A groundbreaking discovery by a research team spearheaded by David Reverter, a distinguished scientist at the Universitat Autònoma de Barcelona (UAB), has illuminated the intricate molecular mechanism governing bacterial cell division. The findings, published in the prestigious journal Nature Communications, pinpoint the pivotal role of the MraZ protein in orchestrating this fundamental biological process by binding to the critical dcw gene cluster. This revelation offers unprecedented insight into how bacteria, the ubiquitous architects of life on Earth, precisely control their replication.
The Genesis of Bacterial Replication: The Role of the dcw Operon
Cell division, the fundamental engine driving the proliferation of all living organisms, is a symphony of precisely coordinated molecular events. In the bacterial realm, this vital process is largely orchestrated by a dedicated genetic command center known as the dcw operon. This operon, a densely packed cluster of genes, carries the blueprint for producing a suite of proteins indispensable for both the physical act of dividing and the construction of the robust scaffolding that defines the bacterial cell wall. Understanding how this operon is activated and regulated is therefore paramount to comprehending bacterial life.
For decades, scientists have recognized the dcw operon as a master regulator of bacterial growth. However, the precise molecular details of its activation have remained elusive. The operon’s genes are switched on through the action of transcription factors, specialized proteins that act as molecular keys, binding to specific DNA sequences known as promoters. These promoters serve as ignition points, signaling where the cellular machinery should begin transcribing the genetic code into functional proteins. The promoter region of the dcw operon, in particular, has been a focal point of research due to its critical role in initiating the cascade of events leading to cell division.
MraZ: The Conductor of the Cell Division Orchestra
Among the key players in this genetic orchestra, the MraZ protein has emerged as a critical conductor. Intriguingly, MraZ is not only a transcription factor but also the very first gene within the dcw operon in virtually all bacterial species. This auto-regulatory loop suggests a sophisticated feedback mechanism where the output of the operon (the proteins for cell division) also controls its own initiation. When MraZ successfully activates the dcw operon, it sets in motion the production of the essential proteins that enable bacteria to split into two daughter cells. Consequently, MraZ functions as the central regulator, dictating the pace and execution of cell division across a vast spectrum of bacterial life.
The significance of this regulatory system cannot be overstated. Bacterial cell division is a rapid and efficient process, crucial for everything from the colonization of diverse environments to the complex symbiotic relationships bacteria form with other organisms. Dysregulation of this process can lead to profound consequences, impacting microbial populations, disease progression, and even ecological balances.
Illuminating the Molecular Dance: Advanced Imaging Techniques
The breakthrough in understanding the MraZ-driven regulation came through the meticulous work of David Reverter’s research group at the UAB. Leveraging cutting-edge structural biology techniques, including X-ray crystallography and cryo-electron microscopy (cryo-EM), the team was able to visualize the molecular choreography of MraZ binding to the dcw operon’s promoter with unprecedented clarity.
X-ray crystallography, a technique that uses X-rays to determine the atomic and molecular structure of a crystal, has long been a cornerstone of structural biology. Cryo-electron microscopy, on the other hand, allows researchers to image biological molecules at near-atomic resolution by freezing them in a thin layer of ice, preserving their native state. The synergistic application of these powerful methods provided a dynamic, high-resolution view of the molecular interactions at play.
The research focused on Mycoplasma genitalium, a bacterium chosen for its notably compact genome, making it an ideal model organism for detailed genetic and structural studies. This bacterium, frequently encountered in laboratory research due to its genetic simplicity, offered a clear window into the fundamental mechanisms of bacterial cell division.
Atomic Precision: Unveiling the MraZ-DNA Interaction
The promoter region of the dcw operon, the target of MraZ’s regulatory action, is characterized by a specific DNA sequence. It comprises four repetitive segments, often referred to as "boxes," each consisting of six nucleotide bases. These repetitive DNA sequences are crucial for recognizing and binding transcription factors.
