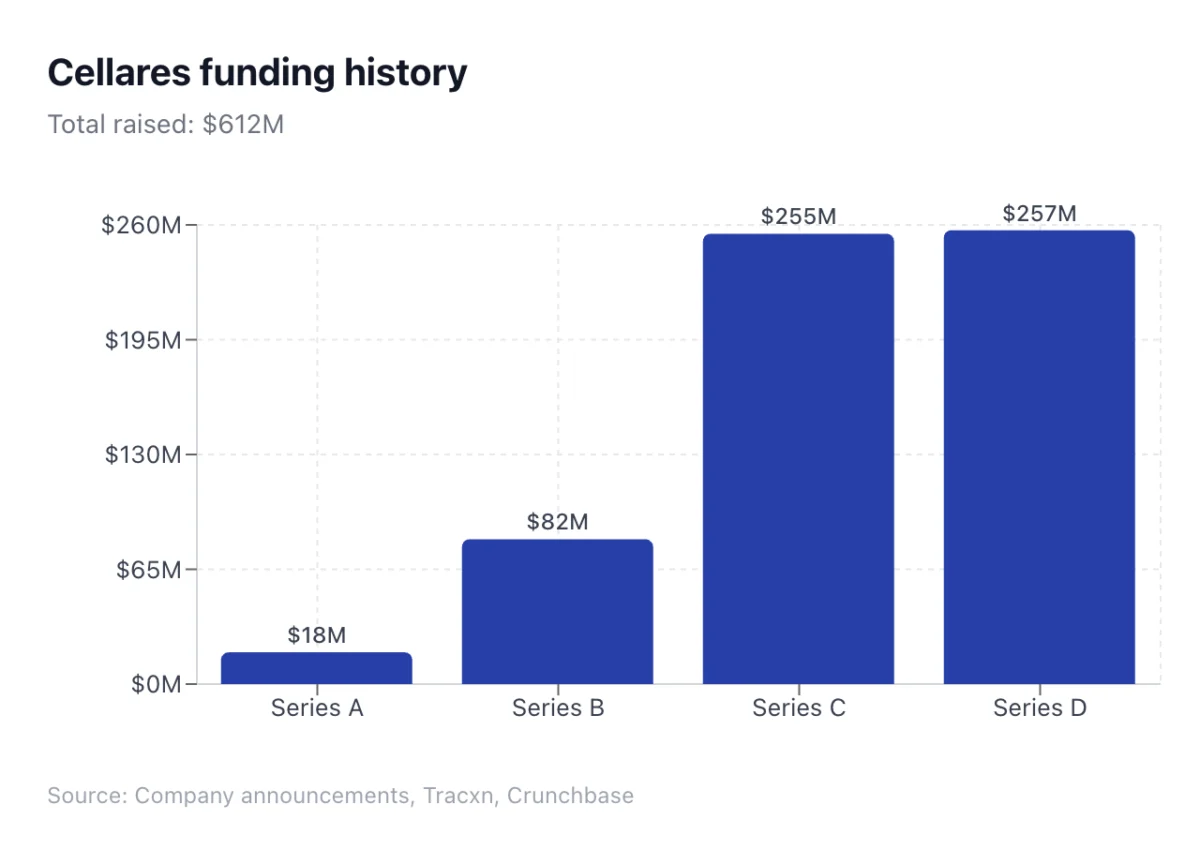
The burgeoning field of cell and gene therapy, heralded as a revolutionary frontier in medicine, is currently grappling with a formidable challenge: the escalating cost and complexity of manufacturing. This bottleneck, often referred to as the "manufacturing bill," threatens to impede patient access and stifle the full potential of these transformative treatments. In response to this critical industry need, Cellares, a South San Francisco-based biotechnology company, announced today the successful closure of a $257 million Series D funding round. This substantial capital injection is earmarked to fuel the global expansion of its automated "Smart Factories," which the company asserts will drastically reduce the capital expenditure and labor costs that have historically plagued cell therapy production.
