In the enduring debate over how the human brain develops its capacity for memory and spatial navigation, the concept of the "tabula rasa," or blank slate, has long held sway in popular imagination. This theory suggests that we are born with a clean slate, and that every connection, every memory, and every learned behavior is written upon this void through experience. However, groundbreaking research from the Institute of Science and Technology Austria (ISTA) has provided a starkly different perspective. Led by Magdalena Walz Professor for Life Sciences Peter Jonas and ISTA alumnus Victor Vargas-Barroso, the team has demonstrated that the brain’s primary memory center, the hippocampus, actually begins as a "tabula plena"—a full slate—which is then meticulously refined and pruned into an efficient system.
The study, recently published in the prestigious journal Nature Communications, challenges the conventional "building block" model of neural development. Instead of starting with a few connections that multiply over time, the research reveals that the hippocampal network begins in a state of exuberant, almost chaotic connectivity. As the organism matures, the brain does not simply add more wires; it selectively removes the unnecessary ones. This process of refinement is what ultimately allows for the sophisticated storage and retrieval of complex information.
The Architecture of Memory: Understanding the CA3 Circuit
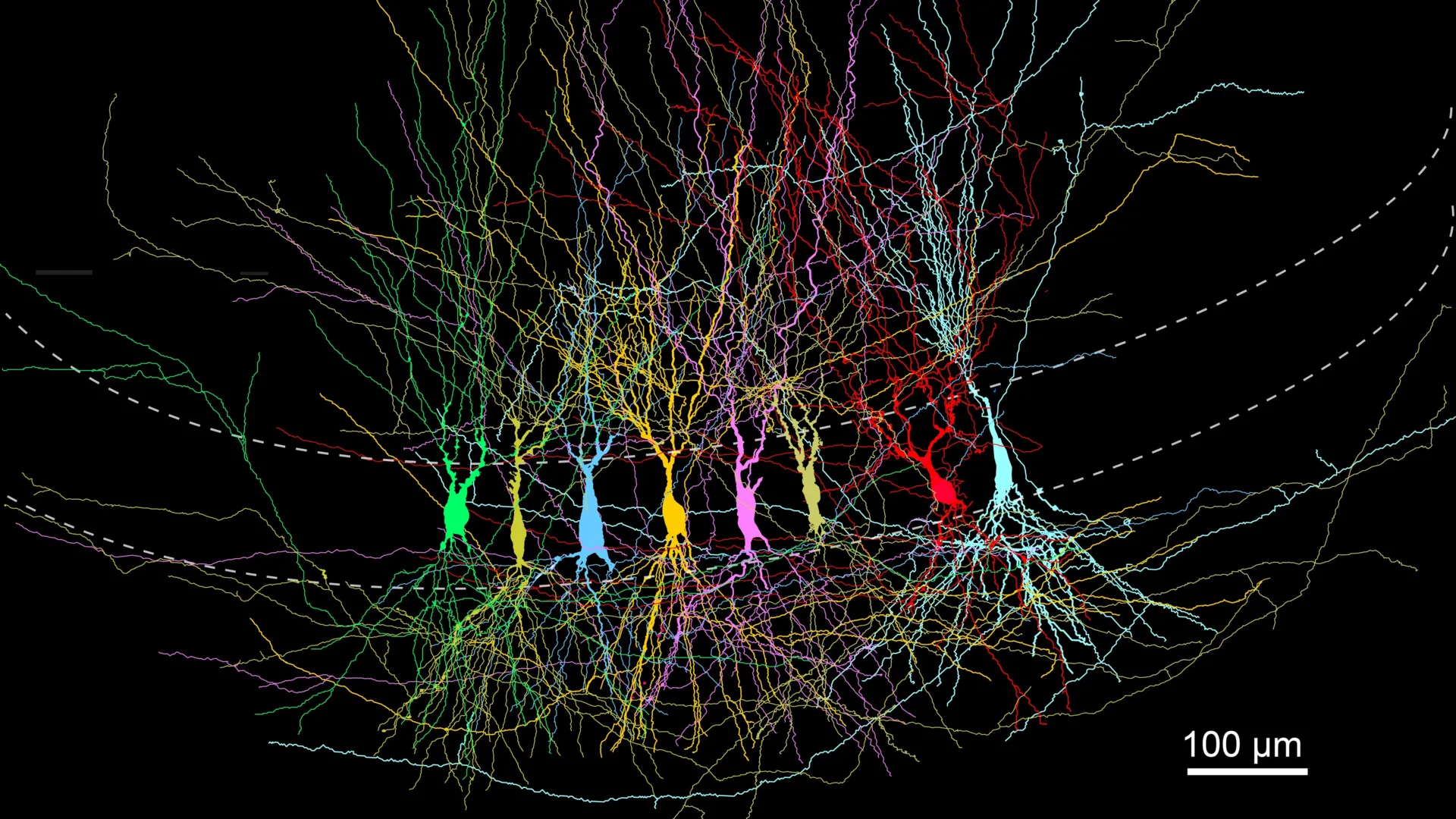
To understand the significance of this discovery, one must first look at the specific geography of the brain under investigation. The researchers focused their efforts on the CA3 region of the hippocampus. This area is often described by neuroscientists as the "auto-associative" network of the brain. It is comprised of pyramidal neurons that are essential for pattern completion—the ability to recall a whole memory from just a small fragment of information, such as remembering a childhood birthday party from the mere scent of a candle.
The CA3 network relies on a complex web of recurrent connections, where neurons link back to themselves and their neighbors. These connections are governed by synaptic plasticity, the biological process by which the strengths of connections between neurons are modified. In a mature brain, these connections are highly specific and efficient. The ISTA team sought to determine how this precision is achieved: is it built from scratch, or is it carved out of a larger, unrefined mass?
A Longitudinal Chronology of Brain Development
