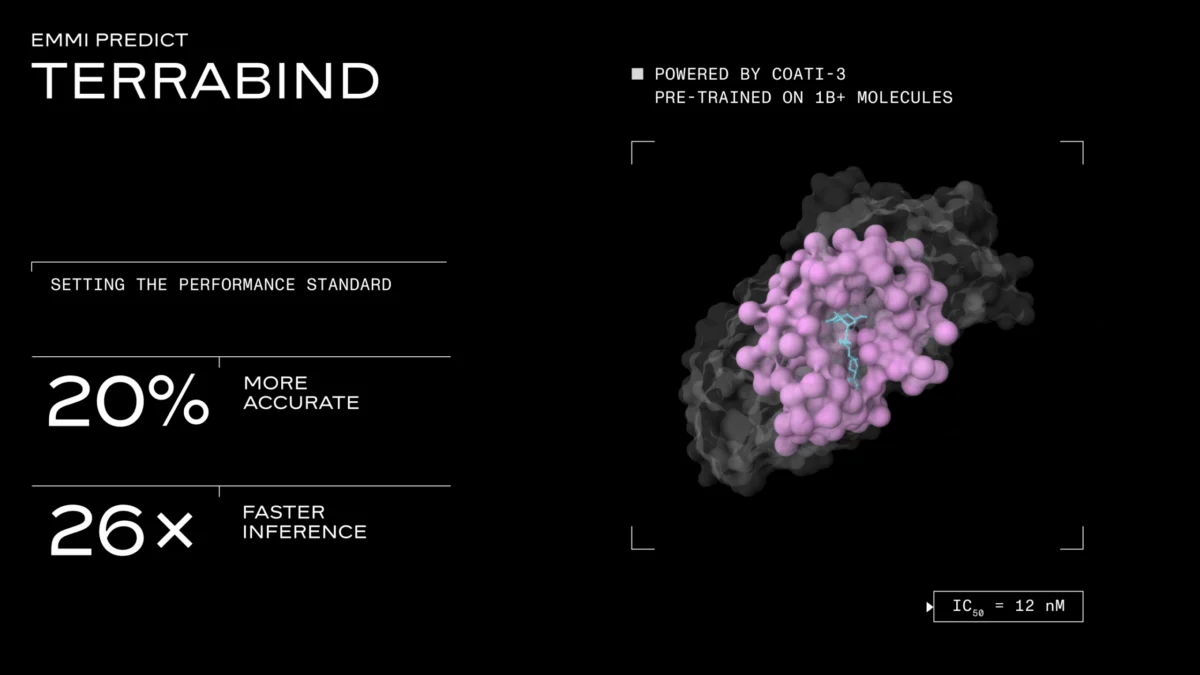
In a significant development poised to reshape the landscape of AI-driven drug discovery, Terray Therapeutics, a Los Angeles-based startup, has unveiled TerraBind, a novel potency prediction model that claims a staggering 26x faster inference speed and 16-20% improved accuracy compared to leading diffusion-based models like Boltz-2. Crucially, TerraBind also introduces built-in uncertainty quantification, a feature Terray’s leadership asserts is vital for practical application in real-world drug discovery programs. This announcement positions TerraBind as a potential disruptor, addressing what Terray identifies as a fundamental bottleneck in current AI architectures for small-molecule binding affinity prediction.
The Evolution of AI in Drug Discovery and Boltz-2’s Benchmark
The application of artificial intelligence in drug discovery has witnessed rapid advancements in recent years, promising to accelerate the identification of novel therapeutic candidates and reduce the exorbitant costs and timelines associated with traditional methods. A pivotal moment occurred last June when a collaboration between MIT and Recursion Pharmaceuticals led to the release of Boltz-2. This open-source AI model marked a significant milestone, becoming the first of its kind to jointly predict protein structure and small-molecule binding affinity. Building on the momentum of its widely adopted predecessor, Boltz-1, Boltz-2 achieved accuracy comparable to highly sophisticated, physics-based free-energy perturbation methods, but at an astounding speed — roughly 1,000 times faster. Its open-source nature further democratized access to advanced computational tools, fostering innovation across the industry.
Despite the breakthroughs represented by Boltz-2 and similar models, Terray Therapeutics argues that a common architectural flaw inherently limits their utility in the demanding environment of drug development. According to Dr. Jacob Berlin, co-founder and CEO of Terray, the prevailing approach among most structure-based potency prediction models, including Boltz-2 and those developed by other prominent players like Isomorphic Labs, shares a sequential structure: encode the molecule and protein, generate a detailed all-atom 3D pose via a diffusion step illustrating how the molecule fits the protein, and then derive the potency prediction from that generated pose.
