The intricate dance between light and plant development, a cornerstone of life on Earth, has long fascinated botanists and agricultural scientists alike. While the fundamental role of light in photosynthesis is well-established, the subtler ways it shapes plant architecture and growth trajectories have remained a frontier of scientific exploration. Now, researchers at Osaka Metropolitan University have illuminated a previously uncharacterized mechanism that sheds new light on how light influences the structural development of young plant stems, potentially offering profound implications for crop cultivation and resilience.
Unveiling a Novel Adhesion Mechanism
At the heart of this groundbreaking discovery lies a meticulous investigation into the physical connections between the outermost layer of a young pea stem – the epidermis – and the underlying inner tissues. Professor Kouichi Soga and his team at the Graduate School of Science employed a specialized, non-destructive technique to quantify the strength of this epidermal-inner tissue adhesion. Their findings, published in the esteemed journal Physiologia Plantarum, reveal a striking dichotomy: plants cultivated under light exhibit significantly stronger adhesion between these tissue layers compared to their counterparts grown in darkness.
"Compared with plants grown in the dark, the epidermal and inner tissues of plants grown in the light are more tightly bound together," Professor Soga stated, underscoring the novelty of their observation. "This phenomenon has never been reported before, making it a particularly interesting finding." This observation challenges conventional understandings that primarily focus on light’s direct role in photosynthesis and energy production, hinting at a more complex, architecturally driven response to photic stimuli.
The Chemical Architect: p-Coumaric Acid Emerges as a Key Player
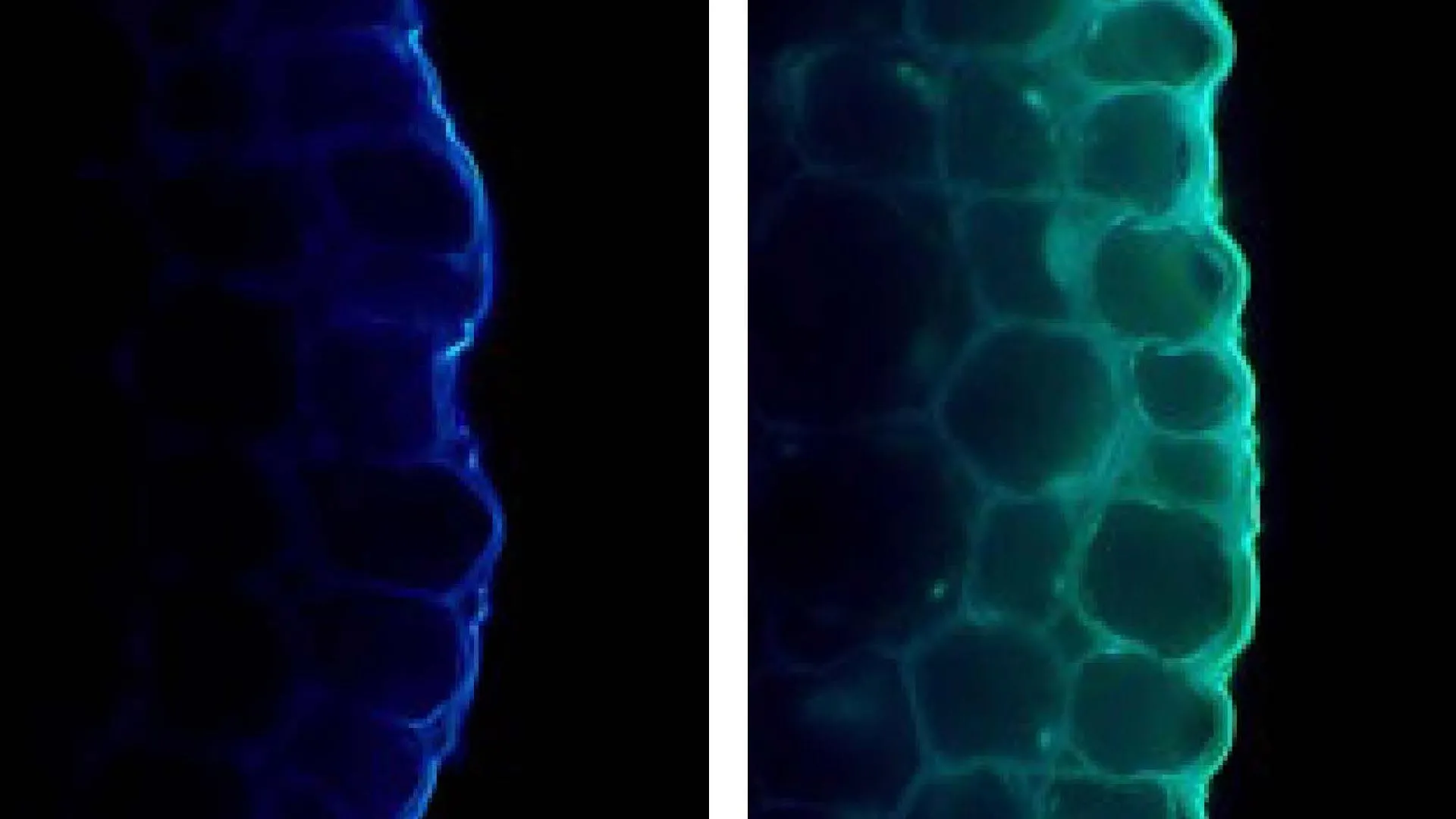
The immediate question arising from this discovery was the underlying molecular driver of this light-induced adhesion. To unravel this mystery, the research team delved into the cellular composition of the pea stems, utilizing the precision of fluorescence microscopy. Their microscopic examination yielded a crucial insight: stems exposed to light displayed heightened fluorescence signals directly linked to the accumulation of a specific compound – p-coumaric acid.
