A research team at King’s College London has announced the discovery of a pioneering molecular form of aluminum that could fundamentally alter the landscape of industrial chemistry. Led by Dr. Clare Bakewell, a Senior Lecturer in the Department of Chemistry, the researchers have successfully synthesized highly reactive aluminum molecules capable of severing some of the most resilient chemical bonds known to science. This breakthrough, recently detailed in the journal Nature Communications, introduces a stable yet potent alternative to the expensive and environmentally taxing rare earth metals that currently dominate the global manufacturing and energy sectors.
The core of the discovery lies in the creation of a "cyclotrialumane," a unique compound featuring three aluminum atoms arranged in a precise triangular, or trimeric, configuration. While aluminum is the most abundant metal in the Earth’s crust, its application in complex chemical catalysis has historically been limited compared to transition metals like platinum, palladium, and rhodium. The new structure developed by Dr. Bakewell’s team overcomes these traditional limitations, demonstrating a level of reactivity and stability that allows it to mimic—and in some cases, exceed—the performance of its more expensive counterparts.
The Architecture of Cyclotrialumane: A Structural Milestone
The identification of the cyclotrialumane structure represents a significant milestone in main-group chemistry. For decades, the challenge for chemists has been to create aluminum compounds that remain stable enough for practical use while maintaining the high reactivity required to facilitate chemical transformations. The King’s College London team achieved this balance by stabilizing the triangular aluminum core, ensuring it remains intact even when dissolved in various chemical solutions.
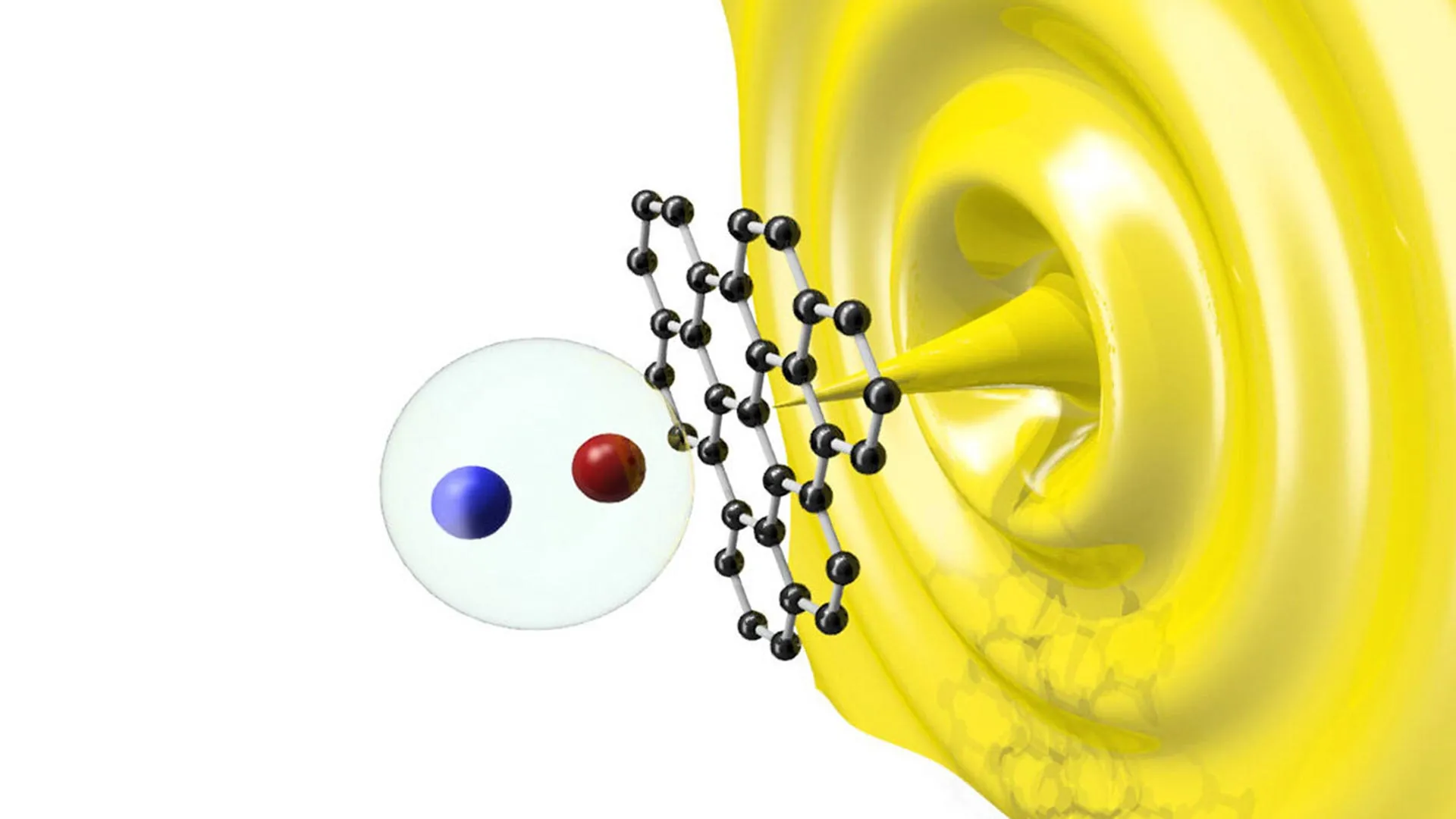
This stability is critical for industrial applications, where catalysts must survive the rigors of a reaction environment without breaking down prematurely. The trimeric structure allows the molecule to engage in "multi-center" bonding, a phenomenon where the electrons are shared across the three aluminum atoms. This configuration provides the necessary electronic environment to break strong covalent bonds, such as those found in dihydrogen (H2) and various hydrocarbons.
In laboratory trials, the cyclotrialumane demonstrated the ability to split dihydrogen molecules and facilitate the step-by-step insertion of ethene, a fundamental building block in the petrochemical industry. Furthermore, the reaction with ethene resulted in the formation of unprecedented five- and seven-membered aluminum and carbon rings. These structures are not merely academic curiosities; they represent entirely new pathways for synthesizing complex organic molecules that were previously difficult or impossible to create using conventional catalysts.
