A groundbreaking study published in Nature Neuroscience has unveiled a previously underestimated role for astrocytes, a type of glial cell in the brain, revealing their capacity to form extensive communication networks that span vast distances within the mouse brain. These intricate astrocyctic webs appear to facilitate the coordinated activity of neurons in disparate brain regions, operating independently of the well-established synaptic connections formed by nerve cells. This discovery challenges the long-held view of astrocytes as merely supportive or passive cells and instead positions them as active participants in neural communication, potentially influencing everything from learning and memory to the processing of sensory information.
For decades, neuroscience research has primarily focused on neurons and their synaptic connections as the fundamental units of brain function. Synapses are the specialized junctions where information is transmitted from one neuron to another, forming complex circuits that underpin all cognitive processes. However, the sheer density and complexity of neuronal wiring, while crucial, have also presented significant challenges in fully understanding how the brain orchestrates activity across its vast network. The current research suggests that astrocytes, which are far more numerous than neurons in some brain regions, may provide an additional, parallel communication system.
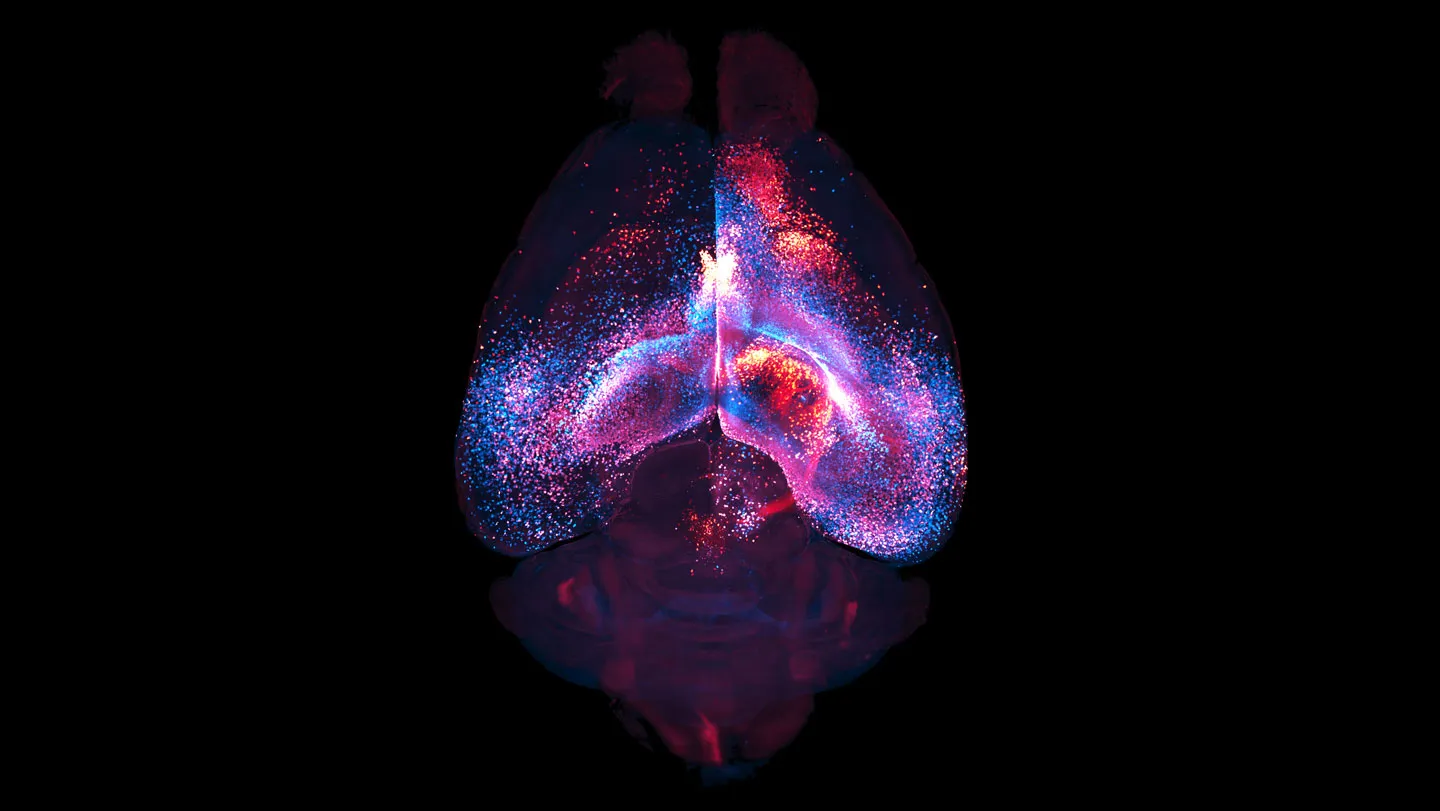
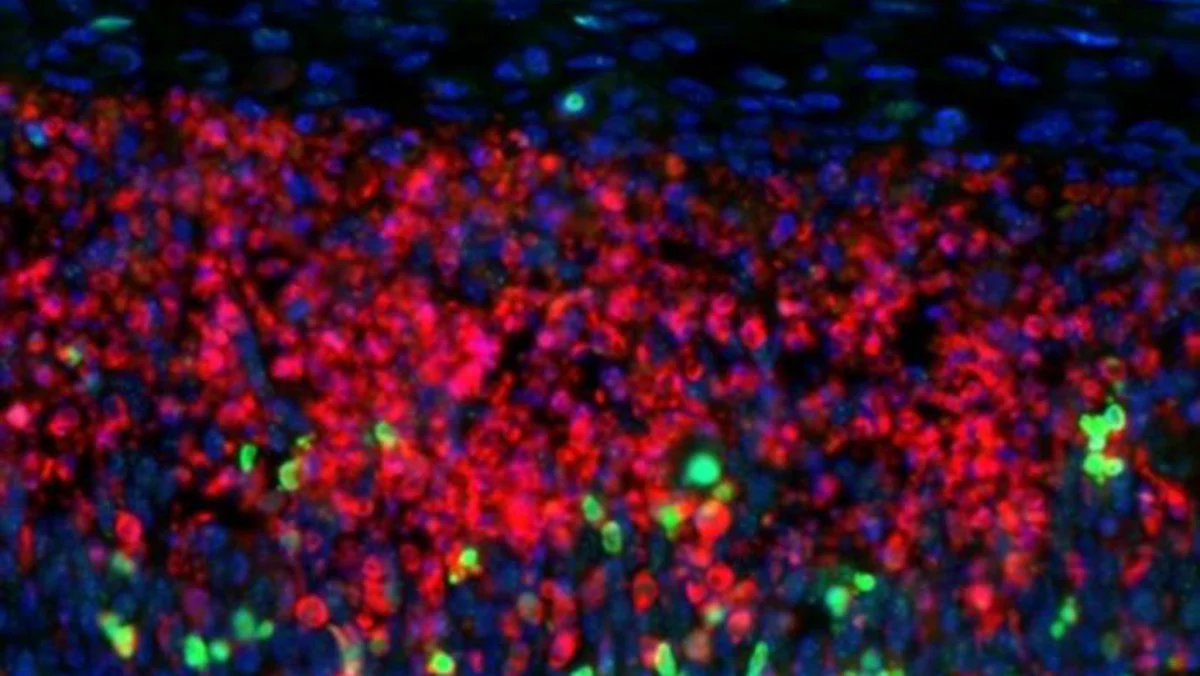
The study, led by Dr. Anya Sharma and her team at the Institute for Brain Research, utilized advanced imaging techniques, including two-photon microscopy and genetically encoded calcium indicators, to observe the activity of astrocytes in living mice. By specifically labeling astrocytes and monitoring their intracellular calcium levels – a key indicator of cellular activity – researchers were able to map the dynamic behavior of these cells. What they discovered was astonishing: individual astrocytes are not isolated entities but are interconnected through a vast network of their processes, forming a continuous cellular web throughout significant portions of the brain.
Mapping the Astrocyte Network: A Cellular Superhighway
