Eli Lilly’s investigational drug, retatrutide, a pioneering first-in-class GIP, GLP-1, and glucagon triple hormone receptor agonist, has delivered extraordinary results in a pivotal Phase 3 trial. The company recently announced that retatrutide demonstrated an average weight loss of 16.8% in participants with type 2 diabetes over a 40-week period, alongside significant improvements in glycemic control. These findings underscore the potential of this multi-targeted therapeutic to redefine the treatment landscape for type 2 diabetes and its closely associated comorbidities.
Unpacking the Pivotal Phase 3 Results
The TRANSCEND-T2D-1 trial, a crucial component of retatrutide’s extensive clinical development program, unequivocally met its primary and key secondary endpoints. Participants receiving retatrutide achieved superior A1C reduction and weight loss compared to those on placebo. On average, individuals experienced a substantial reduction in their body weight by 16.8%, translating to an average loss of 36.6 pounds. A particularly noteworthy aspect highlighted by the data is the sustained nature of this weight reduction; no weight-loss plateau was observed, with participants continuing their downward trajectory throughout the 40-week study duration. This continuous efficacy suggests a profound and durable metabolic impact.
Beyond its impressive effect on body weight, retatrutide also exhibited robust glycemic control. Participants saw an average A1C reduction of up to 2.0%. A1C, or glycated hemoglobin, is a critical measure reflecting average blood glucose levels over the preceding two to three months. A reduction of this magnitude is clinically highly significant, indicating improved long-term glucose management and a decreased risk of diabetes-related complications such as cardiovascular disease, kidney disease, and neuropathy. For many individuals with type 2 diabetes, achieving an A1C below 7% is a primary treatment goal, and retatrutide’s performance indicates a strong capacity to help patients reach and maintain this target.
Beyond Glycemic Control: Comprehensive Cardiometabolic Benefits
The impact of retatrutide extends beyond mere weight and A1C metrics, demonstrating a broader beneficial effect on several key cardiovascular risk factors. The trial data revealed clinically meaningful improvements from baseline across indicators vital for cardiometabolic health. These included reductions in non-HDL cholesterol, triglycerides, and systolic blood pressure.
Non-HDL cholesterol, a comprehensive measure of "bad" cholesterol particles, and triglycerides are both well-established markers for cardiovascular disease risk. Elevated levels contribute to atherosclerosis, the hardening and narrowing of arteries, which can lead to heart attacks and strokes. Similarly, high systolic blood pressure is a major risk factor for heart disease and stroke. The ability of retatrutide to positively influence these parameters suggests a holistic approach to managing the multifaceted health challenges faced by individuals with type 2 diabetes, many of whom also contend with metabolic syndrome and an elevated risk of cardiovascular events. This comprehensive profile positions retatrutide not just as an anti-diabetic or weight-loss drug, but as a potential agent for overall cardiometabolic risk reduction.
The Science Behind the Success: A Triple Agonist Approach
Retatrutide’s innovative mechanism of action as a triple hormone receptor agonist is central to its remarkable efficacy. It simultaneously targets receptors for three distinct incretin hormones: Glucose-dependent insulinotropic polypeptide (GIP), Glucagon-like peptide-1 (GLP-1), and Glucagon. This multi-pronged approach distinguishes it from existing therapies and offers a synergistic benefit.
- GIP (Glucose-dependent insulinotropic polypeptide): This hormone primarily enhances insulin secretion from pancreatic beta cells in a glucose-dependent manner, meaning it only stimulates insulin release when blood glucose levels are high, reducing the risk of hypoglycemia. GIP also plays a role in fat deposition and metabolism, and its agonism contributes to the overall metabolic improvements observed.
- GLP-1 (Glucagon-like peptide-1): Already a well-established target for diabetes and obesity treatments, GLP-1 agonists work by stimulating insulin release, suppressing glucagon secretion (which normally raises blood sugar), slowing gastric emptying, and promoting satiety. These actions collectively lead to improved glycemic control and significant weight loss. Drugs like semaglutide (Ozempic, Wegovy) and liraglutide (Victoza, Saxenda) have revolutionized treatment in this space.
- Glucagon: Historically known for its role in raising blood glucose, glucagon’s agonism in the context of a triple agonist might seem counterintuitive for diabetes treatment. However, recent research has unveiled a more complex and nuanced role. Glucagon receptor agonism can increase energy expenditure, promote lipolysis (fat breakdown), and potentially enhance satiety through different pathways than GLP-1. When carefully balanced with GIP and GLP-1 agonism, glucagon’s effects can contribute to greater weight loss and improved metabolic health without exacerbating hyperglycemia, especially given the glucose-dependent nature of GIP and GLP-1’s insulinotropic effects.
