QUAD is marketed by MEDVi as a sublingual, four-drug cocktail for erectile dysfunction, combining three well-known PDE5 inhibitors—sildenafil, tadalafil, and vardenafil—with apomorphine, a drug primarily approved in the U.S. for advanced Parkinson’s disease. This combination, sold as a compounded drug, bypasses the rigorous premarket review process mandated by the U.S. Food and Drug Administration (FDA) for safety, effectiveness, and quality, raising profound questions about patient safety and regulatory compliance.
Unpacking QUAD: A Risky Cocktail
The Nature of Compounded Drugs
Compounded drugs are custom-made medications prepared by a pharmacist for an individual patient based on a prescription from a licensed practitioner. Unlike commercially manufactured drugs, they are not FDA-approved, meaning they have not undergone the extensive testing and review process required to ensure their safety, efficacy, and consistent quality. While legitimate compounding serves a vital role for patients with specific needs (e.g., allergies to inactive ingredients in commercial products, need for different dosage forms), the proliferation of companies mass-producing "essentially-a-copy" compounded drugs raises significant regulatory red flags. The FDA explicitly states that it does not review compounded drugs for premarket approval.
The PDE5 Inhibitors: A Triple Threat?
Sildenafil (Viagra), tadalafil (Cialis), and vardenafil (Levitra) are all FDA-approved PDE5 inhibitors, widely used and effective treatments for erectile dysfunction. They work by increasing blood flow to the penis, facilitating an erection. While individually approved and generally well-tolerated, they carry known side effects, including headache, flushing, dyspepsia, and visual disturbances. Crucially, they are contraindicated with nitrates due to the risk of dangerous hypotension (severely low blood pressure) and require caution with alpha-blockers and certain blood pressure medications.

The FDA’s official labeling for sildenafil explicitly warns against combining PDE5 inhibitors, stating that such combinations "have not been studied" and "may further lower blood pressure," concluding that "the use of such combinations is not recommended." This warning underscores the potential for additive hypotensive effects when multiple drugs from this class are taken simultaneously. QUAD’s formulation, which includes all three major PDE5 inhibitors, directly contravenes this established medical guidance. The rationale for combining three drugs that share the same mechanism of action, especially without any clinical data on the safety and efficacy of such a combination, is medically unsound and poses an elevated risk of adverse events.
Apomorphine: A Troubled History in ED Treatment
The inclusion of apomorphine further complicates QUAD’s safety profile. In the U.S., apomorphine is FDA-approved solely for the treatment of motor fluctuations, or "OFF episodes," in adults with advanced Parkinson’s disease, where it acts as a dopamine agonist. It is also used in veterinary medicine to induce vomiting in dogs.
Its history as an ED treatment is fraught with issues. In Europe, apomorphine was marketed for erectile dysfunction starting in May 2001 under the brand name Uprima, but its marketing authorization expired in May 2006. The developer, Abbott Laboratories, chose not to renew it for commercial reasons, largely influenced by underwhelming efficacy and common side effects. A UK study involving over 11,000 patients found that a staggering two-thirds discontinued its use because it simply "didn’t work," while nausea was a fairly common complaint. While a 2020 systematic review in Archives of Sexual Behavior offered a more positive outlook, noting tolerability at lower doses (2 mg and 3 mg), it conceded that discontinuation rates due to adverse events climbed significantly at higher doses.
