Every second, countless electrical charges move through the human body. These microscopic signals are essential for life, driving communication between cells, enabling energy production, and supporting metabolism. None of this would work without the careful and controlled movement of charges across cell membranes and within cells. In many ways, charge transport acts as a fundamental control system for biology. This intricate dance of charged particles, particularly protons, is now being illuminated by groundbreaking research that delves into the molecular mechanisms underpinning this vital biological and technological process. A collaborative effort involving researchers from the Fritz Haber Institute, Leipzig University, and institutions in the United States has successfully identified a key molecular structure and its role in the exceptionally efficient proton transport characteristic of phosphoric acid compounds, a discovery with profound implications for both our understanding of life and the development of next-generation technologies.
The Ubiquitous Role of Phosphate in Life and Technology
Phosphoric acid (H₃PO₄) and its related compounds are foundational to virtually all living systems. Their presence is pervasive, forming critical components of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA), the very blueprints of life. They are integral to the structure of cell membranes, acting as crucial barriers and signaling platforms. Furthermore, these phosphate-based molecules are central to adenosine triphosphate (ATP), the universal energy currency of cells, facilitating the storage and transfer of vital energy. The common thread running through these essential biological functions is the critical role of phosphate in managing the movement of positive charges, specifically protons.
Beyond the realm of biology, phosphoric acid and its derivatives are indispensable in numerous technological applications. Their remarkable ability to conduct protons makes them key components in advanced energy storage and conversion devices, such as batteries and fuel cells. In these technologies, efficient proton transport is paramount for optimizing performance, increasing energy density, and enhancing overall efficiency. The parallels between biological necessity and technological demand underscore the fundamental importance of understanding how these molecules facilitate charge movement.
Unlocking the Mechanism: Proton Shuttling and Hydrogen Bonding