The simultaneous activation of these three distinct yet interconnected pathways provides a more potent and comprehensive metabolic modulation compared to single or even dual agonists. This "first-in-class" designation highlights a significant leap in pharmacological strategy, moving towards a more complete physiological restoration of metabolic balance.
Addressing the Dual Epidemic: Type 2 Diabetes and Obesity
Type 2 diabetes and obesity represent two of the most pressing global health epidemics of our time, often coexisting and exacerbating each other. Obesity is a primary driver of insulin resistance, a hallmark of type 2 diabetes, making the management of both conditions intrinsically linked. For many individuals, particularly those with type 2 diabetes, achieving meaningful and sustained weight loss has historically been an uphill battle, often complicated by the metabolic changes associated with the disease itself and the side effects of some older diabetes medications.
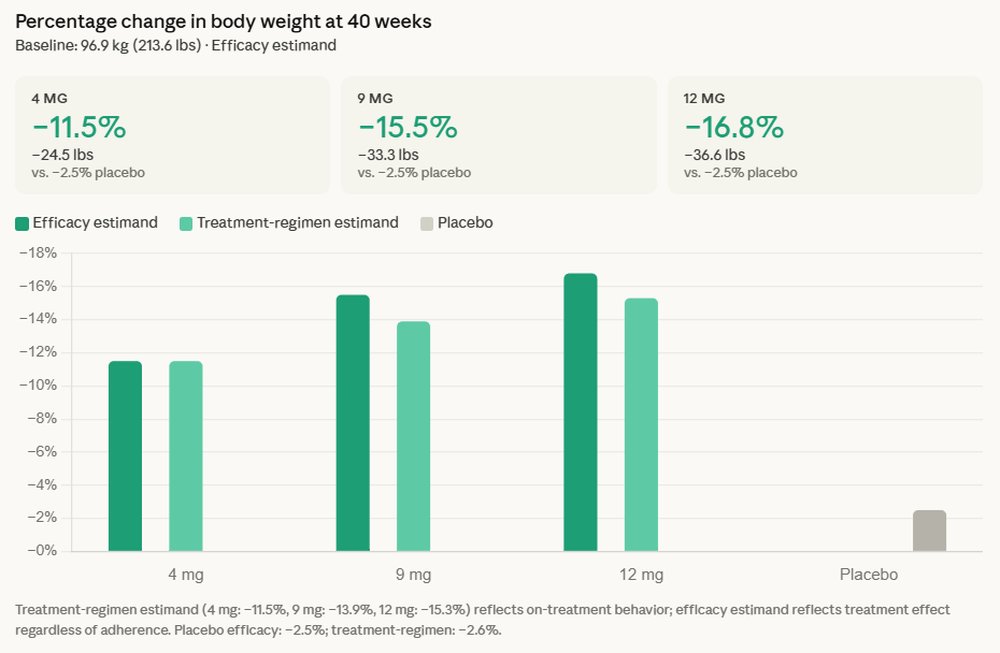
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, underscored this challenge, stating, "For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes." His remarks highlight the critical need for therapeutic innovations that can effectively tackle both aspects concurrently. Retatrutide’s ability to deliver substantial A1C reduction alongside significant weight loss directly addresses this dual burden, offering a potential solution that has long been sought by patients and clinicians. The sustained weight loss observed, without a plateau, is particularly encouraging for patients who often experience a deceleration or reversal of weight loss over time with other interventions.
Eli Lilly’s Dominance in the Cardiometabolic Space
Eli Lilly has established itself as a frontrunner in the development of groundbreaking treatments for cardiometabolic diseases. The success of tirzepatide, their dual GIP/GLP-1 agonist marketed as Mounjaro for type 2 diabetes and Zepbound for chronic weight management, has already revolutionized the field. Tirzepatide has demonstrated unprecedented weight loss results (up to 22.5% in non-diabetic individuals with obesity) and superior A1C reductions compared to earlier GLP-1 agonists.
Retatrutide represents the next evolution in Lilly’s robust pipeline, building upon the foundational success and understanding gained from tirzepatide. By adding glucagon agonism to the GIP and GLP-1 framework, Lilly is pushing the boundaries of incretin-based therapies, aiming for even greater efficacy in weight management and metabolic control. This strategic investment in multi-agonist compounds underscores Lilly’s commitment to addressing the complex and interconnected nature of metabolic disorders, positioning them as a dominant force in this rapidly expanding therapeutic area. The company’s focus on this class of drugs is a clear indication of their belief in the potential for these molecules to significantly improve patient outcomes across a spectrum of conditions.
