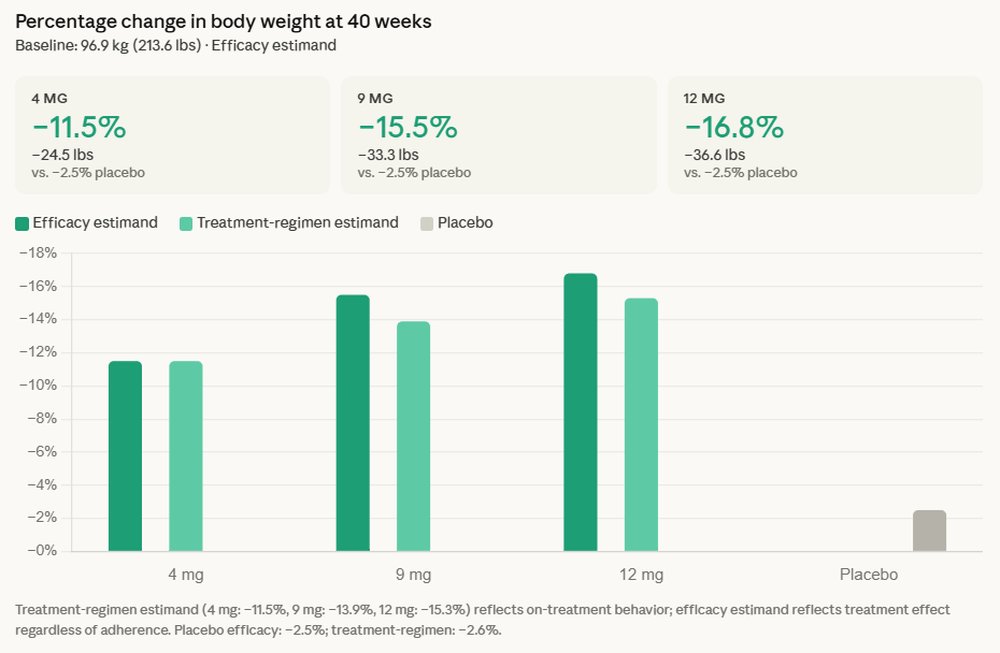
Eli Lilly and Company today announced groundbreaking results from a Phase 3 clinical trial, revealing that its investigational triple agonist, retatrutide, led to an average weight loss of 16.8% in participants with type 2 diabetes. This significant outcome, alongside superior A1C reduction, positions retatrutide as a potentially transformative therapy in the complex landscape of cardiometabolic health, addressing both glycemic control and weight management, which have historically presented persistent challenges for patients.
A New Frontier in Metabolic Disease Treatment
Retatrutide is a first-in-class investigational therapeutic that uniquely targets three key hormone receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon. This multi-pronged mechanism of action differentiates it from existing single or dual-agonist therapies, offering a novel approach to metabolic regulation. The drug successfully met its primary and key secondary endpoints in the 40-week trial, demonstrating superior A1C reduction and substantial weight loss when compared to a placebo. Participants experienced an average A1C reduction of up to 2.0% and an average weight loss of 36.6 pounds (approximately 16.6 kg). Crucially, the trial observed no weight-loss plateau, with participants continuing their weight loss trajectory throughout the 40-week study period, suggesting potential for even greater reductions over a longer duration.
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, underscored the significance of these findings. "For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes," Custer stated. "With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes." His comments reflect the long-standing unmet need in managing the intertwined conditions of type 2 diabetes and obesity, which often exacerbate each other and lead to a cascade of further health complications.
Beyond glycemic control and weight reduction, retatrutide also demonstrated clinically meaningful improvements across several key cardiovascular risk factors. These included reductions in non-HDL cholesterol, triglycerides, and systolic blood pressure, all of which are critical indicators for cardiovascular health and frequently elevated in individuals with type 2 diabetes and obesity. The safety profile observed in the trial was consistent with similar incretin-based therapies, with common adverse effects including nausea, diarrhea, and vomiting. These side effects are generally manageable and often diminish over time as patients adjust to the medication.