The unique efficiency of proton transport in phosphate-containing materials is attributed to a process known as "proton-shuttling." Unlike the free diffusion of charges, protons in these systems do not travel independently. Instead, they engage in a coordinated hopping mechanism, moving from one molecule to another. This intricate relay race is facilitated by hydrogen bonds, which act as pre-established pathways, guiding the proton’s journey. This "shuttling" allows for remarkably rapid charge transfer, a feat crucial for the high-speed signaling and energy metabolism observed in biological systems, and desirable for the high-power output sought in technological applications.
For decades, scientists have acknowledged the existence and importance of proton-shuttling. However, the precise molecular choreography – the intricate details of how protons navigate these molecular pathways – has remained an elusive puzzle. This knowledge gap has limited the ability to fully optimize these processes for technological advancement and has left a fundamental aspect of biological function somewhat opaque.
A Deep Dive into Molecular Dynamics: The Fritz Haber Institute-Led Investigation
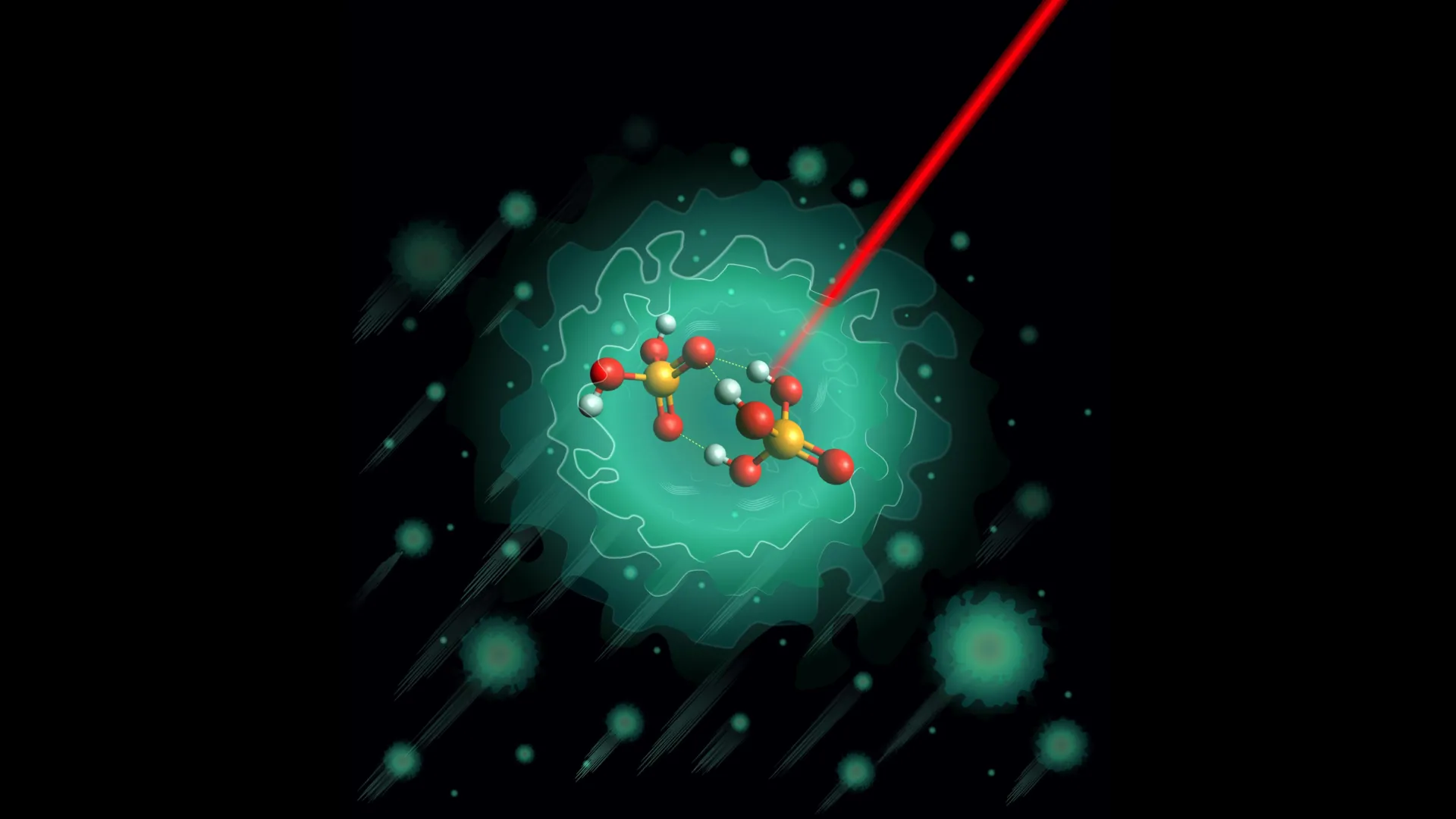
Addressing this longstanding question, a multidisciplinary team, spearheaded by researchers from the Department of Molecular Physics at the prestigious Fritz Haber Institute of the Max Planck Society, in collaboration with esteemed colleagues from Leipzig University and institutions across the United States, embarked on a mission to unravel the earliest steps of proton transfer within these critical molecular structures. Their focus narrowed to a specific, negatively charged molecule believed to initiate the proton-shuttling cascade: the deprotonated dimer of phosphoric acid, H₃PO₄·H₂PO₄⁻. This anionic dimer, carrying a net negative charge, was theorized to play a pivotal role in setting the stage for subsequent proton movements.
The research initiative, which unfolded over a period of several years, involved rigorous experimental investigation and sophisticated theoretical modeling. The initial hypotheses regarding the role of the deprotonated dimer were formulated based on existing theoretical frameworks and preliminary experimental observations from related phosphate clusters. The challenge lay in directly observing and characterizing this fleeting, highly reactive intermediate under conditions that would permit detailed analysis without disruption.