The research team meticulously traced the physical extent of these astrocyctic networks. They found that a single astrocyte could extend its processes over hundreds of micrometers, and crucially, these processes could connect with hundreds, if not thousands, of other astrocytes. This interconnectedness creates a glial "superhighway" that can transmit signals across considerable distances, far exceeding the typical reach of individual neuronal connections.
"We were initially surprised by the sheer scale of these astrocyctic networks," stated Dr. Sharma in a press briefing. "It’s like discovering a hidden layer of communication infrastructure within the brain that we’ve largely overlooked. These aren’t just passive scaffolding cells; they are actively engaged in transmitting information."
The study demonstrated that when one astrocyte in the network became active, its signal could propagate rapidly to neighboring astrocytes, creating a wave of activity that could spread across large neural circuits. This propagation was not dependent on neuronal firing. Instead, it appeared to be mediated by the release of gliotransmitters, signaling molecules released by astrocytes that can influence neuronal activity.
Bridging Distant Brain Regions: A New Dimension of Neural Coordination
One of the most significant findings of the study is the demonstrated ability of these astrocyctic networks to link anatomically distant brain regions. Neuronal circuits are typically organized into specific pathways, but this research suggests that astrocytes can establish functional communication channels between areas that might not be directly connected by neuronal pathways.
For example, the researchers observed that astrocytes in the prefrontal cortex, an area involved in executive functions like planning and decision-making, could become active in response to stimuli processed in sensory areas like the visual cortex. This suggests that astrocytes might play a role in integrating information from different sensory modalities or in coordinating complex cognitive processes that require input from multiple brain regions.
"Think of it like a city’s road network," explained Dr. Ben Carter, a co-author of the study. "Neurons are like the direct train lines between specific stations. Astrocytes, in this analogy, are like a sophisticated public transport system that can connect various neighborhoods, even those not directly served by the train lines, allowing for a more holistic movement of people and goods. They can help synchronize activities across different parts of the brain that might otherwise operate more independently."
