A groundbreaking study led by scientists at the Universitat Autònoma de Barcelona (UAB) has elucidated the intricate molecular mechanism governing bacterial cell division, a fundamental process for all life. The research, published in the prestigious journal Nature Communications, identifies the MraZ protein as a pivotal regulator, precisely controlling the activation of the dcw gene cluster, which dictates the essential steps of bacterial reproduction and cell wall construction. This discovery marks a significant leap forward in our understanding of bacterial biology, with potential implications for antibiotic development and microbial engineering.
The Essential Blueprint for Bacterial Reproduction: The dcw Operon
For bacteria to thrive and multiply, they must execute a complex and tightly regulated process of cell division. This biological ballet involves the coordinated action of numerous proteins and regulatory elements. At the heart of this intricate system in most bacterial species lies the dcw operon, a compact genetic unit that houses the crucial instructions for building the bacterial cell and orchestrating its division. The dcw operon is not merely a collection of genes; it represents a master control panel, ensuring that the necessary cellular machinery is synthesized at the right time and in the correct quantities.
The genes within this operon are activated by specialized proteins called transcription factors. These molecular scouts bind to specific DNA sequences known as promoters, which serve as the starting points for gene expression. The promoter region is a critical signaling hub, essentially telling the cellular machinery where to begin reading the genetic code to produce proteins. In the case of the dcw operon, one particular transcription factor, MraZ, stands out. Significantly, MraZ is also the very first gene within the dcw operon itself, forming a self-regulatory loop. When MraZ is activated, it initiates the transcription of the entire operon, leading to the production of proteins indispensable for bacterial division. Consequently, MraZ acts as the master switch, dictating the pace and initiation of cell division across a vast spectrum of bacterial species.
Visualizing the Molecular Dance: Advanced Imaging Techniques Uncover MraZ’s Mechanism
The UAB research team, under the leadership of Professor David Reverter from the Department of Biochemistry and Molecular Biology and a researcher at the Institute of Biotechnology and Biomedicine of the UAB (IBB-UAB), has now provided an unprecedented, high-resolution view of this regulatory process. Their investigation employed cutting-edge structural biology techniques, including X-ray crystallography and cryo-electron microscopy (cryo-EM). These powerful tools allowed the scientists to peer into the molecular interactions with remarkable clarity, revealing how the MraZ protein engages with the promoter of the dcw operon in Mycoplasma genitalium.
Mycoplasma genitalium was chosen for this study due to its exceptionally small genome, which simplifies genetic analysis and makes it a valuable model organism for fundamental research in bacterial genetics. The choice of this microorganism allowed the researchers to focus on the core regulatory mechanisms without the complexity of a larger, more elaborate genetic architecture.
Atomic Precision: The Dynamic Deformation of MraZ
The promoter region of the dcw operon, as revealed by the study, possesses a unique structure. It features four repeating segments, often referred to as "boxes," each consisting of six nucleotides. These repeating DNA sequences are crucial for their role in regulating transcription initiation.