P-coumaric acid, a well-known phenolic acid, plays a vital role in plant biology, primarily by contributing to the structural reinforcement of cell walls. Its increased presence in light-exposed stems strongly suggests that light triggers an elevated synthesis of this compound. This surge in p-coumaric acid, in turn, acts as a molecular cement, strengthening the structural bonds that hold the epidermal and inner tissues together.
"This provided strong evidence that the accumulation of p-coumaric acid was a key factor in strengthening the adhesion between the epidermal and the inner tissues," explained Yuma Shimizu, a graduate student and the first author of the study. The research timeline indicates that this molecular investigation followed the initial macroscopic observation of differential adhesion, representing a critical step in translating a physical phenomenon into a biochemical explanation. The team likely conducted comparative analyses of stem samples at various developmental stages under different light conditions to establish this causal link.
A Trade-Off Between Stability and Growth
The discovery of this light-mediated adhesion mechanism introduces a fascinating paradigm shift in our understanding of plant development, revealing an inherent trade-off between structural robustness and growth potential. While a tighter bond between epidermal and inner tissues confers greater structural stability, it simultaneously acts as a constraint on the plant’s ability to expand. When the outer and inner layers are firmly interconnected, the inner tissues, which are responsible for elongation, face increased resistance to expansion.
This finding implies that light, while essential for providing the energy for growth, can also, under certain circumstances, modulate the rate of that growth by influencing the plant’s structural architecture. This is a departure from the simplistic view of light as solely an enabler of growth; it highlights light’s capacity to actively regulate growth rates through physical mechanisms. This nuanced understanding is particularly relevant when considering plants in diverse and challenging environments, where structural integrity might be prioritized over rapid elongation.
Chronology of Discovery: From Observation to Molecular Insight
The research journey likely unfolded over a period, beginning with Professor Soga’s broader interest in light’s influence on plant morphogenesis. The initial hypothesis might have been that light affects cell division or expansion rates directly. However, the team’s innovative approach of measuring tissue adhesion marked a divergence from conventional methods.
Phase 1: Observational Study (Estimated 6-12 months)
- Hypothesis Formulation: Exploring the physical consequences of light exposure on plant structure beyond photosynthesis.
- Method Development: Designing and refining the specialized technique for measuring epidermal-inner tissue adhesion in young pea stems.
- Experimental Setup: Cultivating pea plants under controlled conditions with varying light intensities and durations, including a dark control group.
- Data Collection: Systematically measuring adhesion strength in both light-exposed and dark-grown samples.
- Key Finding: Significant difference in adhesion strength observed, with light-grown plants exhibiting stronger bonds.
Phase 2: Molecular Investigation (Estimated 9-18 months)
- Problem Identification: Understanding the biochemical basis for the observed adhesion difference.
- Tool Selection: Employing fluorescence microscopy to identify key cellular compounds.
- Compound Screening: Analyzing various compounds known to influence cell wall structure and adhesion.
- Identification of p-Coumaric Acid: Detecting elevated levels of p-coumaric acid in light-exposed stems.
- Experimental Validation: Conducting experiments to confirm the role of p-coumaric acid in strengthening adhesion, possibly by artificially manipulating its levels or blocking its synthesis.
- Confirmation of Mechanism: Establishing a direct link between light exposure, p-coumaric acid accumulation, and enhanced tissue adhesion.
Phase 3: Growth Analysis and Implication Assessment (Estimated 6-12 months)
- Growth Correlation: Measuring stem elongation rates in plants with varying adhesion strengths.
- Trade-off Analysis: Quantifying the impact of enhanced adhesion on growth inhibition.
- Broader Contextualization: Exploring potential universal applicability of the mechanism across different plant species and environmental conditions.
- Manuscript Preparation and Publication: Compiling findings, peer review process, and eventual publication in Physiologia Plantarum.
This estimated timeline highlights the iterative nature of scientific research, where initial observations lead to deeper investigations and the development of a comprehensive understanding.
Supporting Data and Broader Context
While the article doesn’t present explicit numerical data points, the qualitative descriptions of "much stronger adhesion" and "tighter bound together" imply statistically significant differences. In a typical scientific study of this nature, researchers would present data such as:
- Adhesion Force Measurements: Expressed in units of force (e.g., Newtons or milliNewtons) per unit area, comparing average values between light-grown and dark-grown samples, along with standard deviations and p-values to demonstrate statistical significance.