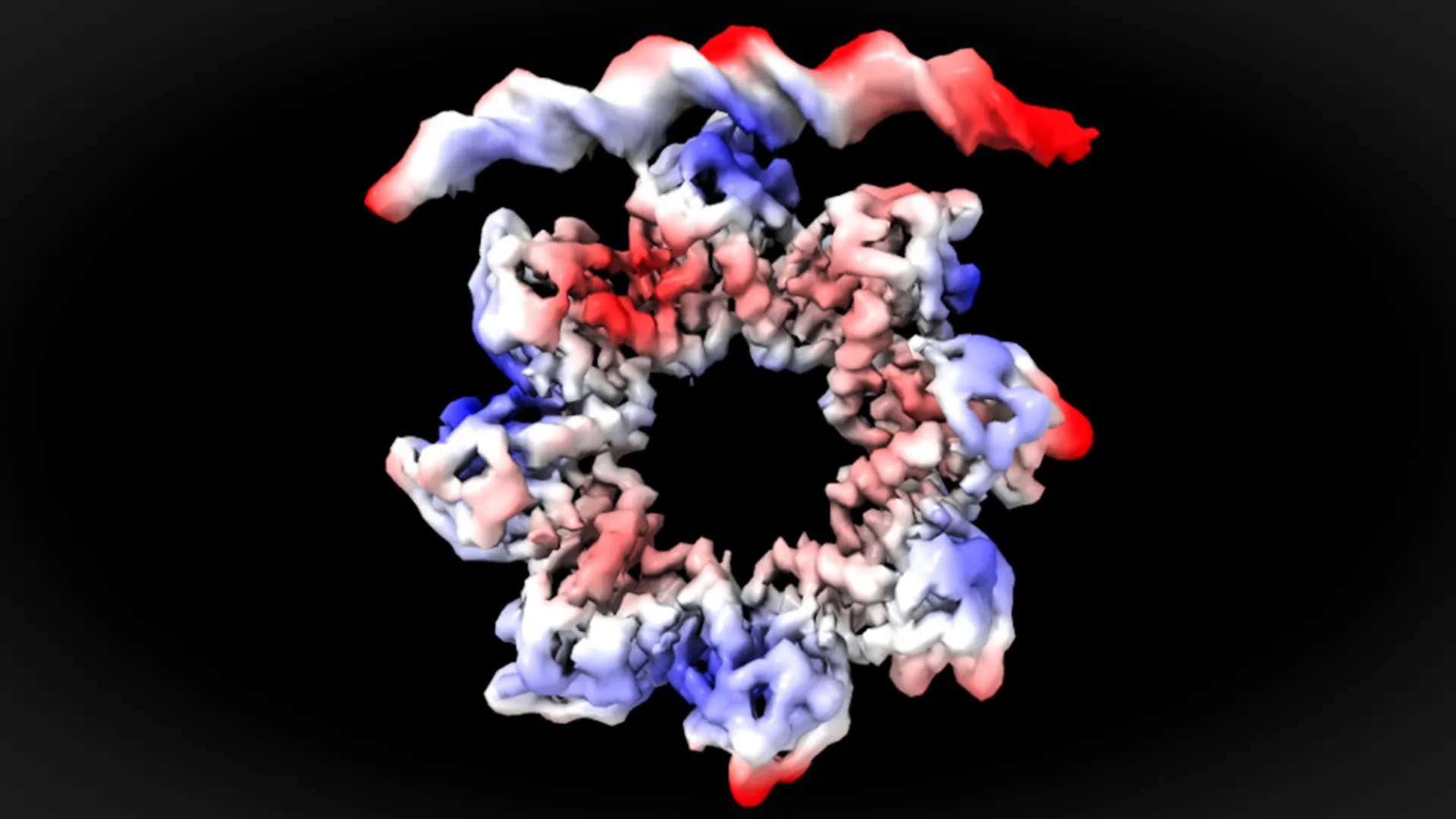
Through meticulous cryo-EM analysis, the UAB team was able to observe the precise manner in which the MraZ protein engages with these DNA sequences. Their findings revealed a surprising conformational change within the MraZ protein itself, a necessary adaptation for effective binding.
"This is a surprising observation," explained David Reverter. "The MraZ protein is an octamer formed by eight identical subunits joined in the shape of a donut, but with a curvature that would never allow the union with the four ‘boxes’ of the promoter. However, to regulate cell division we see how the donut breaks and deforms in such a way that four of the subunits can join the four boxes of the promoter."