The research team conducted their study using mouse models, which provide a reliable proxy for mammalian brain development. To capture the full trajectory of hippocampal maturation, the researchers identified three distinct chronological stages for observation:
- Early Postnatal Period (Days 7–8): This stage represents the infant brain, where sensory inputs are just beginning to be processed and long-term memory structures are in their infancy.
- Adolescence (Days 18–25): A period of rapid learning and environmental interaction, where the brain is highly sensitive to external stimuli and undergoes significant structural changes.
- Adulthood (Days 45–50): The stage at which the brain’s circuits have largely stabilized and reached peak operational efficiency.
By comparing these three stages, the team was able to map the physical and functional evolution of the CA3 network. The results were counterintuitive. Instead of a sparse network becoming more complex, the researchers found that the youngest brains had the highest density of connections. These early connections, however, were random and lacked the organizational logic required for advanced cognitive functions.
Technical Precision: Patch-Clamping and Laser Activation
The methodology employed by Vargas-Barroso and Jonas was as rigorous as the findings were surprising. To measure the microscopic electrical signals passing between neurons, the team utilized the "patch-clamp" technique. This method involves using a tiny glass micropipette to "clamp" onto a specific part of a neuron—such as the presynaptic terminals or the dendrites—to record the flow of ions through individual channel proteins.
Complementing this, the researchers used advanced imaging and laser-based optogenetics. By using lasers to activate specific neural connections with millisecond precision, they could observe how a signal traveled through the CA3 network. This allowed the team to visualize the "noise" in the infant brain—a dense thicket of signals firing in every direction—and contrast it with the "signal" of the adult brain, where pathways were clear, direct, and purposeful.
Data Analysis: The Pruning Model of Neural Efficiency
The data collected from these experiments confirmed a shift from a dense, random network to a streamlined, optimized one. In the early postnatal stage, the probability of two neurons being connected was significantly higher than in adulthood. However, these connections were often weak and lacked the specificity needed for memory storage.
