For decades, the sense of smell has remained the most enigmatic of the human senses, a biological frontier that resisted the clear mapping applied to vision, hearing, and touch. While the scientific community has long understood how the retina organizes light into a visual field or how the cochlea maps sound frequencies, the olfactory system appeared to be a chaotic wilderness of neurons. However, a landmark study led by researchers at Harvard Medical School has finally brought order to this sensory mystery. By creating the first comprehensive map of smell receptors in the nose, scientists have discovered that the olfactory system is not a random distribution of cells, but a highly organized architectural marvel characterized by precise horizontal bands.
The research, published in the journal Cell, represents a paradigm shift in neurobiology. Led by Sandeep Robert Datta, a professor of neurobiology in the Blavatnik Institute at Harvard Medical School, the study utilized advanced genetic sequencing and spatial mapping to analyze millions of neurons. The findings reveal a level of structural sophistication that challenges thirty years of scientific assumptions and provides a new blueprint for understanding how the brain interprets the chemical world.
The Architecture of Scent: From Chaos to Order
In the traditional view of olfaction, the neurons responsible for detecting odors were thought to be scattered somewhat haphazardly across the olfactory epithelium—the specialized tissue inside the nose. While researchers knew that different neurons expressed different receptors, the prevailing theory suggested that these neurons were distributed in broad, ill-defined zones, with a high degree of randomness within those areas.
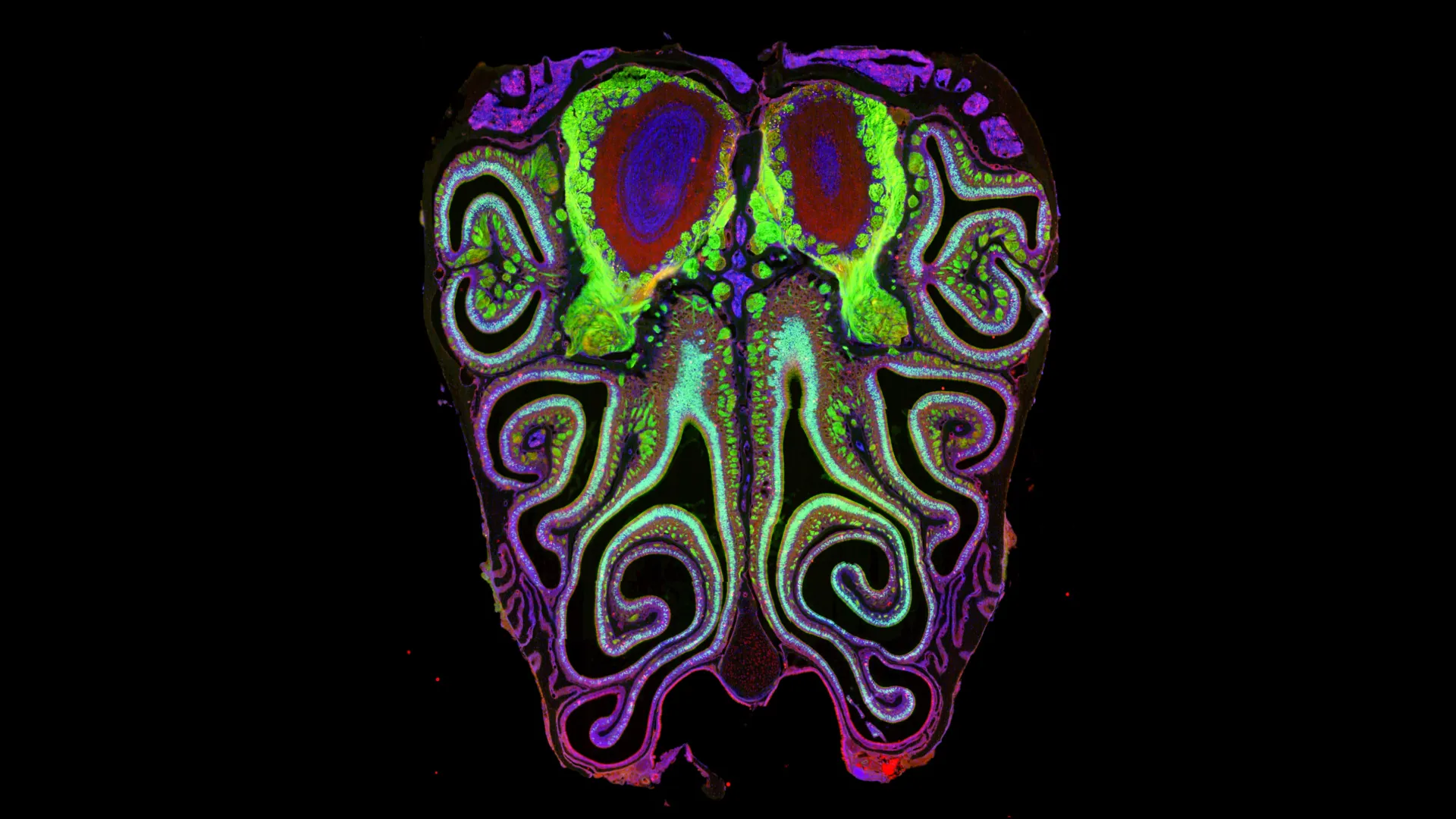
The new Harvard study systematically dismantles this notion. By analyzing the noses of more than 300 mice, Datta’s team identified that more than a thousand types of smell receptors are arranged in a series of tightly packed, overlapping horizontal stripes. These stripes run from the top of the nasal cavity to the bottom, with each receptor type assigned to a specific geographic coordinate. This organization is so consistent that it was nearly identical across every animal studied, suggesting a fundamental biological law governing the layout of the olfactory system.
"Our results bring order to a system that was previously thought to lack order," Datta explained. This conceptual shift is significant because it suggests that the position of a neuron in the nose is intimately tied to its function. The "where" of a smell receptor is just as important as the "what," creating a spatial logic that the brain can use to decode complex odors.
