Researchers at Washington University School of Medicine in St. Louis have unveiled a breakthrough in neurodegenerative research that could fundamentally alter the trajectory of Alzheimer’s disease treatment. By repurposing the principles of cancer immunotherapy, the scientific team has successfully engineered astrocytes—the most abundant support cells in the central nervous system—to identify, target, and eliminate the toxic amyloid beta plaques that characterize the disease. This experimental approach, detailed in a study published on March 5 in the journal Science, offers a potential transition from the current regimen of lifelong, frequent infusions toward a more sustainable, long-term cellular therapy.
The development comes at a pivotal moment in the history of Alzheimer’s care. For decades, the medical community struggled to produce treatments that did more than manage symptoms. The recent emergence of monoclonal antibodies, such as lecanemab and donanemab, marked the first era of disease-modifying therapies. While these drugs have proven effective at slowing cognitive decline—typically granting patients an additional 10 months of independent living—they are hampered by significant logistical hurdles. Patients must undergo intravenous infusions once or twice a month, a process that is both time-consuming and expensive. The new "CAR-astrocyte" therapy, however, suggests that a single injection could potentially achieve superior results with significantly less patient burden.
The Evolution of the Amyloid Hypothesis and Current Clinical Standards
To understand the significance of the Washington University study, one must look at the "amyloid hypothesis," which has dominated Alzheimer’s research for over thirty years. This theory posits that the accumulation of amyloid beta protein in the brain is the primary driver of the disease. These proteins clump together to form "plaques," which disrupt cell-to-cell communication and trigger an inflammatory response that eventually kills neurons.
Current FDA-approved monoclonal antibodies work by circulating in the bloodstream, crossing the blood-brain barrier in small amounts, and binding to these plaques to signal the immune system to remove them. While revolutionary, the efficacy of these treatments is limited by the "half-life" of the antibodies; once the drug clears the system, the therapeutic effect wanes, necessitating the next dose. Furthermore, these treatments rely on the brain’s existing immune cells, known as microglia, to do the heavy lifting. In a brain ravaged by Alzheimer’s, these microglia are often "exhausted" or dysfunctional, limiting the overall impact of the medication.
Repurposing CAR-T Technology for the Central Nervous System