As the mice moved into adolescence and adulthood, the overall number of connections decreased. This process, known as synaptic pruning, is not a sign of decay but of maturation. "Intuitively, one might expect that a network grows and becomes denser over time," Professor Peter Jonas explained. "Here, we see the opposite. It follows what we call a pruning model: it starts out full, and then it becomes streamlined and optimized."
This optimization is critical for reducing "background noise" in the brain. If every neuron is connected to every other neuron, the system becomes overwhelmed by signals, making it impossible to isolate a specific memory. By trimming away the excess, the brain increases its signal-to-noise ratio, allowing for faster and more accurate information processing.
Why the Brain Starts Full: The Evolutionary Logic of Tabula Plena
The discovery of the "tabula plena" model raises a fundamental biological question: why would nature expend the energy to create an over-connected brain only to tear much of it down? The ISTA researchers suggest that this is an evolutionary strategy designed for speed and flexibility.
The hippocampus is responsible for integrating disparate types of information—sights, sounds, smells, and spatial data—into a single, cohesive memory. This is an incredibly complex task. If the brain started as a true "tabula rasa," neurons would have to physically grow toward one another to establish connections based on new experiences. This "growth-on-demand" model would be slow and might miss critical windows for learning.
By starting with an "exuberant" network where almost everything is already connected, the brain bypasses the need for initial structural growth. Instead, it can immediately begin the process of "selective strengthening." Connections that are used and reinforced by experience are kept and strengthened, while those that are unused are pruned away. This allows the infant brain to be "pre-wired" for connection, ensuring that as soon as an experience occurs, the hardware is already in place to record it.
Broader Implications for Neurodevelopmental Research
The findings of this study have significant implications for our understanding of neurodevelopmental disorders. Conditions such as autism spectrum disorder (ASD) and schizophrenia have long been linked to abnormalities in synaptic pruning.
In the case of ASD, some researchers hypothesize that the brain fails to prune effectively, leading to a "hyper-connected" state where sensory input is overwhelming and difficult to process. Conversely, in schizophrenia, there is evidence of excessive pruning during adolescence, leading to a loss of critical connections required for cognitive coherence.
By establishing a clear baseline for how a healthy hippocampus prunes itself, the ISTA research provides a vital "roadmap" for clinicians. Understanding the timeline of when these connections should be trimmed allows researchers to identify exactly when and where developmental processes might go off course.
Reactions from the Scientific Community
While the ISTA study focused on mice, the fundamental principles of hippocampal development are highly conserved across mammalian species, including humans. Experts in the field of neurobiology have hailed the study as a significant step forward in reconciling the "nature vs. nurture" debate.
Independent analysts suggest that this research shifts the focus from "how we learn" to "how we refine what we already have." This has implications not only for biology but also for artificial intelligence. Modern neural networks in AI are often designed to grow and add parameters; however, this research suggests that an AI modeled after the human brain might be more effective if it started with a massive, random set of connections that were then pruned down through training—a process already being explored in some "sparse" AI models.
Conclusion: The Refined Mind
The work of Peter Jonas and Victor Vargas-Barroso at ISTA provides a profound insight into the mechanics of the mind. It suggests that we do not enter the world as empty vessels waiting to be filled, but as intricate, over-connected systems waiting to be refined. Our experiences do not just add to who we are; they carve us out of a block of potential.
As the brain moves from the dense randomness of infancy to the elegant precision of adulthood, it demonstrates a remarkable biological efficiency. The "full slate" of the hippocampus ensures that we are ready to learn from the moment of birth, while the subsequent pruning ensures that our memories are clear, our navigation is accurate, and our cognitive resources are used with maximum effectiveness. This paradigm shift from a blank slate to a pruned slate fundamentally alters our understanding of human development, placing the emphasis on the brain’s incredible ability to self-optimize through the simple, yet powerful, act of letting go of the unnecessary.














Leave a Reply