This remarkable structural plasticity of the MraZ protein underscores the sophisticated evolutionary solutions that nature employs to ensure essential biological processes occur with fidelity. The octameric donut-like structure of MraZ, normally ill-suited to the linear arrangement of the DNA boxes, undergoes a dramatic reconfiguration. It essentially "breaks" and reshapes itself, allowing four of its subunits to precisely dock with the four repetitive DNA sequences on the promoter. This precise alignment is the critical step that triggers the transcription of the downstream genes, initiating the cell division cascade.
A Quantum Leap in Fundamental Understanding
The ability to directly visualize the atomic-level interaction between MraZ and the promoter DNA that initiates cell division marks a significant leap forward in the field. Prior to this research, scientists relied heavily on indirect evidence derived from biochemical experiments and computational modeling to piece together the workings of this regulatory system. While these methods provided valuable insights, they lacked the direct, visual confirmation that structural biology can offer.
This direct visualization provides empirical validation for theoretical models and opens new avenues for understanding the nuances of transcriptional regulation in bacteria. It moves beyond inferential knowledge to concrete, observable evidence of molecular mechanics.
A Universal Mechanism? Implications for Bacterial Diversity
The implications of this discovery extend far beyond the specific bacterium studied. David Reverter expressed confidence that the regulatory system identified is likely conserved across a vast majority of bacterial species. "This is universal to most bacteria, since all MraZ proteins are very similar, have the same octamer structure, and the DNA sequences of the promoters of the operons that regulate cell division are also similar," Reverter stated.
This universality suggests that the fundamental mechanism of MraZ-mediated dcw operon activation is a deeply ingrained feature of bacterial biology, honed over millions of years of evolution. This conserved nature has profound implications for various fields, including medicine, biotechnology, and fundamental biology.
International Collaboration Fuels Discovery
The success of this research was a testament to robust international collaboration. The UAB team, comprising researchers from the Institute of Biotechnology and Biomedicine and the Department of Biochemistry and Molecular Biology, worked in close partnership with the ALBA synchrotron, a Spanish research facility providing powerful X-ray sources, and the cryo-electron microscopy service at the Institute of Genetics and Molecular and Cellular Biology in Strasbourg, France. This synergistic effort, combining expertise and cutting-edge infrastructure, was instrumental in achieving the high-resolution structural data.
Broader Impact and Future Directions
The deciphering of the MraZ regulatory mechanism has far-reaching implications.
Antimicrobial Development: Understanding how bacteria initiate cell division is a critical target for developing new antimicrobial therapies. Many existing antibiotics target cell wall synthesis, a process directly influenced by the dcw operon. By precisely understanding how MraZ controls this operon, researchers can potentially design novel compounds that specifically inhibit MraZ function or disrupt its interaction with the DNA, thereby halting bacterial replication. This could lead to the development of antibiotics that are more targeted and less prone to resistance.
Biotechnology and Synthetic Biology: In the realm of biotechnology, controlling bacterial growth is essential for various industrial processes, such as fermentation for producing biofuels, pharmaceuticals, and food products. A deeper understanding of cell division regulation could allow for more precise control over bacterial cultivation, optimizing yields and improving efficiency. Furthermore, synthetic biologists could leverage this knowledge to engineer bacteria with specific growth characteristics or to create novel microbial systems for diverse applications.
Fundamental Biological Research: This discovery deepens our fundamental understanding of gene regulation and protein-DNA interactions, cornerstones of molecular biology. It provides a detailed case study of how complex biological processes are controlled at the molecular level, offering insights applicable to other regulatory systems in bacteria and even in more complex organisms. The observed structural plasticity of MraZ serves as a compelling example of protein adaptability in response to environmental cues or DNA binding partners.
Ecological Significance: Bacteria are the most abundant life forms on Earth, playing critical roles in virtually every ecosystem. Their ability to rapidly divide and adapt is key to their ecological success. Understanding the regulation of this fundamental process provides a window into the dynamics of microbial communities, their interactions, and their impact on global biogeochemical cycles.
The research team anticipates that future studies will delve deeper into the specific structural changes that MraZ undergoes and explore potential allosteric regulators that might influence this transformation. Further investigations into the variations of MraZ and dcw promoters across different bacterial species will also shed light on the evolutionary trajectory of this crucial regulatory system. This ongoing research promises to continue unraveling the intricate molecular symphony that governs bacterial life, with profound implications for science and human well-being.











Leave a Reply