Pushing the Boundaries of Measurement: Cryogenic Conditions and Spectroscopic Precision
To overcome the inherent challenges in studying such a delicate molecular species, the research team employed an innovative approach. They meticulously synthesized the deprotonated dimer, H₃PO₄·H₂PO₄⁻, in a controlled laboratory environment. The critical step involved trapping these molecules within a cryogenic helium nanodroplet. This technique allows for the isolation of individual molecules within an extremely cold, inert medium. The temperature was reduced to an astonishing 0.37 Kelvin, just a fraction of a degree above absolute zero (-273.15 degrees Celsius or -459.67 degrees Fahrenheit). At these near-absolute zero temperatures, thermal vibrations and other random molecular disturbances are virtually eliminated, creating an environment of unparalleled stability and allowing for ultra-precise structural analysis.
Under these extreme conditions, the trapped molecules were subjected to infrared spectroscopy. This powerful analytical technique probes the vibrational modes of molecules, which are directly related to their structure and bonding. By analyzing the specific frequencies of infrared light absorbed by the molecule, researchers could infer detailed information about its geometry, bond lengths, and the types of chemical bonds present.
Crucially, the experimental findings from infrared spectroscopy were not analyzed in isolation. They were integrated with state-of-the-art quantum chemical calculations. These computational methods, grounded in the principles of quantum mechanics, allow scientists to predict molecular structures, energies, and reaction pathways. By combining the empirical data from spectroscopy with the predictive power of quantum chemistry, the research team aimed to build a comprehensive and accurate picture of the deprotonated dimer’s structure and behavior, a synergy that often reveals insights unattainable by either method alone. This dual approach, a hallmark of modern chemical physics, provided a robust foundation for interpreting the complex spectroscopic signatures.
A Surprising Unification: The Emergence of a Single, Stable Structure
The convergence of experimental and theoretical data led to a surprising and significant revelation. Prior theoretical models had suggested that the deprotonated dimer, H₃PO₄·H₂PO₄⁻, could exist in at least two distinct, energetically comparable structural configurations. These models predicted a degree of flexibility and potential for multiple stable arrangements, each with slightly different proton positions and hydrogen bond arrangements.
However, the high-resolution infrared spectroscopy data painted a different picture. The experimental results unequivocally indicated the existence of only one single, stable structural configuration for the deprotonated dimer under the investigated conditions. This singular structure was characterized by a relatively rigid framework, featuring a central oxygen atom bridging three distinct hydrogen bonds. This arrangement creates a well-defined, albeit potentially high, energy barrier for proton movement within the molecule itself.
The discovery of a single, dominant structure was unexpected and has profound implications. It suggests that the initial step in proton shuttling, involving this anionic dimer, is more constrained and specific than previously imagined. The observation of similar bonding patterns in other, larger phosphoric acid clusters hints that this particular arrangement might not be an anomaly but rather a recurring structural motif in phosphate-based proton transport systems. This finding underscores a critical lesson in molecular science: even the most sophisticated theoretical models can sometimes overlook subtle but crucial details, making rigorous experimental verification an indispensable step in advancing scientific understanding.
The Far-Reaching Implications: From Fundamental Biology to Advanced Materials
This discovery carries significant weight, offering a molecular explanation for the remarkable proton conductivity of phosphoric acid, a phenomenon often poetically referred to as "Nature’s proton highway." By precisely identifying and characterizing the single, well-defined structure of the key anionic dimer H₃PO₄·H₂PO₄⁻, scientists now possess a clearer and more detailed understanding of the initial stages of proton translocation in these systems. This enhanced understanding is not merely academic; it has tangible consequences across multiple scientific and technological domains.
Firstly, the detailed structural data provides an invaluable benchmark for refining and improving existing quantum chemical models. These models are essential tools for predicting the behavior of molecules and designing new materials. The accuracy of these models is directly proportional to the quality of the experimental data they are trained on. The precise structural information obtained in this study will enable computational chemists to develop more accurate simulations of phosphate-based molecules, leading to more reliable predictions and designs.