Economic and Geopolitical Drivers for Aluminum Research
The push to find alternatives to transition metals is driven by a combination of economic volatility and geopolitical necessity. Currently, the production of everything from pharmaceuticals to smartphone components and catalytic converters relies on Platinum Group Metals (PGMs) and Rare Earth Elements (REEs).
According to market data, the price of platinum often fluctuates between $900 and $1,100 per ounce, while palladium can reach even higher peaks depending on supply chain stability. In contrast, aluminum is priced by the metric ton, typically trading at approximately $2,200 to $2,500 per ton. As Dr. Bakewell noted, this makes aluminum roughly 20,000 times less expensive than the precious metals it seeks to replace.
Beyond cost, the geographical concentration of rare metal reserves presents a significant risk to global supply chains. A vast majority of the world’s transition metals are extracted from a small number of regions, including South Africa, Russia, and China. Political instability or trade disputes in these areas can lead to immediate price spikes and manufacturing delays. Aluminum, being ubiquitous and easily extracted from bauxite ore found on nearly every continent, offers a level of resource security that rare earth metals cannot match.
Chronology of Catalyst Development and the Shift to Main-Group Elements
To understand the significance of this discovery, it is essential to view it within the broader timeline of chemical evolution:
- Late 19th – Early 20th Century: The industrial revolution saw the rise of the Haber-Bosch process and other catalytic methods relying heavily on iron and nickel.
- Mid-20th Century: The discovery of organometallic catalysis led to the dominance of transition metals like palladium and rhodium, which became the industry standard for complex cross-coupling reactions (winning several Nobel Prizes).
- Early 2000s: Concerns over "peak minerals" and the environmental cost of mining sparked the "Green Chemistry" movement. Researchers began exploring "Frustrated Lewis Pairs" and other methods to make main-group elements (like boron and phosphorus) behave like transition metals.
- 2010s-2020s: Advances in ligand design allowed for the stabilization of low-valent aluminum and magnesium compounds.
- 2024: The King’s College London team successfully synthesizes the cyclotrialumane, proving that a stable, trimeric aluminum structure can perform advanced bond-breaking tasks traditionally reserved for heavy transition metals.