The investment round, co-led by prominent financial institutions BlackRock and Eclipse, underscores a growing belief in the industrialization of biomanufacturing as the viable path forward for advanced therapies. New strategic investors joining this round include T. Rowe Price, Baillie Gifford, Duquesne Family Office, and Intuitive Ventures—the venture capital arm of Intuitive Surgical, a pioneer in robotic surgery known for its da Vinci system. Further bolstering the diverse investor syndicate are EDBI, the corporate investment arm of Singapore’s Economic Development Board, and Gates Frontier, the venture investment firm founded by Bill Gates. This blend of financial and strategic investors highlights the broad recognition of Cellares’ potential to disrupt a critical sector.
The Manufacturing Bottleneck in Cell and Gene Therapy
Cell and gene therapies represent a paradigm shift in medical treatment, offering curative potential for a range of diseases previously considered untreatable, including certain cancers, genetic disorders, and autoimmune conditions. These therapies, particularly autologous cell therapies like CAR-T, involve extracting a patient’s own cells, genetically modifying them in a laboratory, expanding them, and then reinfusing them into the patient. While immensely promising, the manufacturing process is inherently complex, resource-intensive, and bespoke.
Traditional cell therapy manufacturing is characterized by its manual nature, requiring highly skilled personnel to perform numerous delicate steps in pristine, controlled environments (cleanrooms). This manual intervention introduces significant variability, increases the risk of contamination, and drives up labor costs. Moreover, the stringent Good Manufacturing Practice (GMP) requirements necessitate extensive quality control and assurance protocols, adding further layers of complexity and expense. The specialized infrastructure, including dedicated cleanrooms and advanced analytical equipment, demands substantial upfront capital investment. Consequently, the cost of manufacturing a single dose of cell therapy can often run into hundreds of thousands of dollars, making these life-saving treatments prohibitively expensive for many healthcare systems and patients.
This manufacturing burden has had a tangible impact on the broader cell and gene therapy ecosystem. Venture funding in the sector, after reaching a peak in 2021, has experienced a sharp decline, plunging more than 80%. This contraction reflects investor apprehension regarding the long-term return on investment for therapies that, despite their clinical efficacy, face significant hurdles in achieving scalable, cost-effective production. The industry urgently requires solutions that can de-risk manufacturing, improve efficiency, and ultimately lower the cost of goods, thereby expanding market access and accelerating therapeutic development.
Cellares’ Solution: Industrializing Production with "Smart Factories"
Cellares is positioning itself as a pivotal solution to this industry-wide challenge through its innovative approach to industrializing cell therapy manufacturing. The company’s core offering revolves around its proprietary hardware-and-software platform, the Cell Shuttle, and its vision for globally interconnected "Smart Factories." This model aims to transform the labor-intensive, manual processes into a fully automated, scalable, and standardized manufacturing workflow.
The Cell Shuttle is designed as an automated, end-to-end production platform capable of handling multiple cell therapy modalities. It encapsulates the entire manufacturing process within a closed, modular system, from cell input to final product formulation. This closed-system design significantly reduces the risk of contamination, a critical concern in cell therapy production where sterility is paramount. The automation minimizes human touchpoints, thereby reducing labor costs, human error, and variability between batches. The platform integrates robotics, advanced process controls, and sophisticated software to ensure precise execution of each step, from cell culture and expansion to purification and final fill.
Complementing the Cell Shuttle is Cell Q, an automated testing suite. Quality control and release testing are crucial but time-consuming aspects of cell therapy manufacturing, often taking weeks and contributing significantly to the overall cost and time-to-patient. Cell Q aims to automate these analytical processes, providing rapid, reliable, and consistent quality assessments. By integrating automated testing, Cellares seeks to accelerate batch release times, further reducing costs and ensuring that therapies can reach patients more quickly.

A Landmark Series D Round and Strategic Investor Confidence
The $257 million Series D funding round represents a significant vote of confidence in Cellares’ technology and business model. The co-leadership by BlackRock, a global investment management corporation, and Eclipse, a venture capital firm known for its focus on industrial technologies, signals a strong belief in the financial viability and transformative potential of Cellares’ automation strategy.