Unmasking the Bottleneck: The Diffusion Dilemma
Dr. Berlin succinctly articulates Terray’s critique with a compelling analogy: "It’s almost like you’re trying to decide the income of people by first looking at pictures of how they dress, instead of directly trying to predict the income. We only care how well the molecule works." This perspective highlights Terray’s belief that the diffusion-based pose generation step, while providing visually rich structural data, is an indirect and computationally expensive detour when the ultimate goal is potency prediction. Terray’s internal analysis suggests this diffusion step accounts for approximately 96% of the total computational cost in such models.
The "age of AI abundance," as Dr. Berlin terms it, presents a paradox: while AI can generate and predict properties for millions of molecules, the practical constraint remains the ability to physically synthesize and test only a fraction of these – typically tens or hundreds. "Picking those correctly is actually the entire value proposition," Berlin emphasizes. This underscores the critical need for not just high-throughput prediction, but also highly accurate and reliable selection of the most promising candidates to maximize the efficiency of experimental validation.
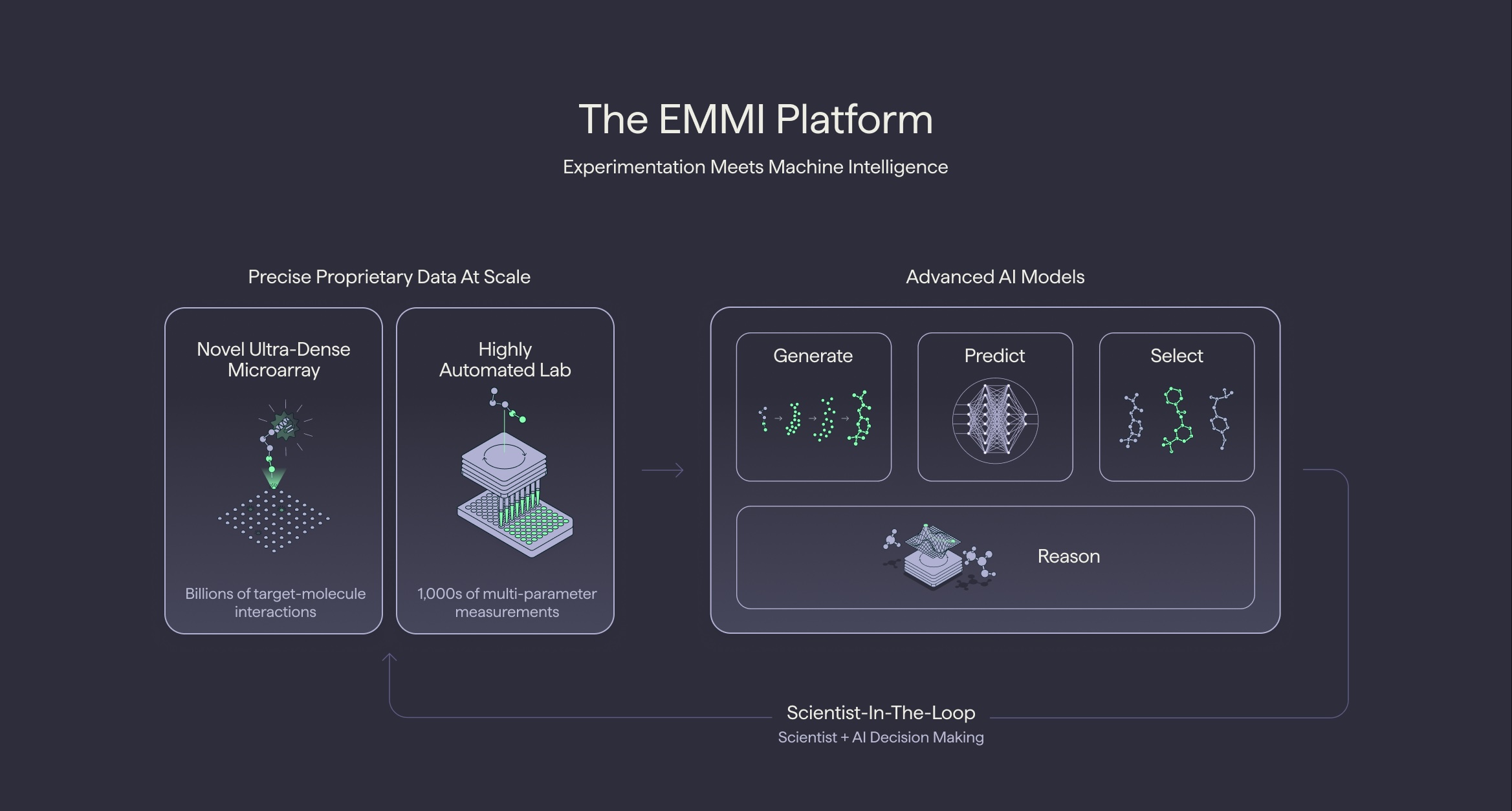
TerraBind’s Architectural Innovation: Direct Potency Prediction
Terray’s core architectural insight for TerraBind lies in the complete elimination of this diffusion-based pose generation step. Instead, TerraBind leverages Terray’s proprietary chemical foundation model, COATI (which has its code publicly available on GitHub), to encode molecules and proteins into a shared representation space. This encoded information is then directly fed into a Pairformer module. This module is designed to resolve structural features and predict potency without ever generating an explicit, all-atom 3D picture of the protein-ligand complex.
