Mitochondria, the indispensable powerhouses of the cell, are renowned for their critical role in energy production, fueling every cellular function. This vital role is underpinned by their possession of a unique genetic blueprint, mitochondrial DNA (mtDNA). Each eukaryotic cell harbors hundreds, if not thousands, of copies of this mtDNA, meticulously organized into compact structures known as nucleoids. For decades, scientists have observed a remarkable regularity in the spatial arrangement of these nucleoids within the mitochondrial matrix. This precise organization is not merely aesthetic; it is fundamental to ensuring the faithful inheritance of mtDNA during cell division and the equitable expression of its genes throughout the vast mitochondrial network. When this delicate balance is disrupted, the consequences can be severe, manifesting in a spectrum of debilitating metabolic and neurological disorders, including liver failure, encephalopathy, and age-related neurodegenerative conditions such as Alzheimer’s and Parkinson’s disease.
Unraveling a Century-Old Enigma: The Mystery of Nucleoid Spacing
The consistent and orderly distribution of mtDNA nucleoids within mitochondria has long presented a significant puzzle in cell biology. Despite their crucial importance, the precise mechanisms by which cells achieve and maintain this regular spacing have remained elusive. Existing hypotheses, often invoking processes like mitochondrial fusion and fission, or the action of molecular tethers, have proven insufficient. "Proposed mechanisms related to mitochondrial fusion, fission, or molecular tethering cannot explain it, since nucleoid spacing is maintained even when they are disrupted," explains Suliana Manley, a distinguished professor at the Laboratory of Experimental Biophysics (LEB) at EPFL. This observation highlights the inadequacy of current models to account for the robust nature of nucleoid organization.
A Paradigm Shift: The Rediscovery of Mitochondrial Pearling
In a breakthrough that promises to redefine our understanding of mitochondrial biology, Professor Manley and her postdoctoral fellow, Juan Landoni, have identified the long-sought mechanism responsible for nucleoid spacing. Their groundbreaking research, published recently in a leading scientific journal, points to a process that has historically been relegated to the fringes of scientific inquiry: "mitochondrial pearling." This phenomenon, characterized by transient, bead-like deformations of the mitochondrial network, has now been revealed as a dynamic and essential choreographer of mtDNA distribution.
Mitochondrial Pearling: A Dynamic Ballet of DNA Distribution
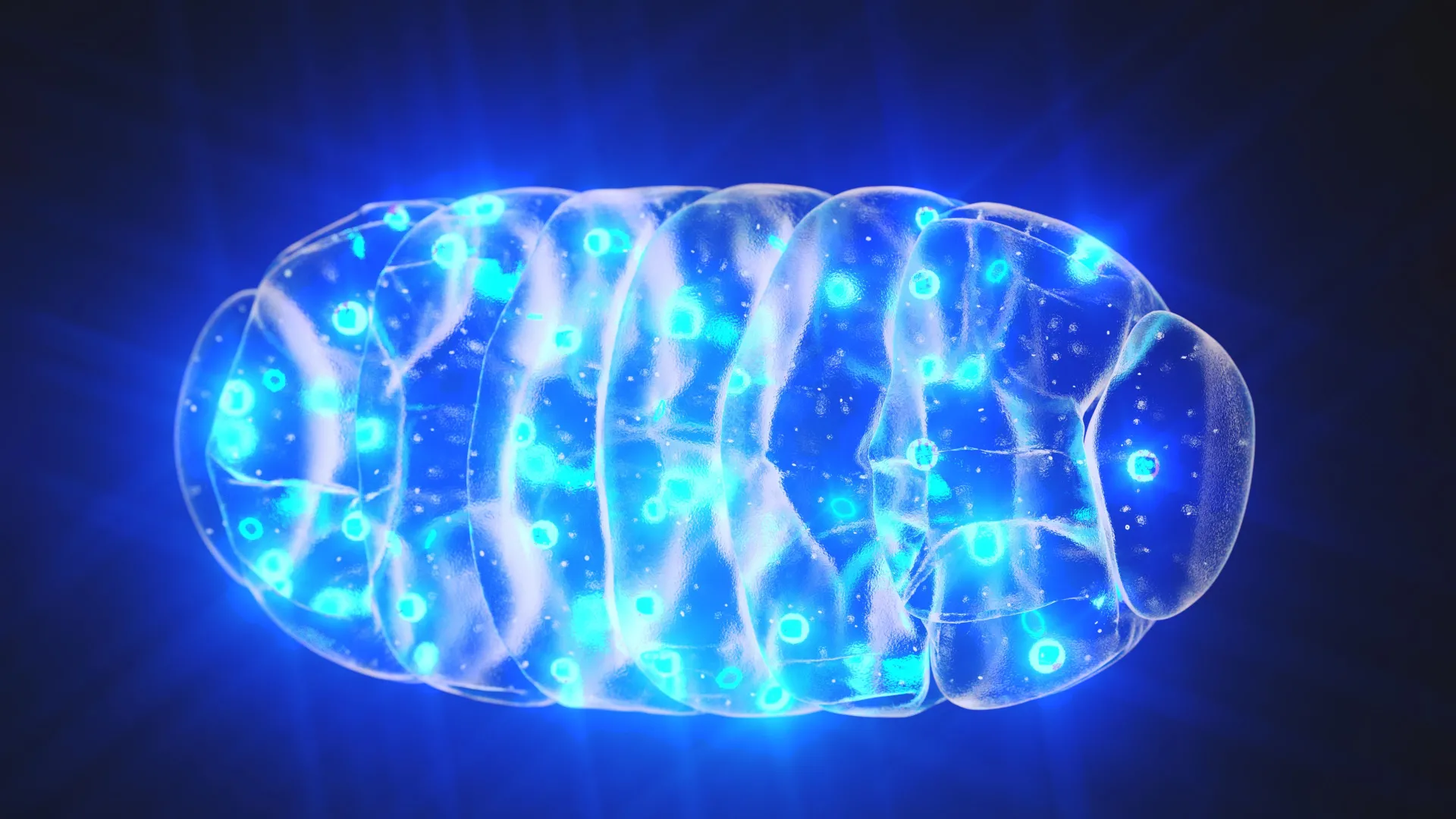