The research team, led by senior author Marco Colonna, MD, the Robert Rock Belliveau, MD, Professor of Pathology at WashU Medicine, looked to oncology for a solution. Chimeric Antigen Receptor (CAR) T-cell therapy has transformed the treatment of certain blood cancers. In CAR-T therapy, a patient’s own T-cells are genetically modified to express a receptor that acts as a "homing device," allowing them to recognize and kill specific cancer cells.
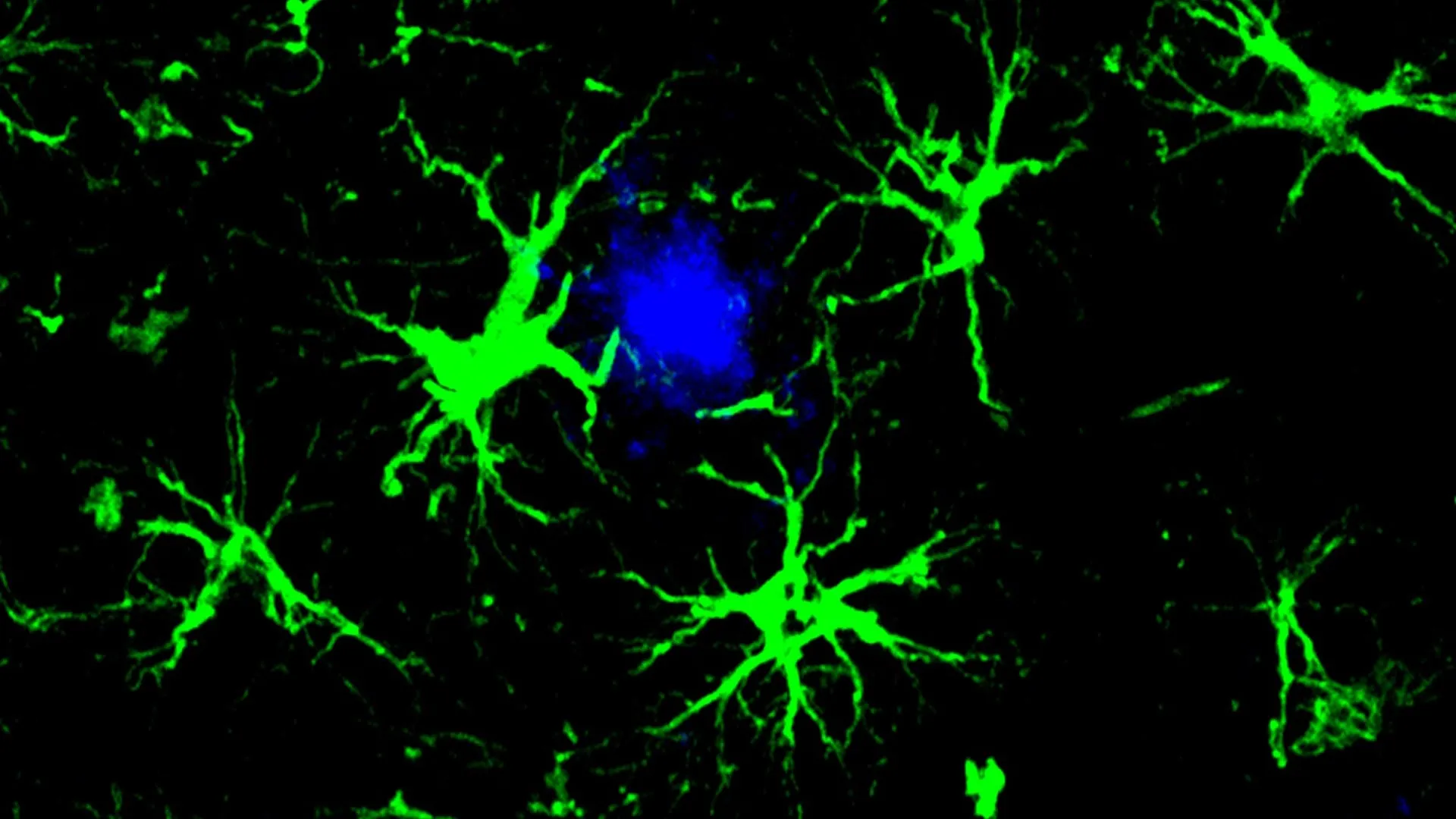
In the context of the brain, however, T-cells can be dangerous, as excessive T-cell activity in the central nervous system often leads to severe neuroinflammation. Instead of T-cells, the researchers focused on astrocytes. Historically viewed as mere "glue" holding neurons together, astrocytes are now understood to be vital players in brain metabolism, the maintenance of the blood-brain barrier, and the regulation of synapses.
"This study marks the first successful attempt at engineering astrocytes to specifically target and remove amyloid beta plaques in the brains of mice with Alzheimer’s disease," Dr. Colonna stated. By equipping these native brain cells with a CAR homing device, the scientists effectively turned the brain’s own infrastructure into a specialized cleaning crew.
Methodology: From Genetic Engineering to In Vivo Testing
The study was spearheaded by first author Yun Chen, PhD, during his time as a graduate student in the laboratories of Dr. Colonna and David M. Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology. The team utilized a viral vector—a harmless virus—to deliver the CAR gene directly into the brains of mice.
The engineered CAR was specifically designed to recognize amyloid beta. Once the astrocytes expressed this receptor on their surface, they gained the ability to physically latch onto amyloid proteins. Unlike normal astrocytes, which might ignore plaques or even contribute to scarring around them, these "CAR-astrocytes" were programmed to "swallow" and degrade the toxic aggregates through a process called phagocytosis.
The researchers conducted a longitudinal study using two distinct groups of mice genetically predisposed to develop Alzheimer’s-like symptoms. These mice typically begin showing significant plaque buildup by six months of age, mirroring the early-to-mid stages of human Alzheimer’s.
Chronology of Findings: Prevention and Reduction
The results of the trial were categorized by the timing of the intervention, providing a clear timeline of the therapy’s potential efficacy at different stages of disease progression.
1. The Preventative Phase
In the first group, the viral vector was administered to young mice before any detectable plaques had formed. The researchers then monitored the mice for several months. By the time the mice reached the six-month mark—the age at which their brains would normally be saturated with amyloid—the treated mice showed a near-total absence of plaque. The CAR-astrocytes had successfully acted as a surveillance system, clearing amyloid proteins as soon as they appeared and preventing the formation of the toxic clusters entirely.
