New research emerging from Arizona State University is fundamentally reshaping our understanding of bacterial locomotion, revealing that these microscopic organisms possess a remarkable suite of strategies for movement and dissemination, even when their primary propulsion systems fail. For decades, the scientific community has largely attributed bacterial mobility to flagella, slender, whip-like appendages that rotate to propel cells through liquid environments. However, groundbreaking studies published by ASU researchers demonstrate that microbes can effectively spread across surfaces through mechanisms entirely independent of these familiar structures, opening new avenues for combating infections and understanding microbial ecosystems.
The Silent Spread: Uncovering "Swashing"
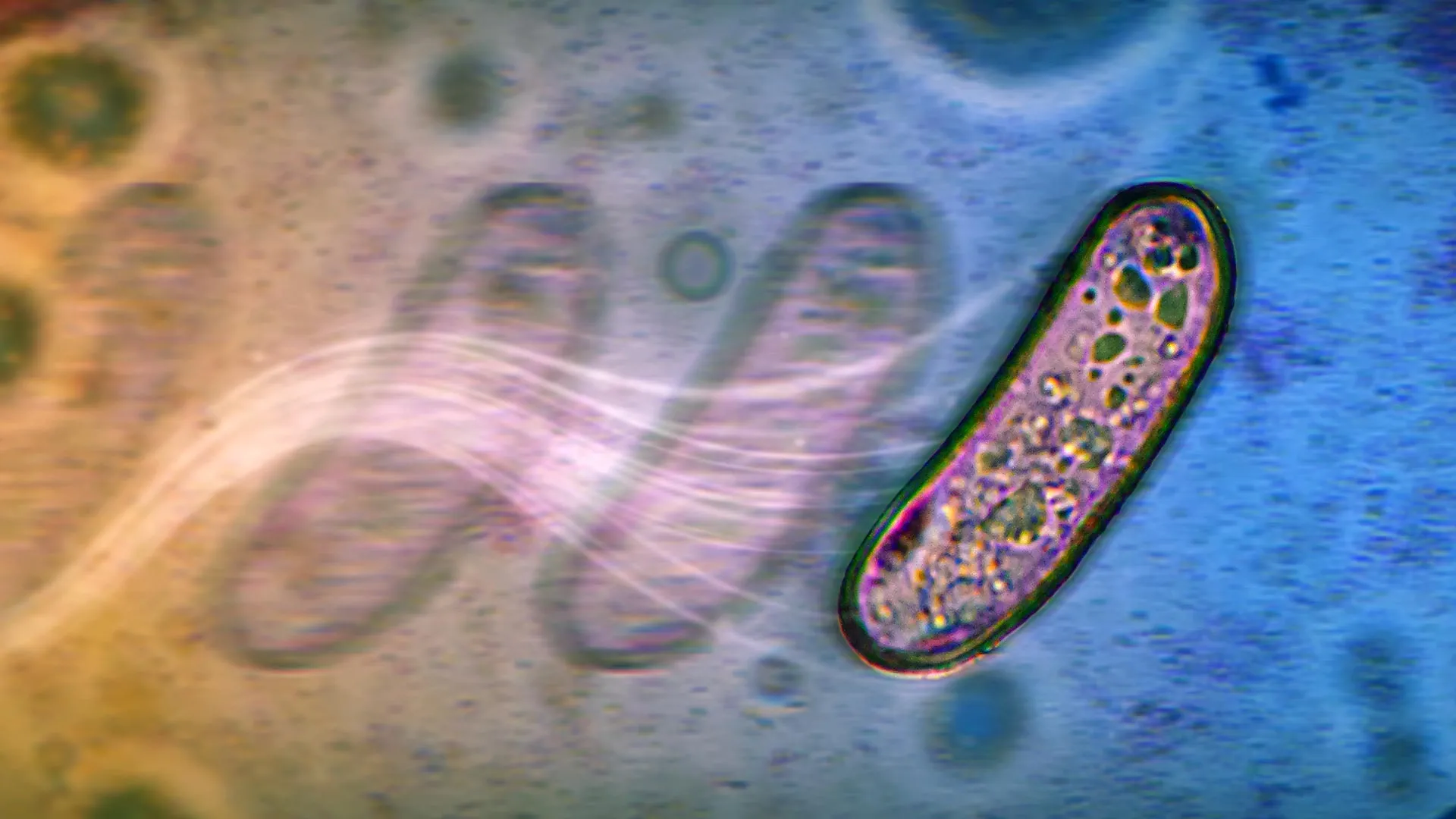
The first of these pivotal studies, led by researcher Navish Wadhwa from ASU’s Biodesign Center for Mechanisms of Evolution and the Department of Physics, has identified a novel mode of bacterial migration dubbed "swashing." This phenomenon allows common pathogens like Salmonella and Escherichia coli (E. coli) to traverse moist surfaces even when their flagella are experimentally disabled. The research, published in the prestigious Journal of Bacteriology and recognized as an Editor’s Pick, reveals that these bacteria generate their own motive force through their metabolic processes.
