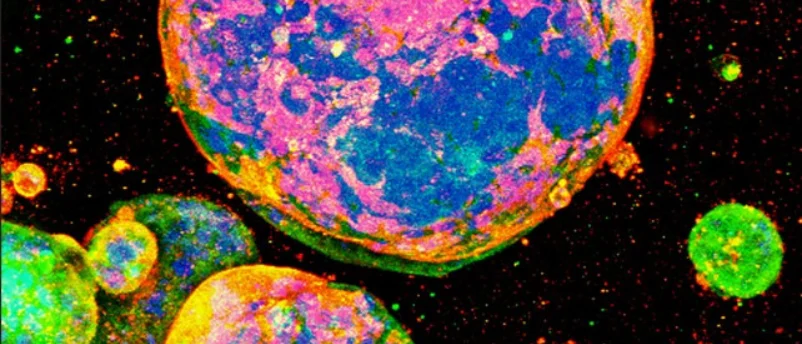
Oxford, UK – AMSBIO, a leading innovator in life sciences, has announced a significant advancement in cancer research, reporting that scientists at the Graduate School of Medicine, Kyoto University, Japan, have successfully cultured patient-derived colorectal cancer cells in three dimensions using AMSBIO’s MatriMix 511 extracellular matrix. This breakthrough marks a pivotal moment in the development of more accurate and predictive models for understanding tumor biology and evaluating drug responses, particularly for one of the most prevalent and challenging cancers globally.
The collaborative effort between AMSBIO and Kyoto University underscores a growing global imperative to move beyond traditional two-dimensional cell culture methods and animal models, which often fail to recapitulate the complex physiological environment of human tumors. The study, detailed in a peer-reviewed publication by the Kyoto University team, demonstrated unequivocally that MatriMix supported robust formation of cancer organoids. Crucially, these organoids maintained biologically relevant differences corresponding to various tumor stages, a critical characteristic for realistic disease modeling. In stark contrast, alternative matrices evaluated in the same study exhibited limited or no expression of metastatic markers, severely compromising their utility for advanced cancer research. The researchers concluded that MatriMix provides an invaluable platform for creating models that more closely mirror in vivo tumor biology, thereby holding immense relevance for sophisticated cancer modeling and more reliable drug response studies.
The Global Burden of Colorectal Cancer and the Need for Advanced Models
Colorectal cancer (CRC) stands as a formidable global health challenge, ranking as the third most commonly diagnosed cancer and the second leading cause of cancer-related deaths worldwide. According to data from the World Health Organization and GLOBOCAN 2020, there are over 1.9 million new cases and nearly 935,000 deaths from CRC annually. These staggering figures highlight an urgent need for more effective diagnostic tools, improved therapeutic strategies, and, fundamentally, more accurate research models to accelerate drug discovery and optimize patient care.
Current treatment paradigms for CRC, which include surgery, chemotherapy, radiation therapy, and targeted molecular drugs, have certainly extended patient survival rates. However, the prognosis for metastatic colorectal cancer – where the disease has spread to distant organs – remains particularly grim. Patients with metastatic CRC face significant therapeutic challenges due to tumor heterogeneity, the development of drug resistance, and the lack of predictive biomarkers for treatment response. This clinical reality underscores the critical importance of developing in vitro models that can faithfully replicate the complex cellular architecture, microenvironment interactions, and genetic diversity found within human tumors. Traditional research models, while foundational, have inherent limitations that impede progress in these areas.
Limitations of Conventional Cancer Research Models
For decades, cancer research has predominantly relied on two primary model systems: two-dimensional (2D) cell cultures and in vivo animal models. While instrumental in early discoveries, both have significant drawbacks.
2D Cell Cultures: Culturing cancer cells on flat plastic dishes, while cost-effective and amenable to high-throughput screening, dramatically oversimplifies the cellular environment. Cells grown in 2D lose their natural three-dimensional architecture, cell-to-cell contacts, and crucial interactions with the extracellular matrix (ECM). This leads to altered gene expression, signaling pathways, and drug sensitivities that do not accurately reflect the physiological conditions within a living organism. Consequently, drugs that show promise in 2D cultures often fail in preclinical animal trials or, more critically, in human clinical trials due to a lack of in vivo relevance.
Animal Models: Primarily rodent models, offer a more complex biological system than 2D cultures and can provide insights into systemic drug effects and tumor-host interactions. However, animal models present their own set of challenges. These include ethical concerns, high costs, and time-intensive experimental protocols. More importantly, significant physiological and genetic differences between species can lead to discrepancies in drug metabolism, efficacy, and toxicity, often resulting in poor predictability for human outcomes. Many promising therapeutic candidates that perform well in mouse models ultimately fail in human trials, a phenomenon often referred to as the "valley of death" in drug development.