Mitochondrial pearling, a temporary morphing of the mitochondrial structure into a "beads on a string" formation, plays a pivotal role in the precise segregation and redistribution of mtDNA clusters. During these brief but frequent transformations, the tightly packed nucleoids are encouraged to separate and spread out more evenly. This dynamic redistribution is the key to maintaining the consistent, regular spacing that is critical for both mtDNA inheritance and gene expression. The process, it appears, is far from an anomaly; it is an elegant and energy-efficient biological solution.
Visualizing the Unseen: Advanced Microscopy Illuminates Mitochondrial Dynamics
To unravel the intricacies of mitochondrial pearling, the EPFL research team employed a sophisticated arsenal of cutting-edge imaging techniques. Their meticulous observations were conducted on living cells, allowing them to capture the dynamic behavior of mitochondria and their enclosed DNA in real-time. This multidisciplinary approach integrated super-resolution microscopy, which provides unparalleled detail of cellular structures, with correlated light and electron microscopy (CLEM), enabling the correlation of live-cell fluorescence imaging with high-resolution ultrastructural information. Complementing these were phase contrast microscopy techniques, which allowed for the visualization of cellular structures without the need for staining, thereby minimizing interference with natural biological processes.
These advanced tools were instrumental in enabling the researchers to track individual nucleoids, document rapid and transient changes in mitochondrial shape, and gain unprecedented insights into the internal organization of these vital organelles. The ability to observe these phenomena in living cells, rather than relying solely on static snapshots, was crucial for understanding the dynamic nature of pearling.
The Mechanics of Pearling: A Temporal and Spatial Analysis
Live-cell imaging revealed the remarkable frequency of pearling events, with mitochondria undergoing these transformations several times per minute. During these moments, the elongated, tubular mitochondria briefly constrict at regular intervals along their length, forming the characteristic "pearls." Intriguingly, the distance between these constrictions closely mirrors the typical spacing observed between nucleoids in their quiescent state.
While most of these bead-like segments were found to harbor a nucleoid near their center, the formation of pearling structures was not entirely dependent on the presence of mtDNA. This suggests that the physical framework for pearling might be established independently of the DNA payload.
As the pearling process unfolds, larger aggregations of nucleoids frequently undergo fragmentation, breaking down into smaller clusters. These smaller clusters then migrate and settle into adjacent "pearls." Upon the mitochondrion’s eventual return to its characteristic tubular morphology, the nucleoids remain separated, thereby preserving their even distribution throughout the organelle. This cyclical process ensures a constant state of organized dispersion.