Specifically, TerraBind-Struct operates at a coarse-grained level, focusing only on protein Cβ atoms and ligand heavy atoms. This avoids the computationally intensive iterative denoising and all-atom coordinate generation characteristic of diffusion models. The result of this streamlined approach is a dramatic reduction in inference time. Benchmarked end-to-end on a single A6000 GPU (196 tokens, 10 samples), TerraBind achieves an inference speed of 1.04 seconds per complex, a substantial improvement over Boltz-2’s 27.8 seconds per complex. This 26x speed advantage is not merely incremental; it fundamentally changes the scale at which computational screening can be performed.
Transforming Virtual Screening: Million-Scale Efficiency
The practical implications of TerraBind’s speed are profound. Terray integrates TerraBind into a sophisticated two-tiered screening pipeline. The first tier utilizes an ultra-fast, sequence-only model, TerraBind-Seq, which Terray claims is 20,000 times cheaper than public alternatives. This initial screening rapidly sifts through vast chemical spaces, identifying a manageable subset of promising candidates. These candidates then undergo a more rigorous structure-based evaluation using TerraBind-Struct. Even at this second tier, TerraBind-Struct remains 20 times cheaper and faster than Boltz-2. The cumulative effect of this pipeline is the ability to conduct virtual screening at a million-molecule scale, a significant leap from the industry standard which typically processes thousands of molecules. This expansion of screening capacity dramatically increases the probability of discovering novel hits and leads.
Beyond Speed: Demonstrating Superior Accuracy
A natural concern with stripping out a major architectural component like diffusion-based pose generation is the potential compromise on accuracy. However, Terray’s internal benchmarks suggest the opposite. "You would worry that you’re trading speed and scale for quality, but we don’t," Berlin stated. "The quality is best in class: 20% better than Boltz, roughly on par with what Isomorphic put out, but it does 26 times more and quantifies the uncertainties."
Terray put TerraBind through rigorous testing on two independent held-out test sets: the publicly recognized CASP16 binding affinity challenge and a proprietary dataset comprising over 25,000 assay readouts spanning 18 distinct targets. On both datasets, TerraBind demonstrated a 16-20% improvement in Pearson correlation relative to Boltz-2. Specifically, on the proprietary dataset, TerraBind outperformed Boltz-2 on 15 out of 18 targets. Terray attributes these accuracy gains to an unexpected benefit: by removing the diffusion head, the Pairformer module is "forced" to fully resolve structural features itself, rather than offloading that complex task to a downstream generative step. While TerraBind produces coarse-grained geometry rather than the publication-quality, all-atom poses generated by diffusion models, Terray asserts that this level of structural detail is entirely sufficient for reliable pose prediction and affinity ranking, aligning with the practical needs of drug discovery.
It is important to note that while the CASP16 results are publicly verifiable, the proprietary dataset of 25,000 assay readouts is not. However, the open-source nature of Terray’s COATI foundation model code provides a level of transparency regarding its underlying methodology.
The Game-Changing Element: Uncertainty Quantification
While speed and accuracy are crucial metrics, Dr. Berlin contends that the feature most models entirely lack – and the one that holds the greatest practical value in drug discovery – is the ability to quantify the model’s confidence in each prediction. In conventional drug discovery workflows, after generating and scoring a multitude of molecules, researchers typically employ a "greedy" selection strategy, picking the top-ranked candidates for synthesis and experimental testing.
However, this approach carries a significant risk. As Terray explains in its EMMI platform documentation, model uncertainty is often correlated. If, for instance, the top 25 molecules all share a common structural motif that the model inadvertently overvalues, a greedy selection could result in a batch where every molecule either succeeds or fails together. While enforcing structural diversity among selected candidates can mitigate some risks, it doesn’t account for shared uncertainties that might exist even among structurally distinct molecules. This can lead to entire experimental campaigns yielding little actionable data, slowing down drug programs.