Comparative Data: Aluminum vs. Precious Metals
The potential impact of this research is best illustrated by comparing the properties and availability of aluminum against traditional catalytic metals:
| Property | Aluminum (Al) | Platinum (Pt) | Palladium (Pd) |
|---|---|---|---|
| Abundance in Crust | ~8.1% (Most abundant metal) | ~0.0000005% | ~0.0000015% |
| Relative Cost | $1 (Baseline) | ~$20,000+ | ~$25,000+ |
| Environmental Impact | High recycling rate; open-pit mining | High; toxic byproducts; energy-intensive | High; specialized refining required |
| Primary Use | Construction, Packaging | Catalytic converters, Jewelry | Electronics, Industrial catalysts |
By leveraging the high abundance and low cost of aluminum, the King’s College London team is addressing the "sustainability gap" in chemical manufacturing. The ability to perform high-level chemistry with an element that is already a staple of global industry could lead to a massive reduction in the carbon footprint associated with metal extraction and refining.
Environmental Implications and Green Chemistry
The environmental benefits of substituting aluminum for rare earth metals extend beyond the reduction of mining activity. Transition metal catalysts often require complex ligand systems—organic "wrappers" that help the metal do its job—which can be toxic or difficult to dispose of. Additionally, many transition metal processes require high temperatures and pressures, contributing to the energy intensity of chemical plants.
The reactive aluminum trimer identified by Dr. Bakewell’s team operates under conditions that suggest a more energy-efficient pathway. Because the molecule is inherently highly reactive due to its unique triangular strain and electronic structure, it may allow for chemical reactions to occur at lower temperatures. This aligns with the principles of Green Chemistry, which prioritize the use of non-toxic, abundant materials and the minimization of waste.
Furthermore, the "chain growth" capability observed with ethene suggests that this aluminum compound could be used in the production of biodegradable plastics or specialized polymers. If aluminum-based catalysts can replace the heavy-metal catalysts currently used in polymer synthesis, the resulting products would be easier to recycle and less likely to leach harmful metallic residues into the environment.
Expert Analysis and Industry Response
While the research is still in the "exploratory phase," as Dr. Bakewell described it, the scientific community has reacted with cautious optimism. Independent analysts suggest that the primary hurdle will be the sensitivity of these aluminum compounds to air and moisture. Unlike platinum, which is chemically inert and easy to handle, reactive aluminum molecules typically require an inert atmosphere (such as nitrogen or argon) to remain stable.
"The discovery of the cyclotrialumane is a masterclass in molecular engineering," says an independent inorganic chemist not involved in the study. "The challenge now is to see if these five- and seven-membered rings can be produced at scale and if the catalyst can be ‘recycled’ within a reaction cycle. If they can achieve a high turnover frequency, this could be a game-changer for the plastics and pharmaceutical industries."
Dr. Bakewell and her team are currently focusing on expanding the library of reactions these aluminum trimers can facilitate. The team is particularly interested in whether the unique reactivity of the 5- and 7-membered aluminum-carbon rings can be harnessed to create new types of materials with distinctive mechanical or electronic properties.
Looking Forward: From the Lab to the Factory Floor
The transition from a laboratory discovery to an industrial application is a journey that typically takes a decade or more. However, the economic incentives for this specific breakthrough are unusually high. Companies in the chemical sector are under increasing pressure from both regulators and consumers to "decarbonize" their supply chains and reduce reliance on "conflict minerals" or materials sourced from ecologically sensitive areas.
The next steps for the King’s College London researchers involve optimizing the synthesis of the cyclotrialumane to ensure it can be produced reliably and cost-effectively. They will also investigate the "catalytic cycle"—the ability of the molecule to perform a reaction and then return to its original state to start again. This "recyclability" at the molecular level is what defines a true catalyst and is the key to its industrial viability.
As the global community moves toward a circular economy, the ability to unlock the hidden potential of earth-abundant materials like aluminum is paramount. The work of Dr. Bakewell’s team suggests that the future of high-tech chemistry may not lie in the rarest elements on the periodic table, but in the most common ones, reimagined through innovative molecular design.
In her concluding remarks regarding the study, Dr. Bakewell emphasized the dual nature of the discovery: "What’s special about this work is that we’re pushing the boundaries of chemical knowledge… this chemistry could support a transition to cleaner, greener and cheaper chemical production, whilst making new discoveries along the way." By bridging the gap between fundamental science and industrial necessity, the King’s College London team has provided a glimpse into a more sustainable future for global chemistry.













Leave a Reply