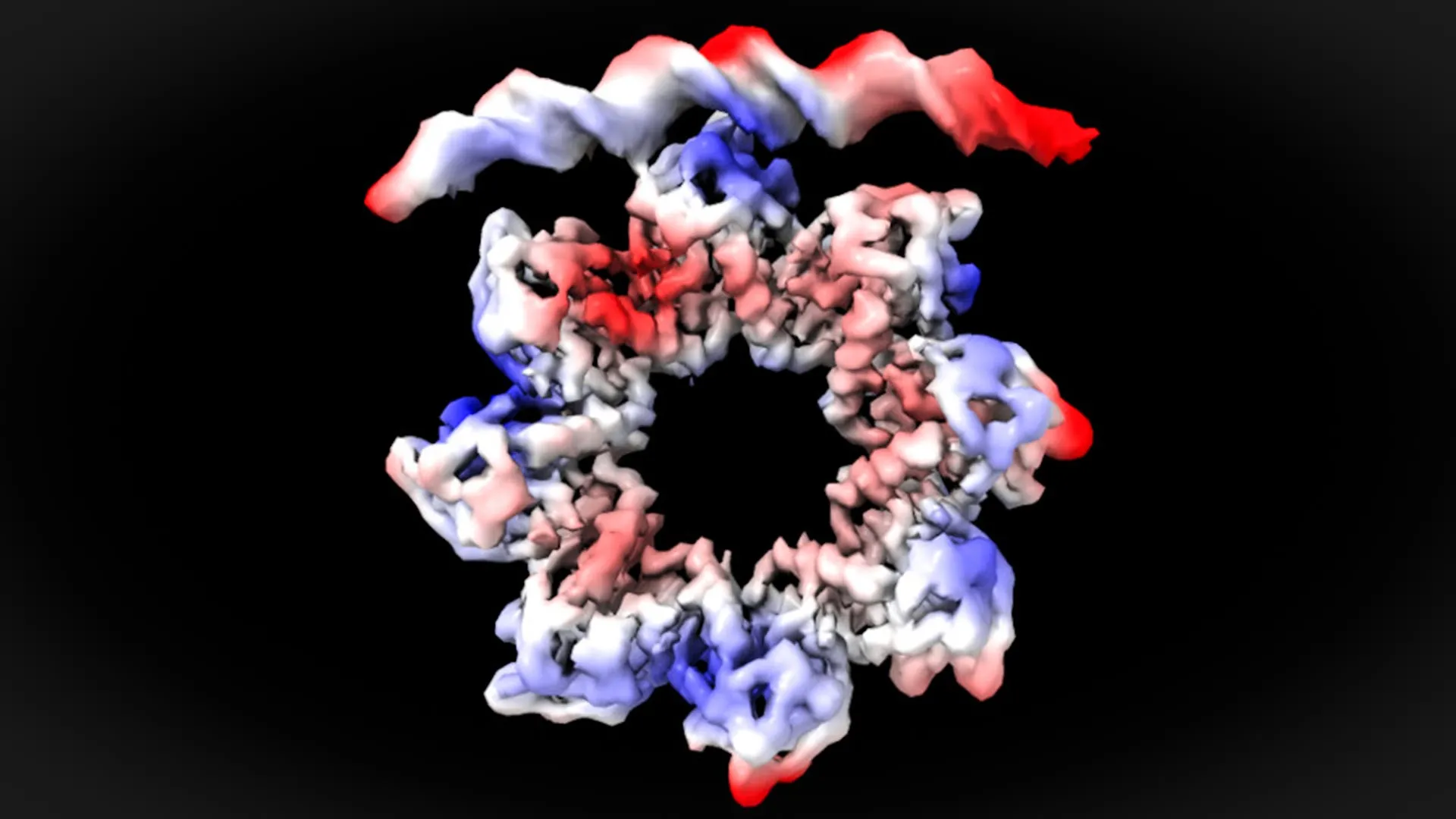
Through meticulous analysis using cryo-electron microscopy, the UAB team achieved near-atomic resolution of the interaction between the MraZ protein and the DNA bases within these four repeated boxes. The findings were not only precise but also profoundly surprising. They observed that for MraZ to effectively bind to the promoter and initiate gene expression, it must undergo a significant and dynamic structural transformation.
Professor David Reverter explained the remarkable nature of this observation: "This is a surprising observation. The MraZ protein is an octamer formed by eight identical subunits joined in the shape of a donut, but with a curvature that would never allow the union with the four ‘boxes’ of the promoter. However, to regulate cell division we see how the donut breaks and deforms in such a way that four of the subunits can join the four boxes of the promoter." This "breaking and deforming" of the donut-like structure is key. It suggests that MraZ is not a rigid molecule but a highly adaptable one, capable of reconfiguring itself to achieve the precise molecular handshake required for gene activation. This dynamic conformational change is critical for its function as a transcription factor.
A Paradigm Shift in Understanding Bacterial Regulation
The ability to directly visualize the molecular interaction between MraZ and the promoter DNA that triggers cell division represents a significant breakthrough. Historically, researchers relied heavily on indirect methods, such as biochemical experiments and sophisticated computer modeling, to infer the workings of this complex regulatory system. While these approaches provided valuable insights, they lacked the direct, visual confirmation that structural biology now offers. The UAB study provides definitive evidence of the physical mechanism at play, moving beyond inference to direct observation.