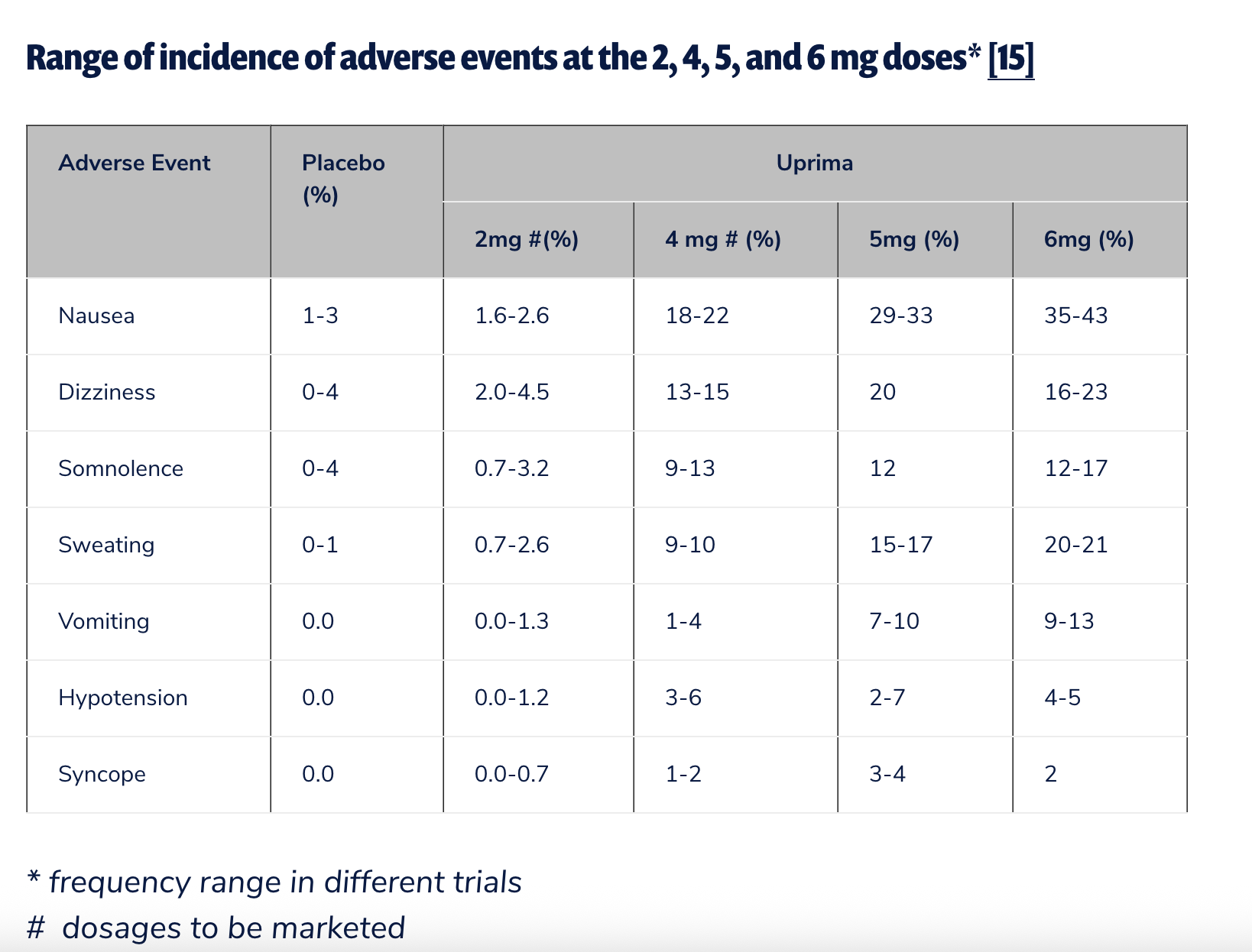
In the U.S., the journey for apomorphine as an ED treatment was even shorter. TAP Pharmaceuticals, a joint venture between Abbott and Takeda, sought FDA approval for sublingual apomorphine (Uprima) but withdrew its application in June 2000. This withdrawal followed an advisory committee flagging serious risks, including hypotension and syncope (fainting). Public Citizen, a consumer advocacy group, vehemently opposed its approval, with Sidney Wolfe, M.D., writing a blistering letter to then-CDER director Janet Woodcock, M.D., urging outright rejection. Dr. Wolfe presciently warned that approval would lead to "serious injuries" and a "near-certain post-market ban." FDA approval for ED in the U.S. never materialized. The potential for hypotension and syncope, as documented by Public Citizen from TAP Pharmaceuticals’ own clinical trial data, showed that between 4% and 5% of patients in the highest dose group experienced hypotension, while 35% to 43% reported nausea.