"We were amazed by the ability of these bacteria to migrate across surfaces without functional flagella," Wadhwa stated in a press release. "In fact, our collaborators originally designed this experiment as a ‘negative control,’ meaning that we expected (once rendered) flagella-less, the cells to not move. But the bacteria migrated with abandon, as if nothing were amiss, setting us off on a multiyear quest to understand how they were doing it."
The mechanism of swashing is elegantly simple yet profound. When bacteria ferment sugars, a common metabolic activity, they produce tiny outward-flowing currents across a wet surface. These currents, driven by the release of acidic byproducts such as acetate and formate, gently push the bacterial colony forward, much like leaves drifting downstream on a shallow stream. This discovery has significant implications, potentially explaining how disease-causing microbes can colonize medical devices, establish infections in wounds, and contaminate food processing equipment—environments where their conventional flagellar propulsion might be hindered or absent.
Sugar as Fuel for Microbial Journeys
The swashing phenomenon is intrinsically linked to the availability of fermentable sugars. Glucose, maltose, and xylose are prime examples of such substrates that, when consumed by bacteria, initiate the cascade leading to fluid current generation. The acidic byproducts of fermentation not only drive the outward flow of water but also create localized changes in pH. Researchers found that even modest alterations in acidity could significantly influence the bacteria’s movement.
Crucially, the absence of fermentable sugars halts swashing. This dependency highlights how sugar-rich environments within the human body, such as mucus linings in the gut or respiratory tract, could become fertile ground for bacterial proliferation and infection spread through this passive, metabolic route. The study’s findings suggest that understanding and manipulating sugar availability could become a novel strategy for infection control.
Further experiments explored the impact of surfactants, molecules commonly found in detergents, on bacterial movement. When added to bacterial colonies, surfactants completely inhibited swashing. Interestingly, these same surfactants had no effect on "swarming," another flagella-driven bacterial motility that enables rapid surface colonization. This stark difference underscores that swashing and swarming are distinct physical processes, offering a potential pathway for targeted intervention. While swarming could be countered by flagella-disrupting agents, swashing might be inhibited by altering surface chemistry or metabolic conditions.
The health implications are far-reaching. Medical implants, catheters, and hospital equipment are all vulnerable to bacterial colonization. If these devices become contaminated, bacteria might spread via swashing, bypassing strategies solely focused on disabling flagella. Treatments could instead target the metabolic pathways that fuel these fluid currents. Similarly, in food processing, understanding how E. coli and Salmonella spread through passive fluid flows can lead to enhanced sanitation protocols by focusing on factors like surface pH and sugar concentrations.
Navigating the Complexities of the Human Body
The environments within the human body present a perfect stage for swashing. Moist interfaces such as gut mucus, wound exudates, and the urinary tract offer the necessary conditions for bacteria to spread, even if their flagella are compromised. This means that bacteria could establish and spread infections in these sites through metabolic self-propulsion, a previously underappreciated mechanism.