The implications of this finding extend far beyond Mycoplasma genitalium. Professor Reverter posits that this regulatory system is likely conserved and widespread across the bacterial kingdom. "This is universal to most bacteria," he stated, "since all MraZ proteins are very similar, have the same octamer structure, and the DNA sequences of the promoters of the operons that regulate cell division are also similar." This universality suggests that the principles uncovered in this study could apply to a broad range of bacteria, including many that are medically important or play significant roles in ecological processes.
A Timeline of Discovery and International Collaboration
The research journey leading to this significant publication involved a concerted effort over a period of several years, culminating in the recent publication in Nature Communications. The initial stages likely involved identifying the dcw operon and its crucial role in bacterial cell division, a well-established area of microbiology. Subsequent research focused on pinpointing the specific transcription factors involved, with MraZ emerging as a key candidate due to its position within the operon and its presumed regulatory function.
The critical phase of the research involved the application of advanced structural biology techniques. X-ray crystallography, a method used to determine the atomic and molecular structure of a crystal, likely provided initial insights into the overall structure of MraZ. However, the dynamic nature of the MraZ-DNA interaction, particularly the conformational changes, necessitated the higher resolution and capability of cryo-electron microscopy. Cryo-EM allows for the imaging of biological molecules in a near-native, frozen state, providing detailed structural information of complexes that may be difficult to crystallize.
The collaboration underpinning this study highlights the interconnected nature of modern scientific research. The UAB team, comprising experts in biochemistry and molecular biology, joined forces with international partners to leverage specialized facilities. The ALBA synchrotron, a facility that produces intense beams of X-rays, likely played a role in the crystallographic aspects of the research, enabling detailed structural analysis. Furthermore, the cryo-electron microscopy service at the Institute of Genetics and Molecular and Cellular Biology in Strasbourg, France, provided access to state-of-the-art equipment essential for visualizing the MraZ-DNA complex at unprecedented resolution. This international synergy was crucial for achieving the comprehensive understanding presented in the study.
Broader Implications: Antibiotic Development and Microbial Engineering
The detailed understanding of how MraZ regulates bacterial cell division carries significant implications for several fields of science and medicine.
-
Antibiotic Development: Bacterial cell division is a prime target for antimicrobial therapies. Many existing antibiotics, such as penicillin, function by disrupting the synthesis of the bacterial cell wall, a process intimately linked to cell division. By understanding the precise molecular machinery that initiates and controls this process, researchers can identify novel targets for the development of new antibiotics. If MraZ or its interaction with the dcw operon can be inhibited, it could effectively halt bacterial growth and replication, offering a new avenue for combating antibiotic-resistant bacteria, a growing global health crisis. The universality of the MraZ system across bacteria further amplifies its potential as a broad-spectrum antibiotic target.
-
Microbial Engineering and Synthetic Biology: Beyond its therapeutic potential, this research could also be instrumental in the field of microbial engineering. For scientists aiming to engineer bacteria for specific industrial or biotechnological purposes, such as the production of biofuels, pharmaceuticals, or enzymes, precise control over bacterial growth and division is paramount. A deep understanding of the regulatory networks, like the one governed by MraZ, allows for more sophisticated genetic engineering strategies to optimize bacterial strains for enhanced productivity or specific functionalities.
-
Fundamental Understanding of Life: At its core, this discovery deepens our fundamental understanding of how life perpetuates itself. Cell division is a universal biological process, and unraveling its intricate molecular mechanisms in bacteria provides foundational knowledge that can be applied to understanding similar processes in other organisms, potentially including eukaryotes. The dynamic nature of protein-DNA interactions revealed in this study also contributes to the broader field of molecular biology, shedding light on the plasticity and adaptability of biological molecules.
The research team’s meticulous work, leveraging advanced imaging techniques and international collaboration, has not only solved a long-standing puzzle in bacterial biology but has also opened new doors for scientific innovation. The identification of MraZ’s dynamic role in orchestrating bacterial cell division is a testament to the power of structural biology and collaborative research in pushing the boundaries of our knowledge.















Leave a Reply