The Perilous Combination: Interactions and Contraindications
The most alarming aspect of QUAD is the synergistic interaction between its components. Combining three PDE5 inhibitors, each capable of lowering blood pressure, with apomorphine, a drug known to cause hypotension and syncope, creates a significantly heightened risk profile. Standard professional drug-interaction databases, widely used by U.S. pharmacies and electronic health records, classify the combination of tadalafil and apomorphine as a "Major" interaction—the highest tier of warning. These databases explicitly cite "additive blood-pressure lowering with risk of dizziness, fainting and changes in heart rate." Furthermore, hypotension itself is flagged as a "Major" disease contraindication for tadalafil, specifically for patients with resting blood pressure below 90/50 mmHg. These databases also trigger a therapeutic-duplication warning when more than one erectile dysfunction agent is prescribed simultaneously, stating that the recommended maximum is one. QUAD, by its very name, contains four active pharmaceutical ingredients, directly contradicting this established safety guideline.
Medvi’s Troubled Trajectory and Regulatory Scrutiny
From Unicorn to Controversy: The NYT Profile and Its Aftermath
MEDVi’s recent trajectory has been characterized by rapid growth and an equally swift backlash. The New York Times profile on April 2, 2026, painted a picture of CEO Matthew Gallagher as a visionary, but this narrative quickly unraveled. Investigative reporting from Futurism, Forrester, Drug Discovery & Development, and Business Insider revealed a company grappling with serious allegations that extend far beyond its ED product line. These include a February 20, 2026, FDA warning letter for misbranding compounded GLP-1 drugs, a number of pending lawsuits, the use of apparently AI-generated "doctor" personas in Meta advertisements, and questions regarding the oversight of its affiliate marketing network. These issues collectively paint a picture of a company potentially prioritizing aggressive growth and marketing over stringent regulatory compliance and patient safety.
FDA’s Stance on Compounding: "Essentially a Copy" and Beyond
The FDA maintains clear policies regarding compounding, particularly concerning products that are "essentially a copy" of commercially available drugs. An April 1, 2026, FDA statement, issued just days before the New York Times profile and this article’s testing, clarified that the agency "intends to consider a compounded drug product to be essentially a copy of a commercially available drug product" when it "contains the same APIs as two or more commercially available drug products in the same, similar or easily substitutable strength." This is highly relevant to QUAD, as sildenafil, tadalafil, and vardenafil are all commercially available as FDA-approved brand-name drugs (Viagra, Cialis, Levitra) and their generic versions.
The FDA guidance provides a narrow exception for cases where a prescriber documents a significant clinical difference for an identified individual patient (e.g., an allergy to an inactive ingredient). It also specifies that the FDA generally does not intend to take action against compounders making "essentially-a-copy" products if they fill "four or fewer prescriptions of that compounded drug product during a calendar month." This threshold is designed to accommodate genuine individualized compounding by local pharmacies for specific patient needs, not large-scale commercial distribution. Given MEDVi’s apparent operational scale and aggressive marketing, it is highly probable that its sales of QUAD far exceed this minimal threshold, further placing it squarely within the FDA’s regulatory crosshairs.
Moreover, the April 1 FDA document does not specifically address the compounding of drugs for uses outside their FDA-approved indications, which is the case for apomorphine in ED. This gray area, combined with the "essentially-a-copy" issue, creates a complex regulatory challenge for products like QUAD.
The GLP-1 Precedent: A Pattern of Misbranding?
The February 2026 FDA warning letter issued to MEDVi for misbranding compounded GLP-1 drugs (like semaglutide, used for weight loss and diabetes) is a critical piece of context. This letter indicates a prior instance of MEDVi failing to adhere to FDA regulations regarding compounded medications. While an affiliate marketing page for QUAD attempted to distance the ED product line from this specific warning, stating that the "FDA warning letter and the GLP-1 regulatory crackdown are about a different product line" and "did not reference MEDVi’s QUAD ED product line," the precedent suggests a pattern of regulatory challenges for the company across different product categories. This raises questions about MEDVi’s overall commitment to regulatory compliance and patient safety across its portfolio. The Day v. OpenLoop Health federal class action lawsuit in Delaware, which mentions MEDVi in connection with GLP-1 services, further underscores the legal and ethical scrutiny the company faces.