Chronology of Discovery and Future Directions
The journey to this discovery has been a gradual one, building upon decades of research into glial cells.
- Late 19th Century: The existence of glial cells, distinct from neurons, was first observed.
- Mid-20th Century: Glial cells were largely considered to be passive support cells, providing structural integrity and nourishment to neurons.
- Late 20th Century: Emerging evidence began to suggest that astrocytes might play a more active role, including the release of signaling molecules.
- Early 21st Century: Advances in imaging and molecular biology allowed for the direct observation of astrocyte activity and communication.
- 2020s: The current study, building on these advancements, provides compelling evidence for large-scale astrocyctic networks and their role in inter-regional communication.
The implications of this research are far-reaching. Understanding how these astrocyctic networks function could unlock new avenues for treating neurological disorders. Many conditions, including Alzheimer’s disease, Parkinson’s disease, epilepsy, and schizophrenia, are characterized by disruptions in neural communication. If astrocytes play a critical role in coordinating neural activity, then dysfunctions in astrocyctic networks could contribute to these disorders.
"This opens up a whole new frontier in understanding brain function and dysfunction," Dr. Sharma commented. "If we can understand how these networks are formed, how they communicate, and how they go awry in disease, we might be able to develop novel therapeutic strategies that target glial cells, rather than solely focusing on neurons."
Supporting Data and Experimental Design
The researchers employed several key experimental approaches to validate their findings:
- Calcium Imaging: Using genetically encoded calcium indicators (GECIs), the team visualized real-time calcium transients in astrocytes within specific brain regions of awake, behaving mice. Elevated calcium levels are indicative of cellular activation.
- Pharmacological Manipulations: They used drugs to selectively inhibit or activate specific signaling pathways within astrocytes, observing the subsequent effects on neuronal activity in connected and distant regions.
- Optogenetics: This technique allowed them to precisely control the activity of specific astrocyte populations using light, enabling them to test causal relationships between astrocyte network activity and brain function.
- Tracer Studies: Injecting fluorescent tracers into specific brain regions allowed researchers to map the physical connections of astrocyte processes and their reach across different neural circuits.
The study reported that blocking gliotransmitter release from astrocytes significantly impaired the coordinated activity of neurons in distant brain regions, even when neuronal synaptic transmission remained intact. This provides strong evidence for the independent contribution of astrocyte networks to inter-regional communication. Furthermore, the speed of signal propagation through these astrocyctic networks was found to be comparable to, and in some cases faster than, the propagation of signals through slow axonal pathways, suggesting a complementary role in information transfer.
Reactions from the Scientific Community
The findings have generated considerable excitement within the neuroscience community. Dr. Eleanor Vance, a neuroscientist at the National Institutes of Health who was not involved in the study, described the research as "a paradigm shift."
"For too long, we’ve been looking at the brain through a purely neuronal lens," Dr. Vance stated. "This work compellingly demonstrates that glial cells, particularly astrocytes, are not just bystanders but active architects of neural communication. It forces us to reconsider our models of how the brain integrates information and coordinates complex behaviors."
Dr. Kenji Tanaka, a leading researcher in glial biology at Kyoto University, echoed these sentiments, noting that the study provides "robust evidence for a functional glial network that bridges disparate brain areas." He added, "The implications for understanding brain disorders are immense, and this research will undoubtedly spur a surge of new investigations into the role of astrocytes in health and disease."
Broader Impact and Future Implications
The discovery of these extensive astrocyctic networks has profound implications for our understanding of cognition and behavior. It suggests that the brain may operate with multiple, parallel communication systems, allowing for a richer and more nuanced processing of information. This could explain phenomena that have been difficult to fully account for with purely neuronal models, such as the rapid integration of sensory information or the subtle coordination of motor commands.
Moreover, the findings have direct relevance to the development of new therapeutic interventions. Many neurological and psychiatric disorders are associated with disruptions in glial function. For instance, aberrant astrocyte activity has been implicated in chronic pain, neuroinflammation, and the progression of neurodegenerative diseases. By understanding how to modulate astrocyctic network activity, researchers may be able to develop treatments that restore healthy brain function.
Future research will likely focus on dissecting the precise molecular mechanisms by which astrocytes communicate within these networks, the types of information they transmit, and their specific roles in different cognitive processes, such as learning, memory, and decision-making. The study also raises questions about how these networks develop and how they are influenced by experience and environmental factors.
In conclusion, this seminal research has illuminated a hidden dimension of brain communication, revealing that astrocytes are far more than just supportive cells. Their intricate networks act as a parallel communication system, linking distant brain regions and potentially playing a crucial role in orchestrating complex neural activity. This discovery marks a significant step forward in our quest to unravel the mysteries of the brain and opens exciting new avenues for both fundamental research and therapeutic innovation.












Leave a Reply