exercise is dangerous due to fall risks or cardiovascular limitations.
- Astronauts: Long-duration space flight causes significant bone density loss due to the lack of gravity (and thus lack of mechanical loading). Piezo1 activators could mitigate "space osteopenia."
Expert Analysis and Industry Response
Medical analysts suggest that the identification of Piezo1 provides a highly specific target for the pharmaceutical industry. Unlike systemic treatments that can have widespread side effects, a therapy targeting the Piezo1 pathway in bone marrow could potentially offer a more localized and efficient treatment for bone density issues.
Dr. Wang Baile highlighted the social impact of the research, noting that maintaining bone mass is synonymous with maintaining independence. "Our findings open the door to developing drugs that chemically activate the Piezo1 pathway to help maintain bone mass and support independence for those who are most vulnerable," Wang said.
The study also received praise from the international community. Professor Eric Honoré noted that this strategy moves beyond traditional physical therapy, offering a biological shortcut to skeletal health. The research has already garnered interest from biotechnology firms looking to develop small-molecule agonists for Piezo1.
Funding and Institutional Support
The comprehensive nature of the study was made possible through extensive funding from both local and international bodies. Support was provided by the Research Grants Council of Hong Kong, the Health Bureau of the HKSAR Government, and the National Natural Science Foundation of China. International backing came from the Human Frontier Science Program, the French National Research Agency, and several French medical foundations, highlighting the global importance of finding a solution to age-related bone deterioration.
As the team at HKUMed moves toward clinical trials, the focus will remain on the safety and efficacy of potential "exercise mimetics." While a pill will likely never replace the holistic benefits of a healthy, active lifestyle for those who are capable of it, for millions of people worldwide, the discovery of Piezo1 represents a significant step toward a future where a fracture is no longer an inevitable consequence of aging.














Leave a Reply