A Chronology of Olfactory Discovery
To understand the magnitude of this breakthrough, one must look at the timeline of olfactory research. The modern era of smell science began in 1991, when Richard Axel and Linda Buck identified the family of genes that encode odorant receptors. This discovery, which later earned them the Nobel Prize, revealed that mice have approximately 1,000 different types of receptors, while humans have about 400.
For the next thirty years, scientists attempted to map these receptors. Early technology, however, was limited. Researchers could only look at a few receptors at a time, leading to the "zone" theory—the idea that the nose was divided into four or five large regions where certain receptors might be more common, but otherwise mixed randomly.
As genomic tools evolved, the ability to look at the entire repertoire of receptors simultaneously became possible. The Harvard team’s work represents the culmination of this technological evolution, moving from the study of individual genes to the "big data" era of neuroscience. By combining single-cell sequencing with spatial transcriptomics, the researchers were able to pinpoint the exact location of 5.5 million neurons, providing a resolution that was previously unimaginable.
The Complexity Gap: Why Smell Was the Final Frontier
The delay in mapping the sense of smell, compared to vision or hearing, stems from its sheer complexity. In the visual system, the human eye relies on just three main types of photoreceptors to perceive the entire spectrum of color. In contrast, the olfactory system must detect tens of thousands of different chemical molecules.
Mice possess roughly 20 million olfactory neurons, each of which chooses to express only one of the 1,000 available receptor types. When an odor molecule enters the nose, it binds to a specific set of receptors, creating a unique "chemical fingerprint." Mapping how 1,000 different variables are arranged across 20 million points of data is a computational and biological challenge that far exceeds the mapping of the three variables of color vision.
The Harvard study’s ability to find a pattern within this massive dataset is what makes the discovery so profound. It reveals that the brain does not have to sift through a random soup of information; instead, it receives a pre-organized signal from the nose that is already categorized by its spatial origin.
The Biological GPS: The Role of Retinoic Acid
One of the most significant aspects of the study was the identification of the mechanism that creates this map. The researchers discovered that the organization of the nose is guided by a gradient of retinoic acid, a derivative of Vitamin A known to play a crucial role in embryonic development.