Flavobacteria’s Molecular Snowmobile: The T9SS System
In parallel, a second study from ASU delves into the intricate world of flavobacteria, a group of microbes that do not rely on flagella for locomotion. Instead, these bacteria employ a sophisticated molecular machine known as the type 9 secretion system (T9SS) to glide across surfaces. This system functions like a molecular conveyor belt, moving along the cell’s outer membrane to propel the bacterium forward.
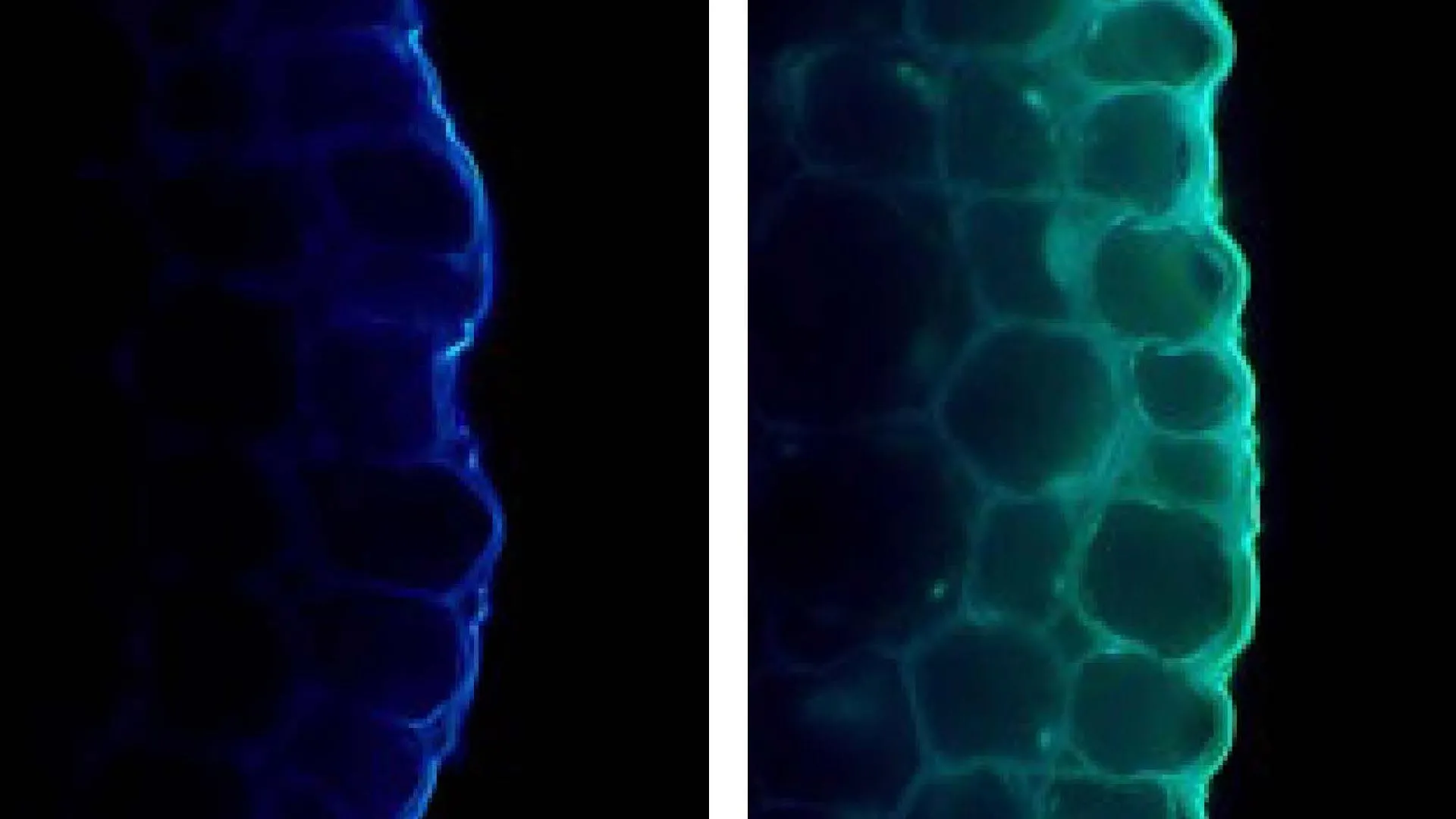
The research, detailed in the journal mBio, uncovered a critical component of the T9SS: a protein named GldJ. This protein acts as a "gear shifter," dictating the direction of the motor that drives the conveyor belt. By removing a small segment of GldJ, researchers observed a reversal in the motor’s rotation from counterclockwise to clockwise, which in turn altered the bacterium’s direction of travel.