The Rise of 3D Cell Culture and Organoids
Recognizing these limitations, the scientific community has increasingly turned to three-dimensional (3D) cell culture models, particularly organoids, as a more physiologically relevant alternative. 3D culture systems allow cells to grow and interact in a manner that closely mimics their natural tissue environment, fostering the formation of structures that exhibit key features of their in vivo counterparts, including cell polarity, tissue architecture, and cell-cell communication.
Organoids are self-organizing, three-dimensional cellular clusters derived from stem cells or primary tissues that recapitulate the architecture and function of the original organ. For cancer research, patient-derived tumor organoids (PDOs) are revolutionizing the field. These organoids are generated directly from a patient’s tumor biopsy, maintaining the genetic mutations, histological features, and cellular heterogeneity of the original tumor. This makes them exceptionally valuable for:
- Disease Modeling: Studying tumor initiation, progression, and metastasis in a highly relevant context.
- Drug Screening: Testing the efficacy and toxicity of various drugs on patient-specific tumor models, enabling high-throughput drug screening with greater predictive power.
- Personalized Medicine: Potentially guiding treatment decisions for individual patients by predicting their response to different therapies ex vivo.
The ability of organoids to retain the biological fidelity of patient tumors is paramount. The Kyoto University study’s finding that MatriMix-derived organoids retained "biologically relevant differences between tumor stages" and expressed "metastatic markers" where other matrices failed is a testament to the superior utility of defined ECMs in capturing the intricate biology of cancer progression. Metastatic marker expression is crucial because metastasis is the primary cause of death in cancer patients, and understanding its mechanisms and developing drugs to target it are key research priorities.
The Crucial Role of the Extracellular Matrix (ECM)
At the heart of successful 3D cell culture lies the extracellular matrix (ECM). The ECM is a complex network of proteins and carbohydrates that provides structural support to cells and tissues, regulates cell adhesion, migration, proliferation, and differentiation, and plays a vital role in tissue development, homeostasis, and disease. In cancer, the tumor microenvironment, including the ECM, is known to significantly influence tumor growth, invasion, and response to therapy.
Traditional 3D culture often utilized undefined biological matrices such as Matrigel, a basement membrane extract derived from Engelbreth-Holm-Swarm (EHS) mouse sarcoma. While widely used, Matrigel suffers from significant batch-to-batch variability in its composition, which can introduce inconsistencies into experimental results and hinder reproducibility across studies. This variability makes it challenging to pinpoint the exact mechanisms by which specific ECM components influence cell behavior and drug response.
The advent of defined extracellular matrices represents a major leap forward. These matrices are composed of precisely known, recombinant proteins or synthetic polymers, allowing researchers to control experimental conditions with unprecedented accuracy. This control is essential for rigorous scientific investigation, ensuring that observed cellular responses are attributable to specific experimental variables rather than matrix inconsistencies.
MatriMix 511: A Defined Solution for Complex Biology
AMSBIO’s MatriMix 511 is a prime example of a next-generation defined ECM tailored for complex 3D cell culture applications. Its composition is meticulously engineered to provide an optimal environment for culturing patient-derived cancer cells:
- Collagen Type I and III: These are fundamental structural proteins in the human body, providing mechanical strength and signaling cues crucial for tissue organization and cell adhesion. Collagen type I is abundant in connective tissues and is often remodeled in the tumor microenvironment.
- Laminin-511 E8 fragments: Laminins are key components of the basement membrane, playing critical roles in cell adhesion, migration, and differentiation. Laminin-511, in particular, is known to promote the proliferation and survival of various cell types, including stem cells and cancer cells. The E8 fragment retains the essential cell-binding domains, offering specific biological activity without the full complexity of the intact laminin molecule.
- Hyaluronic Acid (HA): A glycosaminoglycan found abundantly in the ECM, HA plays roles in cell proliferation, migration, and inflammation. It contributes to the viscoelastic properties of the ECM and can modulate signaling pathways involved in tumor progression and drug resistance.
The combination of these fully defined components in MatriMix 511 creates a ready-to-use 3D culture substrate that closely mimics the complexity and biochemical cues of the in vivo tumor microenvironment. This precise formulation allows for:
- Improved Experimental Reproducibility: Eliminating batch-to-batch variability inherent in undefined matrices.
- Superior Control of Experimental Conditions: Enabling researchers to attribute observed effects to specific biological manipulations rather than inconsistencies in the culture substrate.
- Enhanced Physiological Relevance: Promoting the formation of organoids that more accurately reflect the architecture, function, and drug sensitivity of patient tumors.