Safety Profile and Ongoing Clinical Development
As with any medication, the safety profile of retatrutide is a crucial consideration. The reported common adverse effects, including nausea, diarrhea, and vomiting, are consistent with those observed with other incretin-based therapies, particularly GLP-1 receptor agonists and dual GIP/GLP-1 agonists. These gastrointestinal side effects are generally dose-dependent and tend to be most prominent during treatment initiation and dose escalation, often subsiding over time as the body adjusts. Management strategies, such as gradual dose titration, are typically employed to mitigate these effects. The consistency with established therapies suggests a manageable safety profile within the expected range for this drug class.
The scope of retatrutide’s potential extends far beyond type 2 diabetes. Lilly is currently evaluating the drug in an extensive array of Phase 3 clinical trials, exploring its efficacy and safety across a broad spectrum of indications. These include:
- Obesity and overweight with at least one weight-related medical problem: This is a crucial area, as the weight loss observed in diabetic patients suggests even greater potential for individuals solely focused on weight management.
- Knee osteoarthritis: Obesity is a significant risk factor for osteoarthritis, and weight loss could dramatically improve symptoms and slow disease progression.
- Moderate-to-severe obstructive sleep apnea: Weight loss is a primary treatment for OSA, which is often linked to obesity.
- Chronic low back pain: Similar to knee osteoarthritis, chronic pain conditions can be exacerbated by excess weight, and significant weight loss could offer relief.
- Cardiovascular and renal outcomes: Given the observed improvements in cardiovascular risk factors, dedicated outcome trials are essential to confirm reductions in major adverse cardiovascular events and progression of kidney disease.
- Metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as non-alcoholic fatty liver disease (NAFLD): MASLD is highly prevalent in individuals with obesity and type 2 diabetes, and weight loss is the cornerstone of its management.
This comprehensive clinical program highlights Lilly’s vision for retatrutide as a foundational therapy capable of addressing multiple comorbidities linked to obesity and metabolic dysfunction. The potential for a single drug to positively impact such a diverse range of conditions could simplify treatment regimens, improve patient adherence, and ultimately lead to better overall health outcomes.
Implications for Patients, Clinicians, and the Pharmaceutical Market
The emergence of retatrutide with such compelling data carries profound implications across the healthcare ecosystem.
- For Patients: Retatrutide offers the promise of significantly improved health outcomes. Achieving substantial weight loss and excellent glycemic control, coupled with improvements in cardiovascular risk factors, could dramatically reduce the long-term complications associated with type 2 diabetes and obesity. This translates to a better quality of life, increased mobility, reduced need for other medications, and potentially a longer, healthier lifespan. For those struggling with the emotional and physical burden of chronic weight management and diabetes, retatrutide could represent a transformative new option.
- For Clinicians: The availability of a triple agonist like retatrutide could reshape treatment algorithms for type 2 diabetes and obesity. Clinicians may have a more powerful tool at their disposal to achieve aggressive treatment targets, particularly for patients who have not responded adequately to existing therapies. The comprehensive benefits across multiple risk factors could streamline care, allowing for a more integrated approach to cardiometabolic health. It will also necessitate updated guidelines and education on the optimal use of such advanced therapies.
- For the Pharmaceutical Market: Retatrutide is poised to intensify competition in the already burgeoning market for incretin-based therapies. Lilly, already a leader with Mounjaro/Zepbound, stands to further solidify its market position with a potentially even more efficacious product. This could drive innovation across the industry as competitors strive to develop similar or superior multi-agonist compounds. The economic implications are also significant; while these innovative drugs often come with a high price tag, their potential to prevent costly long-term complications of diabetes and obesity could offer substantial societal savings in the long run, prompting discussions around access, affordability, and insurance coverage.
The Road Ahead: Regulatory Review and Future Prospects
The next critical steps for retatrutide involve the comprehensive compilation of all Phase 3 trial data, followed by submission to major regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Given the strength of the data presented thus far and the urgent unmet medical need in type 2 diabetes and obesity, a swift review process is anticipated, although the exact timeline for approval will depend on the agencies’ assessments.
Should retatrutide gain regulatory approval, it would usher in a new era of metabolic medicine, building on the foundation laid by GLP-1 and dual GIP/GLP-1 agonists. Its potential to address multiple facets of cardiometabolic disease with a single agent could revolutionize patient care, offering a powerful new weapon in the ongoing fight against two of the world’s most pervasive health challenges. The ongoing broad Phase 3 program also hints at a future where retatrutide could become a cornerstone therapy for a wide range of conditions impacted by obesity and metabolic dysfunction, cementing Eli Lilly’s leadership in this vital therapeutic space for years to come.












Leave a Reply