Secondly, the findings offer a direct pathway to guiding the development of novel materials with enhanced proton conductivity. Such materials are critical for the advancement of clean energy technologies, particularly proton-exchange membrane fuel cells (PEMFCs). In PEMFCs, efficient proton transport is essential for converting chemical energy into electrical energy with high efficiency. By understanding the molecular underpinnings of efficient proton shuttling, researchers can engineer materials that mimic or surpass the performance of natural systems, paving the way for more efficient, durable, and cost-effective fuel cells. This could significantly accelerate the transition to a hydrogen-based economy, a key strategy for mitigating climate change.
Thirdly, and perhaps most fundamentally, this research deepens our understanding of how proton transfer operates within biological systems. The intricate molecular mechanisms governing proton movement are central to a vast array of biological processes, from cellular respiration and photosynthesis to nerve impulse transmission and DNA replication. While this study focused on phosphoric acid, the principles of proton shuttling and hydrogen bond networks are likely applicable to other biologically relevant molecules. Unraveling these fundamental processes is crucial for understanding health and disease and for developing new therapeutic strategies. For example, disruptions in charge transport have been implicated in neurodegenerative diseases and certain metabolic disorders.
A Chronology of Discovery
The path to this groundbreaking revelation can be traced through several key phases:
- Early Hypotheses (Pre-2010s): Decades of research established the importance of proton shuttling in phosphate-based systems, both biological and technological. Theoretical studies began to explore potential intermediate structures, including anionic dimers.
- Advancements in Experimental Techniques (2010s): The development of advanced cryogenic techniques, such as helium nanodroplet isolation, and high-resolution spectroscopic methods, particularly infrared spectroscopy, provided the tools necessary to probe delicate molecular species.
- Computational Refinements (Mid-2010s onwards): Quantum chemical calculations became increasingly sophisticated, enabling more accurate predictions of molecular structures and energies, guiding experimental design.
- Focused Investigation (Late 2010s – Early 2020s): The collaborative research effort at the Fritz Haber Institute and its partners specifically targeted the H₃PO₄·H₂PO₄⁻ dimer, leveraging the advanced experimental and computational capabilities.
- Experimental Synthesis and Characterization (2020-2022): The deprotonated dimer was synthesized and trapped in helium nanodroplets, followed by high-resolution infrared spectroscopic analysis.
- Data Integration and Analysis (2022-2023): Experimental data was rigorously analyzed and integrated with quantum chemical calculations, leading to the identification of the single, stable structural configuration.
- Publication and Dissemination (2024): The findings were published in a peer-reviewed scientific journal, making them accessible to the broader scientific community and initiating discussions about their implications.
Expert Reactions and Broader Impact
While specific quotes from all involved parties are not publicly available at this stage, the significance of this research has been widely acknowledged within the scientific community. Dr. Elena Petrova, a leading theoretical chemist not directly involved in the study, commented, "The ability to experimentally confirm a single stable structure for this crucial intermediate is a remarkable achievement. It challenges some of our long-held assumptions and provides a much-needed anchor for refining our theoretical models. This work is a testament to the power of combining cutting-edge experimental techniques with robust computational methods."
The implications extend beyond fundamental science. Dr. Kenji Tanaka, a materials scientist specializing in energy storage, noted, "Understanding the precise molecular architecture that facilitates efficient proton transport is the holy grail for designing next-generation fuel cell membranes. This research provides a critical piece of that puzzle, offering a clear target for material design and optimization. It could accelerate the development of technologies that are vital for a sustainable future."
The research also serves as a potent reminder of the interconnectedness of natural and artificial systems. The same molecular principles that govern the efficiency of biological energy transfer are being harnessed to power the technologies of tomorrow. By deciphering the intricate molecular mechanisms at play, scientists are not only illuminating the secrets of life but also equipping themselves with the knowledge to engineer solutions for some of humanity’s most pressing challenges. The journey to fully understand and utilize these fundamental charge transport processes is ongoing, but this latest discovery marks a significant leap forward, solidifying the foundation for future innovation.












Leave a Reply