The Science Behind the Triple Agonism
To fully appreciate the innovation of retatrutide, it is essential to understand the roles of the three hormone receptors it targets:
- GIP (Glucose-dependent insulinotropic polypeptide): A hormone produced in the gut that stimulates insulin secretion in a glucose-dependent manner, helping to regulate blood sugar. It also plays a role in fat metabolism and energy balance.
- GLP-1 (Glucagon-like peptide-1): Another gut-derived hormone that stimulates insulin secretion, suppresses glucagon secretion (which raises blood sugar), slows gastric emptying, and promotes satiety, leading to reduced food intake and weight loss.
- Glucagon: A hormone primarily known for its role in raising blood glucose levels when they are too low. However, activating the glucagon receptor in combination with GIP and GLP-1 has been hypothesized to enhance energy expenditure, potentially contributing to greater weight loss by increasing metabolism.
Traditional GLP-1 receptor agonists (like semaglutide, found in Ozempic and Wegovy) have revolutionized the treatment of type 2 diabetes and obesity by leveraging the powerful effects of GLP-1. Eli Lilly’s own tirzepatide (Mounjaro for diabetes, Zepbound for weight loss) advanced this by targeting both GLP-1 and GIP receptors, demonstrating superior efficacy over single GLP-1 agonists. Retatrutide represents the next evolutionary step, adding glucagon agonism to the mix. The hypothesis is that by engaging all three pathways, retatrutide can achieve a more comprehensive and potent metabolic effect, leading to even greater improvements in glycemic control and weight reduction, along with potentially broader benefits on lipid profiles and cardiovascular markers. The profound weight loss and sustained trajectory observed in the trial lend strong support to this multi-receptor strategy.
The Global Burden of Cardiometabolic Disease
Type 2 diabetes and obesity are among the most pervasive and costly chronic diseases globally. According to the International Diabetes Federation, over 537 million adults worldwide are living with diabetes, with projections indicating this number will rise to 783 million by 2045. The vast majority of these cases are type 2 diabetes, often linked to overweight and obesity. Similarly, the World Health Organization reports that obesity has nearly tripled worldwide since 1975, with over 1 billion people now obese.
These conditions are not merely isolated health issues; they are interconnected and contribute to a host of severe comorbidities, including cardiovascular disease (heart attack, stroke), kidney disease, non-alcoholic fatty liver disease (now termed metabolic dysfunction-associated steatotic liver disease, MASLD), obstructive sleep apnea, certain cancers, and musculoskeletal problems like knee osteoarthritis. The economic burden is immense, encompassing direct healthcare costs, lost productivity, and reduced quality of life. The development of therapies like retatrutide, which simultaneously address multiple facets of these complex conditions, holds the promise of significantly altering disease progression and improving patient outcomes on a global scale.
Eli Lilly’s Strategic Dominance in Cardiometabolic Health
Eli Lilly has emerged as a frontrunner in the development of innovative treatments for diabetes and obesity. The company’s prior success with tirzepatide, a dual GIP/GLP-1 agonist, has already set a high bar for efficacy in both A1C reduction and weight loss. The positive results for retatrutide reinforce Lilly’s strategic commitment to this therapeutic area and its aggressive pursuit of next-generation molecules. This consistent innovation positions Lilly not only as a market leader but also as a key driver in redefining the standards of care for cardiometabolic diseases.
Analysts are closely watching Lilly’s pipeline, particularly with the competitive landscape heating up with other pharmaceutical companies also investing heavily in incretin-based therapies. The ability of retatrutide to achieve such significant weight loss, especially without a plateau at 40 weeks, suggests it could offer an even more compelling option for patients struggling with weight management in the context of type 2 diabetes. Industry experts anticipate that if these results are sustained and replicated in broader populations, retatrutide could command a substantial share of the growing market for weight loss and diabetes drugs, further solidifying Lilly’s dominant position.