Marketing and Onboarding: Red Flags for Patient Safety
The "Eligibility" Illusion: Flawed Screening Processes
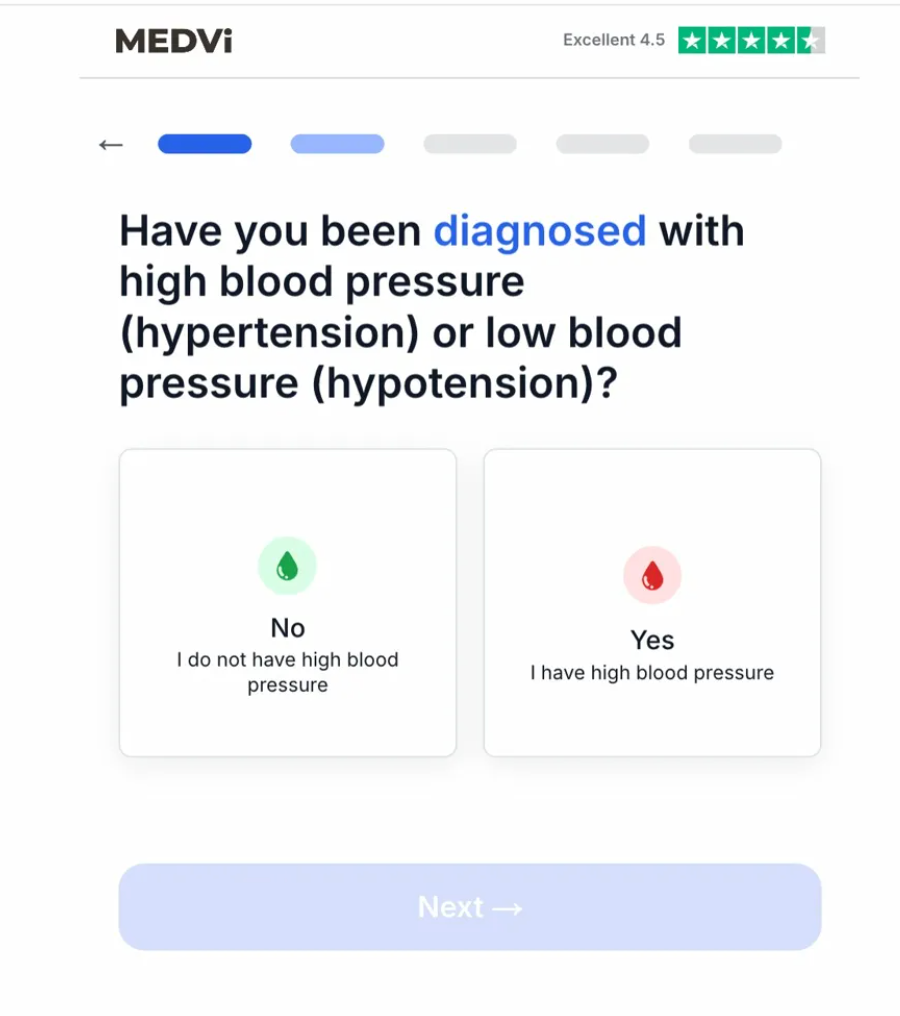
MEDVi’s patient onboarding process for QUAD, as documented in test runs conducted by Drug Discovery & Development on April 7, 2026, revealed significant flaws that could endanger patients. The intake questionnaire, which is supposed to determine eligibility for treatment, exhibited critical shortcomings. For example, a question asking about a diagnosis of "high blood pressure (hypertension) or low blood pressure (hypotension)" offered only two answer cards, both pertaining to high blood pressure. Crucially, no answer card was provided for hypotension, a condition explicitly mentioned in the question and a significant contraindication for the drugs in QUAD. When "Yes, I have high blood pressure" was selected, the intake halted, stating, "Based on your last answer, we cannot complete your assessment." This "gating logic" was designed to fire on hypertension but failed to address the equally, if not more, dangerous condition of hypotension in the context of these medications. This omission suggests a superficial screening process that could allow individuals with critical health risks to proceed toward a potentially harmful prescription.