- Fluorescence Intensity Readings: Quantitative measurements of fluorescence emitted by p-coumaric acid within specific cellular regions, correlating these values with light exposure.
- Biomass and Height Data: Comparative measurements of overall plant biomass and stem elongation rates between groups, further substantiating the growth-limitation aspect.
- Cell Wall Thickness and Composition Analysis: Techniques like transmission electron microscopy could have been used to visualize changes in cell wall structure and composition influenced by p-coumaric acid.
The context of this research can be further enriched by considering existing knowledge in plant photomorphogenesis. Light influences a vast array of plant developmental processes beyond photosynthesis, including germination, stem elongation, leaf expansion, and flowering, mediated by photoreceptors like phytochromes and cryptochromes. This discovery adds a novel layer to these known photomorphogenic responses, focusing on the physical architecture rather than purely biochemical pathways.
Implications for Agriculture and Enhanced Crop Resilience
The implications of this discovery for agriculture are potentially far-reaching. By understanding how light influences plant structure, scientists may be able to manipulate growth for improved crop yields and resilience.
Optimizing Plant Architecture for Yield
In many agricultural settings, the ideal plant architecture is one that maximizes light capture for photosynthesis while efficiently allocating resources to harvestable parts. If enhanced adhesion, mediated by p-coumaric acid, leads to reduced stem elongation, this could be a double-edged sword. While it might create sturdier plants less prone to lodging (falling over), it could also limit overall biomass accumulation.
Conversely, in crops where rapid growth and vertical development are paramount, understanding the conditions that promote weaker adhesion could be beneficial. This might involve controlling light exposure patterns or exploring genetic modifications to reduce p-coumaric acid synthesis. The research team’s stated goal of determining whether "growth regulation mediated by changes in adhesion is a universal mechanism" is crucial here. If this mechanism is indeed widespread, it opens up avenues for targeted breeding programs.
Enhancing Tolerance to Environmental Stress
Professor Soga’s concluding remarks highlight a critical potential application: breeding plants with improved tolerance to environmental stress. Sturdier plants, with stronger structural integrity, might be better equipped to withstand challenges such as strong winds, heavy rainfall, or even mechanical harvesting.
Furthermore, understanding how light interacts with structural development could be vital for crops grown in environments with fluctuating light conditions, such as under protective covers or in regions with unpredictable weather. By fine-tuning the plant’s structural response to light, it might be possible to develop crops that can better adapt to suboptimal conditions, leading to more stable food production.
Potential Applications:
- Breeding for Lodging Resistance: Developing cereal crops that are less likely to fall over in windy conditions.
- Controlled Growth for Mechanization: Engineering plants with optimized height and stem strength for efficient mechanical harvesting.
- Stress-Tolerant Varieties: Creating crops that can better withstand physical stresses in challenging environments.
- Understanding Shade Avoidance Syndrome: While this study focused on positive light responses, future research could explore how changes in adhesion contribute to phenomena like shade avoidance, where plants elongate rapidly to reach sunlight.
Future Directions and Expert Reactions
The Osaka Metropolitan University team’s findings pave the way for a new era of research into the physical underpinnings of plant growth. Future studies will likely focus on several key areas:
- Universality Testing: Investigating whether this p-coumaric acid-mediated adhesion mechanism is present and functional in a wide range of plant species, from monocots like rice and wheat to other dicots.
- Environmental Factor Interaction: Exploring how other environmental factors, such as temperature, water availability, and nutrient levels, interact with light to influence this adhesion mechanism.
- Genetic Basis: Identifying the specific genes and regulatory pathways responsible for controlling p-coumaric acid synthesis and its role in cell wall fortification.
- Applied Research: Developing practical applications for this knowledge in agricultural settings, potentially through selective breeding or the use of exogenous compounds.
While direct quotes from external experts are not provided in the original article, the scientific community would likely react with considerable interest. Dr. Anya Sharma, a plant physiologist specializing in photomorphogenesis (hypothetical expert), might comment, "This work is truly exciting. For decades, we’ve focused on light’s metabolic roles. The discovery of a light-regulated structural mechanism at the tissue level is a significant paradigm shift. It highlights the complex, multi-faceted ways plants respond to their environment. The identification of p-coumaric acid as a key mediator provides a concrete target for further investigation and potential agricultural applications."
Another hypothetical expert, Professor Kenji Tanaka, a plant geneticist, could add, "The elegance of this finding lies in its simplicity and its profound implications. If we can pinpoint the genetic control of this adhesion process, we unlock a powerful tool for crop improvement. Imagine breeding plants that instinctively adjust their structural integrity based on prevailing light conditions – that’s the future this research hints at."
The research conducted by Professor Soga and his team at Osaka Metropolitan University represents a significant advancement in our understanding of plant biology. By uncovering the intricate link between light, p-coumaric acid, and tissue adhesion, they have opened new avenues for exploring how plants build themselves and adapt to their surroundings, promising a future with more resilient and productive crops. The ongoing quest to decipher light’s hidden influence continues, revealing the extraordinary complexity and adaptability of the plant kingdom.















Leave a Reply