This discovery provides a detailed molecular mechanism for how flavobacteria can adjust their movement in response to environmental cues. Such adaptability is crucial for navigating complex surfaces and likely offers an evolutionary advantage by allowing these microbes to efficiently colonize diverse niches. The T9SS system’s ability to precisely tune motility and secretion in dynamic environments is a testament to bacterial evolutionary ingenuity.
Dual-Role Nanogear System: Implications for Health and Microbiome Research
The T9SS system’s influence extends beyond mere locomotion. Its activity has a dual impact on human health, depending on the specific microbial community involved. In the oral microbiome, T9SS-containing bacteria are implicated in gum disease, with the proteins they secrete contributing to inflammation. These inflammatory processes have also been linked to systemic health issues such as heart disease and Alzheimer’s.
Conversely, in the gut microbiome, T9SS activity can be beneficial. Proteins secreted via this system can protect antibodies from degradation, thereby bolstering immune defenses and potentially enhancing the efficacy of oral vaccines.
Understanding the T9SS molecular gearbox offers exciting prospects. Researchers may be able to develop strategies to inhibit the formation of biofilms—sticky microbial communities that are notorious for causing persistent infections and contaminating medical devices. Simultaneously, these insights could be leveraged to promote beneficial microbes and design targeted microbiome therapies.
"We are very excited to have discovered an extraordinary dual-role nanogear system that integrates a feedback mechanism, revealing a controllable biological snowmobile and showing how bacteria precisely tune motility and secretion in dynamic environments," stated lead researcher Shrivastava. "Building on this breakthrough, we now aim to determine high-resolution structures of this remarkable molecular conveyor to visualize, at atomic precision, how its moving parts interlock, transmit force and respond to mechanical feedback. Unraveling this intricate design will not only deepen our understanding of microbial evolution but also inspire the development of next-generation bioengineered nanomachines and therapeutic technologies."
Shrivastava is affiliated with ASU’s Biodesign Center for Fundamental and Applied Microbiomics, the Biodesign Center for Mechanisms of Evolution, and the School of Life Sciences.
A Multifaceted Approach to Microbial Control
Taken together, these two distinct lines of research—fluid surfing and molecular gear-shifting—paint a compelling picture of bacterial adaptability. They underscore that microbes possess a diverse arsenal of motility strategies, making them formidable adversaries in the fight against infection. The traditional focus on disabling flagella, while important, represents only one piece of a much larger puzzle.
The implications for combating bacterial infections are profound. These studies strongly suggest that new, multifaceted approaches are needed. Controlling the environment in which bacteria thrive—managing sugar availability, pH levels, and surface chemistry—emerges as a critical strategy. Furthermore, directly interfering with sophisticated molecular systems like the T9SS gearbox could prove instrumental in preventing bacterial movement and the release of pathogenic proteins.
The Arizona State University research team’s discoveries not only expand the boundaries of our knowledge about microbial life but also provide tangible targets for future interventions. As scientists continue to unravel the intricate mechanisms by which bacteria move and spread, the potential for developing novel, effective treatments against a wide spectrum of bacterial diseases grows ever brighter. The next steps involve not only further elucidating these mechanisms at an atomic level but also translating these fundamental discoveries into practical applications for human health and beyond.











Leave a Reply