The 94% "Success Probability": Unsubstantiated Claims
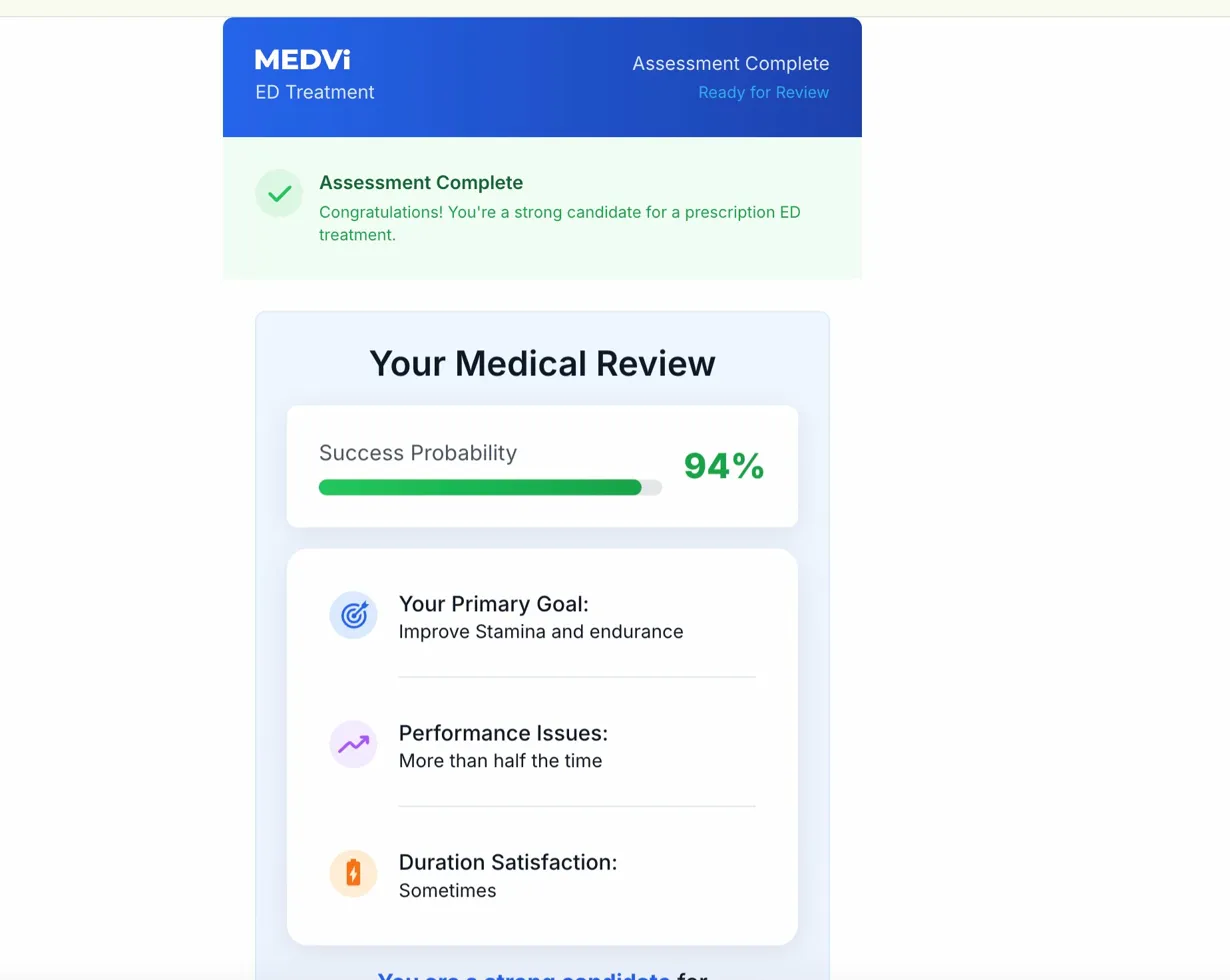
Another concerning aspect of MEDVi’s onboarding process is the claim of a "94% success probability" displayed on the "Your Medical Review" screen for QUAD. This exact same figure has been observed in MEDVi’s GLP-1 weight-loss intake process, as preserved in a screen recording filed as an exhibit in the Day v. OpenLoop Health lawsuit. Across multiple test runs for QUAD with varied answer combinations, this 94% figure remained constant, suggesting it is a fixed, generic claim rather than a personalized, evidence-based assessment. Such a high, undifferentiated success rate for a compounded, unapproved, multi-drug cocktail, especially one with known safety risks, lacks scientific credibility and could mislead patients into believing a level of efficacy and safety that is not supported by data. It can create a false sense of security, potentially influencing patients to overlook crucial warnings.