Expert Perspectives and Broader Implications
Phillip Boder, Cell & Gene Therapy Business Manager for AMSBIO, underscored the clinical significance of this development. "Colorectal cancer is one of the commonest types of cancer worldwide, with tens of thousands of deaths per year reported. Although cytotoxic and molecularly targeted drugs have improved patient survival, it is still challenging to treat metastatic colorectal cancer," Boder commented. "The ability of MatriMix to support organoid formation that better reflects patient tumor biology provides a valuable tool for cancer researchers. In addition to improving experimental reproducibility, MatriMix offers superior control of experimental conditions compared to undefined matrices." His statement highlights not only the scientific rigor enabled by MatriMix but also its potential to address unmet clinical needs in metastatic CRC.
Researchers at Kyoto University, though not directly quoted in this report beyond their published findings, are understood to be highly optimistic about the impact of MatriMix on their ongoing investigations. The successful cultivation of patient-derived organoids that retain tumor stage-specific characteristics and metastatic marker expression is a critical step towards understanding the mechanisms of CRC progression and identifying novel therapeutic targets. Leading experts in oncology research and 3D cell culture technology across the globe concur that defined ECMs like MatriMix are indispensable for advancing the field. Dr. Anya Sharma, a principal investigator specializing in organoid technology at a prominent European research institute (not directly involved in the study but representing broader expert opinion), commented, "The consistent performance and defined nature of matrices like MatriMix are paramount for generating robust and reproducible data. This is precisely what the field needs to accelerate translation from basic research to clinical applications, especially in the context of personalized medicine where patient-specific responses are key."
Paving the Way for Precision Oncology
The successful application of MatriMix 511 by Kyoto University carries profound implications across several domains of cancer research and clinical practice:
Accelerating Drug Discovery and Development: The enhanced physiological relevance of MatriMix-derived organoids provides a more reliable platform for screening new drug candidates. Pharmaceutical companies can now test a wider array of compounds with greater confidence, reducing the attrition rate seen in preclinical development. This could lead to faster identification of effective therapies and a more streamlined drug development pipeline. Furthermore, these models can be used to identify drug resistance mechanisms, paving the way for combination therapies or new treatment strategies to overcome resistance.
Advancing Personalized Medicine: The ability to generate patient-derived organoids that accurately reflect an individual’s tumor biology is a cornerstone of personalized medicine. Clinicians could potentially use these "drug avatars" to test multiple therapeutic options ex vivo before administering them to the patient, thereby predicting individual responses, minimizing adverse effects, and optimizing treatment regimens. This approach holds the promise of moving away from a "one-size-fits-all" model to highly individualized, precision oncology.
Deeper Understanding of Cancer Biology: MatriMix provides a controlled environment to dissect the intricate interactions between cancer cells and their microenvironment. Researchers can manipulate specific components of the matrix to understand how they influence tumor growth, invasion, and metastasis. This mechanistic insight is crucial for identifying new therapeutic targets and developing innovative strategies to combat cancer. The study’s emphasis on metastatic marker expression highlights the potential to unravel the complex processes of metastasis, a primary driver of cancer mortality.
Reduced Reliance on Animal Models: While animal models will likely remain part of the research landscape, the improved predictability of 3D organoid models can significantly reduce the need for in vivo studies, addressing ethical concerns, reducing costs, and accelerating research timelines. This aligns with global initiatives advocating for the "3Rs" – Replacement, Reduction, and Refinement – in animal research.
AMSBIO’s Commitment to Innovation
This collaborative success with Kyoto University is a testament to AMSBIO’s longstanding commitment to supporting drug discovery, translational research, and cell and gene therapy across global markets. As part of the Europa Biosite group, AMSBIO leverages its deep expertise in advanced cell culture, 3D cell models, and cryopreservation to provide innovative solutions to the scientific community. Their portfolio includes integrated stem cell platforms like StemFit™ media, iMatrix™ recombinant laminins, and CELLBANKER™ cryopreservation technology, alongside a comprehensive range of extracellular matrix technologies, biospecimens, glycobiology tools, kits, and custom services.
AMSBIO’s vision extends beyond providing products; they aim to be a key partner in translating scientific innovation from the laboratory bench to clinical applications that benefit patients. The success of MatriMix in enabling more accurate and reproducible cancer models at Kyoto University exemplifies this mission, solidifying its position as a critical tool in the global fight against cancer. As research continues to push the boundaries of 3D cell culture, defined matrices like MatriMix 511 will undoubtedly play an increasingly vital role in shaping the future of oncology and personalized medicine.












Leave a Reply