Orchestrating the Dance: Calcium and Internal Membranes as Regulators
The research team also delved into the regulatory mechanisms that govern mitochondrial pearling. Through a series of genetic and pharmacological experiments, they pinpointed key factors that initiate and control this crucial process. Their findings indicate that the influx of calcium ions into the mitochondria serves as a potent trigger for pearling. Furthermore, internal membrane structures within the mitochondria appear to play a critical role in maintaining the physical separation of nucleoids during and after pearling events.
Conversely, when these regulatory factors are experimentally disrupted, a noticeable shift occurs: nucleoids tend to aggregate rather than maintaining their dispersed and evenly spaced arrangement. This observation underscores the delicate interplay between molecular signals and structural components in ensuring proper mtDNA organization.
A Historical Perspective: From Curiosity to Cornerstone of Biology
The significance of mitochondrial pearling extends beyond its newly elucidated function. The phenomenon itself has a long and curious history in scientific literature. "Since Margaret Reed Lewis first sketched mitochondrial pearling in 1915, it has largely been dismissed as an anomaly linked to cellular stress," notes Landoni. Lewis’s early observations, made with the microscopy techniques of her era, were largely overlooked or misinterpreted for nearly a century.
Now, over a hundred years later, this seemingly peculiar morphology is re-emerging as a fundamental and elegantly conserved mechanism at the very core of mitochondrial biology. The work by Manley and Landoni elevates mitochondrial pearling from a historical curiosity to a cornerstone of our understanding of how cells manage their internal genetic material.
Broader Implications: Health, Disease, and Therapeutic Avenues
The implications of this discovery are far-reaching, extending from fundamental cell biology to the understanding and treatment of human disease. The findings definitively demonstrate that cellular organization relies not solely on intricate molecular machinery but also on fundamental physical processes. This paradigm shift broadens our perspective on cellular regulation.
Understanding the precise mechanisms of mitochondrial pearling and its regulatory controls could unlock critical insights into the pathogenesis of a wide array of diseases intricately linked to mtDNA dysfunction. These include not only the aforementioned neurodegenerative and metabolic disorders but potentially also aspects of aging itself.
The potential therapeutic avenues are substantial. By identifying the molecular triggers and structural components involved in pearling, researchers may be able to develop novel strategies to correct or enhance mtDNA organization in diseased cells. This could involve pharmacological interventions aimed at modulating calcium signaling or targeting specific internal membrane proteins. Such advancements could pave the way for new therapeutic approaches to combat conditions that have, until now, presented significant challenges in treatment.
A Collaborative Endeavor: The Scientific Community’s Response
The scientific community has responded with considerable enthusiasm to the EPFL team’s findings. Leading figures in mitochondrial research have lauded the work as a significant advancement. Dr. Anya Sharma, a renowned mitochondrial geneticist at the National Institutes of Health, commented, "This is a truly remarkable piece of work. For years, we’ve been struggling to explain the consistent spacing of mtDNA, and it turns out a process described over a century ago holds the key. It’s a testament to the power of revisiting old observations with new technologies."
Professor Kenji Tanaka, a cell biologist at the University of Tokyo, echoed this sentiment: "The elegance of mitochondrial pearling as a biophysical mechanism for genome organization is striking. It offers a simple, yet highly effective, solution to a complex cellular problem. This discovery will undoubtedly stimulate a wave of new research in the field."
Future Directions: Expanding the Scope of Investigation
The current research has opened up numerous avenues for future investigation. Scientists are eager to explore the precise molecular players involved in triggering and executing the pearling process. Further studies will likely focus on identifying the specific proteins and lipids that mediate the constrictions and the role of the mitochondrial inner membrane in nucleoid segregation.
Moreover, researchers aim to investigate whether variations in mitochondrial pearling efficiency or regulation contribute to disease susceptibility or progression. Comparative studies across different cell types and organisms could reveal evolutionary adaptations in this fundamental process.
The integration of this newfound knowledge into broader models of mitochondrial dynamics and cellular aging is also a priority. As the field moves forward, the humble yet powerful process of mitochondrial pearling is poised to become a central focus in our quest to understand and combat diseases rooted in the dysfunction of our cellular powerhouses.













Leave a Reply