Broader Implications and Future Trajectory
The potential impact of retatrutide extends far beyond glycemic control and initial weight loss. The observed improvements in cardiovascular risk factors such as non-HDL cholesterol, triglycerides, and systolic blood pressure are particularly encouraging. Cardiovascular disease remains the leading cause of morbidity and mortality in individuals with type 2 diabetes. By positively influencing these markers, retatrutide could offer comprehensive cardiometabolic protection, potentially reducing the incidence of adverse cardiovascular events.
Lilly’s extensive Phase 3 clinical trial program for retatrutide underscores the company’s confidence in its broad applicability and therapeutic potential. The drug is currently being evaluated in several ongoing Phase 3 trials across a spectrum of conditions, including:
- Obesity and overweight with at least one weight-related medical problem: This is a crucial area, as the drug’s profound weight loss capabilities could offer a new standard for a vast patient population.
- Type 2 diabetes: Further trials will solidify its role and gather more extensive data.
- Knee osteoarthritis: Obesity is a major risk factor for osteoarthritis, and significant weight loss could alleviate pain and improve joint function.
- Moderate-to-severe obstructive sleep apnea (OSA): OSA is highly prevalent in obese individuals, and weight loss is a primary treatment strategy.
- Chronic low back pain: Similar to knee osteoarthritis, weight reduction can significantly impact musculoskeletal pain.
- Cardiovascular and renal outcomes: Dedicated trials to definitively assess its long-term protective effects on the heart and kidneys are vital.
- Metabolic dysfunction-associated steatotic liver disease (MASLD): MASLD (formerly NAFLD) is strongly linked to obesity and insulin resistance, and effective weight loss could halt or reverse liver damage.
The breadth of these investigations highlights retatrutide’s potential to become a foundational therapy for a wide array of metabolic and weight-related disorders. If successful across these indications, it could offer a single therapeutic agent capable of addressing multiple, interconnected health issues simultaneously, simplifying treatment regimens and improving patient adherence.
Addressing the Unmet Needs: A Patient Perspective
For patients living with type 2 diabetes and obesity, the prospect of a drug like retatrutide offers significant hope. Current treatment strategies often involve multiple medications to manage blood sugar, weight, and associated comorbidities, leading to complex regimens and potential side effects. A single agent capable of achieving substantial improvements across these domains represents a monumental leap forward. Patient advocacy groups are likely to welcome these results, emphasizing the potential for improved quality of life, reduced medication burden, and a decreased risk of long-term complications. The ability to achieve substantial and sustained weight loss without a plateau could empower patients to reach and maintain healthier weights, a goal that has often been elusive with existing treatments.
From a clinician’s standpoint, retatrutide could provide a powerful new tool in their arsenal. The dual challenge of achieving optimal glycemic control and meaningful weight loss in type 2 diabetes patients has long been a source of frustration. The robust data presented by Lilly suggests that retatrutide could offer a more effective and comprehensive solution, allowing healthcare providers to better manage their patients’ overall metabolic health and potentially prevent the progression of associated complications.
Challenges and Regulatory Pathway
While the initial Phase 3 results are overwhelmingly positive, the path to market involves further rigorous evaluation. Lilly will need to complete its comprehensive Phase 3 program, gathering extensive safety and efficacy data across diverse patient populations and over longer durations, particularly for the cardiovascular and renal outcomes trials. These trials are crucial for establishing the long-term benefits and safety profile required for regulatory approvals globally.
The regulatory submission process, once completed, will involve extensive review by health authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Given the drug’s novel mechanism and strong efficacy, a smooth, albeit thorough, review process is anticipated. The eventual market launch, if approved, would likely face high demand, similar to Lilly’s other successful incretin therapies.
In conclusion, Eli Lilly’s retatrutide has demonstrated exceptional potential in its Phase 3 trial for type 2 diabetes, offering a unique triple-agonist approach that delivers substantial weight loss and significant A1C reduction. These findings, coupled with improvements in cardiovascular risk factors and a manageable safety profile, position retatrutide as a potential game-changer in the treatment of cardiometabolic diseases. As Lilly continues its extensive clinical development program, the medical community and patients alike will eagerly await further data and the eventual regulatory decisions that could usher in a new era of more effective and comprehensive metabolic health management.














Leave a Reply