Retinoic acid acts as a molecular signal, or a "positional cue," that tells developing neurons where they are located along the vertical axis of the nose. Depending on the concentration of retinoic acid a neuron senses, it activates a specific smell receptor. When the researchers experimentally manipulated the levels of retinoic acid, the entire map of receptors shifted. Increasing the acid caused the stripes to move in one direction, while decreasing it caused them to move in the other.
This finding provides a developmental explanation for the map’s precision. It shows that the olfactory system uses a simple chemical gradient to solve the complex problem of organizing a thousand different cell types into a consistent pattern across an entire species.
Alignment with the Brain’s Olfactory Bulb
The implications of the nasal map extend directly into the brain. The researchers found that the horizontal stripes in the nose correspond to specific locations in the olfactory bulb—the first processing station for smell in the brain.
This alignment is critical for neural circuitry. It means that when a scent is detected in a specific stripe in the nose, the information is sent to a specific "address" in the brain. This "nose-to-brain" mapping ensures that the spatial order established in the nasal cavity is preserved as the information moves deeper into the nervous system. This discovery helps explain how the brain can so rapidly and accurately distinguish between similar odors, such as the difference between a lemon and an orange, by utilizing the spatial coordinates of the incoming signals.
Collaborative Validation and Scientific Consensus
The Harvard study did not stand alone. In a rare moment of scientific synchronicity, a separate study led by Catherine Dulac, the Xander University Professor at Harvard, was published in the same issue of Cell with nearly identical findings.
Working independently and using different methodologies, Dulac’s team also observed the organized, striped patterns of receptors in the mouse nose. The fact that two world-class laboratories reached the same conclusion simultaneously provides a powerful validation of the results. It moves the "striped map" theory from a single-study hypothesis to an established scientific fact, providing a firm foundation for all future research in the field.
Clinical Implications: A New Hope for Treating Anosmia
While the study was conducted in mice, the biological principles are expected to have significant relevance for human health. Loss of smell, or anosmia, has historically been a neglected area of medicine, but it gained global attention during the COVID-19 pandemic, which left millions of people with lingering olfactory dysfunction.
Anosmia is more than a mere inconvenience; it is a major health issue. Smell is essential for detecting hazards like gas leaks or spoiled food. It is also intrinsically linked to nutrition, as smell accounts for the majority of what we perceive as flavor. Furthermore, because the olfactory system is directly connected to the brain’s emotional and memory centers (the amygdala and hippocampus), the loss of smell is frequently associated with depression and cognitive decline.
"We cannot fix smell without understanding how it works on a basic level," Datta noted. The discovery of the organized map and the role of retinoic acid opens the door to several potential therapies:
- Stem Cell Regeneration: If scientists know exactly where certain receptors are supposed to be located, they can more accurately direct stem cell therapies to regrow damaged tissue in the correct patterns.
- Targeted Drug Delivery: Understanding the retinoic acid gradient could lead to treatments that "re-tune" the nose after viral infections or trauma.
- Brain-Computer Interfaces: For those with permanent nasal damage, a detailed map of how the nose connects to the olfactory bulb could allow for the development of electronic sensors that bypass the nose and stimulate the brain directly, restoring a sense of smell through technology.
Conclusion: Decoding the Chemical World
The creation of the first detailed map of smell receptors marks the end of an era of mystery and the beginning of a new chapter in sensory biology. By revealing the hidden stripes that organize our olfactory experience, the Harvard team has provided the missing piece of the sensory puzzle.
As researchers move forward, they are already looking to see if this same organization exists in humans. While human noses are structurally different from those of mice, the fundamental genetic and developmental pathways are often conserved across species. If the human nose follows a similar "striped" logic, it will revolutionize our understanding of how we interact with the world around us.
For now, the study stands as a testament to the power of modern genomic technology and the persistence of basic science. By bringing order to the "super-mysterious" world of olfaction, scientists have not only mapped a physical space but have also mapped the potential for new treatments that could one day restore a vital sense to millions of people worldwide. Without this map, as Datta concluded, the scientific community was "doomed to fail" in its efforts to treat smell disorders. With it, the path forward is clearer than ever.














Leave a Reply