The addition of new investors brings both capital and strategic alignment. T. Rowe Price and Baillie Gifford are well-respected asset managers with a history of investing in growth-oriented technology and life sciences companies. Duquesne Family Office, known for its astute investments, further diversifies the funding base. The participation of Intuitive Ventures is particularly noteworthy. Intuitive Surgical’s expertise in robotics and minimally invasive surgical technologies provides a strategic lens on automation and precision, offering potential synergies and validation for Cellares’ hardware-centric approach. Similarly, Gates Frontier’s involvement aligns with Bill Gates’ broader philanthropic and investment interests in global health and technological innovation that addresses critical societal needs. EDBI, representing Singapore’s Economic Development Board, signifies an international recognition of Cellares’ global ambitions and the potential for its technology to foster advanced biomanufacturing capabilities in key regions.
This diverse syndicate reflects a collective conviction that Cellares’ integrated platform holds the key to unlocking the full potential of cell therapies by making them more affordable, accessible, and reliably produced. Investors are betting on Cellares’ ability to create a standardized, industrial-scale manufacturing backbone for an industry that has long struggled with bespoke, high-cost production.
Global Expansion and Ambitious Timeline
With this substantial financing, Cellares is poised to execute an aggressive global factory buildout. The company plans to establish "Smart Factories" in key strategic locations: its headquarters in South San Francisco, California; Bridgewater, New Jersey; Leiden, Netherlands; and Kashiwa City, Japan. This multinational footprint is designed to serve major biopharmaceutical markets across North America, Europe, and Asia, ensuring proximity to clients and patients while facilitating efficient global supply chains.
The strategic choice of locations is deliberate. South San Francisco is a renowned biotech hub, offering access to talent and a vibrant innovation ecosystem. Bridgewater, NJ, positions Cellares to serve the critical East Coast biopharma corridor. Leiden, in the Netherlands, is a rapidly growing life sciences cluster in Europe, providing a gateway to the European market. Kashiwa City, Japan, will anchor Cellares’ presence in the Asia-Pacific region, a significant and expanding market for advanced therapies.
Cellares projects an ambitious timeline for these facilities. The company expects to commence clinical manufacturing in the first half of 2026, marking a critical step toward validating its automated processes in a regulatory-compliant environment. Following successful clinical manufacturing, Cellares aims to achieve commercial-scale capacity by 2027. This rapid scale-up underscores the urgency of the manufacturing bottleneck and the company’s confidence in its ability to deliver industrial-scale solutions in a compressed timeframe.
Market Validation and Key Partnerships
Cellares’ approach is not merely theoretical; it has garnered significant external validation and established crucial industry partnerships. The company has received an FDA Advanced Manufacturing Technology designation, a testament to the innovative nature of its platform and its potential to significantly enhance the efficiency, quality, and reliability of pharmaceutical manufacturing. Such designations are rare and signal regulatory confidence in the technology’s ability to address critical manufacturing challenges.
Perhaps the most significant commercial endorsement comes from a multi-year global manufacturing agreement with Bristol Myers Squibb (BMS), valued at up to $380 million. This partnership demonstrates a major pharmaceutical company’s commitment to leveraging Cellares’ automated platform for the production of its cell therapies. BMS is a leader in oncology and has a significant portfolio of cell therapies, including the CAR-T therapies Breyanzi and Abecma. This agreement provides Cellares with a substantial commercial pipeline and validates its technology’s ability to meet the rigorous demands of a top-tier biopharma partner.

Beyond BMS, Cellares boasts a growing list of notable customers, including Kite (Gilead’s cell therapy arm), City of Hope, and Cabaletta Bio. These partnerships with leading academic centers and biotech innovators further underscore the broad applicability and industry acceptance of Cellares’ automated manufacturing solution across different stages of development and types of cell therapies.