2. The Therapeutic Phase
The second group of mice received the treatment only after they had already developed significant plaque burdens, representing a patient who is already symptomatic. Three months after the single injection, the researchers evaluated the brain tissue. They found that the CAR-astrocytes had reduced the existing amyloid levels by approximately 50 percent compared to a control group. This demonstrated that the therapy was not only a preventative measure but could actively reverse existing pathology.
Supporting Data and Comparative Analysis
The data suggests that the CAR-astrocyte approach could solve several problems inherent in current immunotherapy. In current monoclonal antibody treatments, high doses are required because only a fraction of the drug crosses from the blood into the brain. By using a viral vector to modify cells already residing within the brain, the Washington University team bypassed the blood-brain barrier entirely.
Furthermore, the study observed that the CAR-astrocytes did not appear to trigger the same level of "cytokine storm" or extreme inflammation sometimes seen in CAR-T cancer therapies. Because astrocytes are a natural part of the brain’s ecosystem, they appeared to integrate the new "cleaning" function without disrupting the delicate homeostatic balance of the surrounding neurons.
Expert Reactions and Official Implications
The scientific community has reacted with cautious optimism. David M. Holtzman, MD, a co-author of the study, highlighted the clinical potential of the single-injection model. "Consistent with the antibody drug treatments, this new CAR-astrocyte immunotherapy is more effective when given in the earlier stages of the disease," Holtzman noted. "But where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins."
The Office of Technology Management at Washington University has already assisted the researchers in filing a patent for the CAR-astrocyte engineering method. This move signals a transition from basic laboratory science toward the early stages of pharmaceutical development. Industry analysts suggest that if this technology translates to humans, it could drastically reduce the long-term costs of Alzheimer’s care by eliminating the need for monthly clinical visits and the high manufacturing costs associated with monoclonal antibodies.
Broader Impact: Beyond Alzheimer’s Disease
The implications of this research extend far beyond amyloid plaques. The "homing device" on the CAR can be swapped to target different proteins. This opens the door for treating other neurodegenerative "proteinopathies," such as:
- Parkinson’s Disease: Targeting alpha-synuclein aggregates.
- Frontotemporal Dementia and ALS: Targeting tau or TDP-43 proteins.
- Huntington’s Disease: Targeting huntingtin protein.
Perhaps most exciting is the potential application in neuro-oncology. The research team believes that by modifying astrocytes to recognize markers found on glioblastomas or other aggressive brain tumors, they could create a permanent, internal defense system against cancer. Unlike traditional chemotherapy, which often fails to reach the center of a tumor, engineered astrocytes are already distributed throughout the brain and could provide a localized, sustained attack on malignant cells.
Challenges and Future Directions
Despite the promising results in mice, the road to human clinical trials remains long. One primary concern for researchers is the permanence of the change. Because the viral vector modifies the genetic expression of the astrocytes, the "cleaning" function is essentially permanent. Scientists must ensure that these cells do not eventually begin attacking healthy proteins or cells, a phenomenon known as "off-target effects."
"Although more work needs to be done to optimize the approach and address potential side effects, these results open up an exciting new opportunity," Dr. Colonna emphasized. Future studies will focus on "tuning" the CAR receptors to ensure they only activate in the presence of pathological protein levels, effectively creating a "smart" therapy that turns itself on and off as needed.
Additionally, researchers must determine the safest delivery method for the viral vector in humans. While mice received direct injections, human application might require sophisticated neurosurgical techniques or the development of vectors that can be delivered via the spinal fluid.
Conclusion
The Washington University study represents a paradigm shift in how we approach the "clogged" brain of an Alzheimer’s patient. By moving away from external, temporary fixes and toward the internal, permanent reprogramming of the brain’s own support cells, the medical community may be on the verge of a more definitive solution to the Alzheimer’s crisis. As the global population ages and the prevalence of dementia is expected to triple by 2050, the development of a single-injection therapy could be the key to managing a public health challenge of unprecedented proportions. For now, the "cleaning cells" of St. Louis offer a compelling glimpse into a future where Alzheimer’s is not just managed, but actively dismantled from within.















Leave a Reply