TerraBind addresses this critical challenge with TerraBind-Epinet, a specialized variant that employs epistemic neural networks to estimate model uncertainty for every prediction. This crucial uncertainty information is then fed into an acquisition function known as Expected Maximum (EMAX). EMAX intelligently constructs what Dr. Berlin describes as "hedged baskets" of molecules for synthesis. "The math will tell you: if this molecule is what I think it is, this other molecule is unlikely to be what I think it is, and vice versa," Berlin explains. "So you can build hedged baskets of molecules where if number one works, number ten won’t, which is fine, you got a winner, but if number two doesn’t, number nine probably will. You get rid of the ‘whole set’ type outcome that’s very common in drug discovery, where you make a bunch of molecules and you don’t move your program far enough along."
Terray’s internal benchmarking demonstrates the power of this approach, showing a 6x improvement in selection performance over traditional greedy methods. Retrospective studies further indicate roughly 3x savings in time and cost, directly translating to more efficient and successful drug discovery campaigns. This capability to quantify and leverage uncertainty moves AI from merely predicting to actively guiding experimental design, optimizing resource allocation, and significantly de-risking early-stage drug development.
From Model Advantage to Clinical Impact: The Road Ahead
Dr. Berlin is pragmatic about the journey from computational advantage to tangible drug development outcomes. "Going from model advantage to drug development advantage is actually non-trivial," he admits. "Everybody always skips it: ‘Oh, my model won on this benchmark, problem solved.’ But you have to imbue it in your internal workflows and connect it all the way through."
Terray’s internal pipeline, which is currently focused on immunology, has not yet reached the clinical stage. Therefore, the platform’s claims remain preclinical. However, the company has already achieved significant milestones through strategic partnerships with major pharmaceutical entities, including Bristol Myers Squibb, Gilead Sciences, and Calico. These collaborations suggest a strong industry validation of Terray’s technological approach and potential.
Terray’s Enduring Moat: Proprietary Data and Integrated Workflow
While Terray anticipates that its architectural innovations, particularly the concept of skipping the diffusion step, may be rapidly adopted by competitors, Dr. Berlin believes the company possesses a durable competitive advantage rooted in two key areas: proprietary data and deep workflow integration.
First, Terray claims to have amassed the largest global database of small-molecule binding data, currently exceeding 14 billion unique target-molecule interactions. This immense dataset is not static; it grows at an astonishing rate of approximately 1 billion interactions per quarter. This data is generated through Terray’s proprietary ultra-dense microarray hardware, which is capable of measuring interactions between 32 million molecules and targets of interest within minutes. Crucially, this hardware allows Terray to generate measurements in vast areas of chemical space where no public datasets currently exist. Berlin asserts, "Almost everybody in the AI space except us has focused their work in areas of known chemical space, because they don’t have proprietary hardware and data at the scale we do." Terray’s EMMI platform blog elaborates on this, stating, "Many current ‘AI drugs’ are strikingly similar to previous patents as the models perform well when tasked with finding a better solution to a previously addressed challenge." By venturing into uncharted chemical territory, Terray aims to discover truly novel drug candidates rather than merely optimizing existing ones.
Second, Terray’s advantage stems from its deeply integrated workflow. COATI, the chemical foundation model underpinning TerraBind’s speed, has been in continuous development for years and has been trained on over one billion molecules. It serves as the backbone not just for prediction, but for the entire generate-predict-select cycle that Terray’s scientists utilize daily. Berlin notes a progressive shift within Terray, where scientists are increasingly interacting with the EMMI platform through an AI agent, articulating desired outcomes and allowing the agent to plan and execute the complex workflow, rather than manually sequencing individual models. This level of integration transforms AI from a tool into an intelligent partner in the discovery process.
Investment, Vision, and the Future of Drug Discovery
Terray Therapeutics, founded in 2019, predates both the COVID-19 pandemic and the widespread ChatGPT-fueled AI wave. The company has successfully raised over $200 million in funding, with NVIDIA and Bedford Ridge co-leading its Series B round. NVIDIA is not only an investor but also a technology development partner, underscoring the strategic importance of Terray’s work within the broader AI ecosystem. The TerraBind announcement itself is being co-promoted with NVIDIA, highlighting the collaborative nature of this technological leap.
Dr. Berlin observes the ongoing debate between AI hype and reality as an inherent part of the lengthy drug development timeline. He maintains a long-term perspective on AI’s transformative potential. "In 15 years, when you look back, it’ll be like, ‘Whoa, the drug discovery process changed, the success rate changed, the time to get through all this changed,’" he predicts. "But as you go through it, right up against the frontier, you’ll have people arguing all the way through because one drug will fail, two will succeed, and you’ll have the whole cycle again."
Terray’s TerraBind represents a bold step forward, challenging established AI paradigms and offering a vision for a more efficient, accurate, and strategically guided drug discovery process. By prioritizing direct potency prediction, enhancing speed, and integrating uncertainty quantification, Terray aims to accelerate the journey from novel molecular insights to life-changing therapies, navigating the complexities of drug development with a new level of computational intelligence.














Leave a Reply