The IDMO Model: A Paradigm Shift in Biomanufacturing
Cellares operates on an Integrated Development and Manufacturing Organization (IDMO) model, which represents a departure from the traditional Contract Development and Manufacturing Organization (CDMO) framework. While CDMOs offer outsourced manufacturing services, they often rely on conventional, labor-intensive processes. Cellares’ IDMO model, leveraging its automated "Smart Factory" concept, aims to integrate advanced technology across the entire manufacturing lifecycle, from process development to commercial production.
The company’s central claim is that a single Cellares "Smart Factory" can match the output of approximately 10 conventional CDMO facilities of similar scale. This asserted order of magnitude improvement in efficiency stems from several factors:
- Reduced Footprint: Automation and closed systems require significantly less cleanroom space per unit of output compared to manual processes.
- Lower Labor Costs: The minimal human intervention drastically cuts down on the need for highly skilled, expensive manual labor.
- Faster Turnaround Times: Automated processes, coupled with automated quality control (Cell Q), can accelerate batch production and release.
- Enhanced Consistency and Quality: Robotics and process controls ensure reproducibility and reduce human error, leading to more consistent product quality.
- Higher Throughput: The continuous and integrated nature of the automated system allows for greater volume production.
While these projections are compelling, Cellares acknowledges that its model has "yet to be proven at full commercial throughput." This transparency highlights the critical phase the company is entering, where theoretical efficiency must translate into real-world, large-scale commercial production.
Broader Implications for the Cell and Gene Therapy Industry
Cellares’ success in industrializing cell therapy manufacturing could have profound implications across the biopharmaceutical landscape:
- Increased Patient Access: By significantly driving down the cost of goods for cell therapies, Cellares’ model could make these life-saving treatments more affordable and accessible to a broader patient population globally. This aligns with the ultimate goal of advanced medicine: to deliver effective therapies to those who need them most.
- Accelerated Drug Development and Commercialization: Reduced manufacturing lead times and lower costs could de-risk the development pathway for new cell therapies, encouraging more companies to invest in this field. Faster, more reliable manufacturing means therapies can move from clinical trials to commercial availability more quickly.
- Standardization and Quality Improvement: Automation inherently introduces standardization, reducing variability that can plague manual processes. This leads to more consistent product quality, which is critical for patient safety and regulatory compliance.
- Reshaping the Biomanufacturing Landscape: Cellares’ IDMO model could challenge traditional CDMOs to innovate and adopt more automation, potentially leading to a broader transformation in how advanced therapies are manufactured. It could also encourage a shift towards decentralized manufacturing, with smaller, automated units closer to patient populations.
- Economic Impact: The creation of "Smart Factories" globally could stimulate economic growth, foster high-tech job creation, and establish new centers of biomanufacturing excellence.
Leadership Outlook and Future Trajectory
Fabian Gerlinghaus, CEO and co-founder of Cellares, expressed strong confidence in the company’s trajectory following the Series D funding. He stated that the new capital, combined with existing FDA validation and robust commercial demand, firmly positions Cellares to build the essential manufacturing infrastructure the cell and gene therapy sector critically needs. Gerlinghaus further articulated a clear strategic objective: "This financing puts Cellares on a clear, disciplined path toward becoming a public company."
This statement signals Cellares’ ambition to not only revolutionize manufacturing but also to become a publicly traded entity, indicating a mature business strategy focused on long-term growth and market leadership. The journey from a venture-backed startup to a public company would require continued execution, successful commercial scale-up, and consistent delivery on its promises of efficiency and cost reduction.
The path forward for Cellares, while bolstered by significant capital and strategic partnerships, will involve navigating the complexities of scaling advanced manufacturing technology, maintaining regulatory compliance across multiple geographies, and continuing to innovate in a rapidly evolving scientific field. The company’s ability to demonstrate the promised "order of magnitude" efficiency at full commercial throughput will be the ultimate determinant of its long-term success and its impact on making cell and gene therapies a cornerstone of modern medicine. The industry watches with keen interest as Cellares moves from groundbreaking vision to global manufacturing reality.














Leave a Reply