Affiliate Marketing and Misleading Information
MEDVi’s aggressive marketing strategy, heavily reliant on affiliate pages, further compounds the problem. These pages often present bold claims, such as QUAD "ignites desires" and "[hits] your system in 10 minutes" while offering a "36 hour response window," directly comparing QUAD to FDA-approved Viagra, Cialis, and Levitra in head-to-head bar charts. While some affiliate pages include disclaimers acknowledging that the therapy is "not FDA-approved as a single product" and "may not be suitable for individuals with certain medical conditions or those taking specific medications," these warnings are often buried amidst promotional content. The emphasis on speed and potency, combined with a seemingly high "success probability," can overshadow the critical risks associated with an unapproved, multi-drug combination.
One particular affiliate page, posted on MedicalFoundationofNC.org, exemplifies the complex and sometimes contradictory nature of these marketing efforts. While attempting to clarify that the existing FDA warning letter pertains to GLP-1 products and not QUAD, it still promotes MEDVi’s ED therapies. This page, like others, discloses that it "may earn affiliate commissions," is "not medical advice," and is "not a medical practice, pharmacy, or licensed healthcare provider," and explicitly states it is "not affiliated with The Medical Foundation of North Carolina, Inc., UNC Health Foundation, UNC Health Care, the UNC School of Medicine, or the University of North Carolina at Chapel Hill." Such extensive disclaimers highlight the intricate web of third-party promotion and the potential for confusion among consumers seeking legitimate medical information.
Public Health Implications and Expert Warnings
The public health implications of a product like QUAD are substantial. The stacking of potent medications with overlapping mechanisms of action and adverse effect profiles, particularly in a sublingual formulation that bypasses first-pass metabolism, carries an inherently higher risk of unpredictable and severe side effects.
The Dangers of Hypotension and Cardiovascular Events
The primary concern revolves around the synergistic hypotensive effects of the combined PDE5 inhibitors and apomorphine. Dangerous drops in blood pressure can lead to dizziness, fainting (syncope), falls, and in severe cases, could precipitate cardiovascular events such as heart attacks or strokes, especially in individuals with underlying heart conditions or those taking other medications that affect blood pressure. The lack of robust screening for hypotension in MEDVi’s intake process directly exposes vulnerable patients to these risks. The fact that the FDA explicitly warns against combining PDE5 inhibitors, and drug interaction databases flag the apomorphine-PDE5 inhibitor combination as "Major," should be a clear deterrent for any responsible medical provider.

Lack of Clinical Evidence for Combined Therapies
There is a fundamental absence of clinical trial data supporting the safety and efficacy of QUAD as a single, combined product. Compounded drugs, by definition, do not undergo such trials. For a complex, multi-drug formulation like QUAD, this lack of evidence is particularly concerning. Without controlled studies, the precise pharmacokinetics, pharmacodynamics, and interaction profiles of these four drugs taken simultaneously in a sublingual form remain unknown. This places patients in the unenviable position of being unwitting participants in an unmonitored experiment.
Patient Empowerment and Informed Consent
The aggressive marketing, coupled with simplified and potentially flawed screening, undermines the principle of informed consent. Patients might not fully grasp the unapproved status of QUAD, the significant risks associated with its unique combination of drugs, or the dubious nature of the "success probability" claims. There is a critical need for transparent, comprehensive, and unbiased information to empower patients to make truly informed decisions about their health.
Broader Industry Impact and Future Outlook
The Telehealth and Compounding Nexus
The MEDVi case highlights the ongoing challenges at the intersection of rapidly expanding telehealth services and compounding pharmacies. Telehealth has democratized access to healthcare, but it also presents new avenues for aggressive marketing and potentially lax oversight when combined with the less-regulated domain of compounding. The speed and convenience offered by these platforms must be balanced against stringent adherence to medical ethics, regulatory standards, and patient safety protocols.
Regulatory Challenges and Enforcement
The FDA and other regulatory bodies face an uphill battle in keeping pace with innovative, yet sometimes questionable, business models in the pharmaceutical space. The MEDVi case underscores the need for clearer guidelines, more proactive enforcement, and potentially updated legislation to address the mass production and marketing of compounded drugs, particularly when they mimic or combine FDA-approved medications in unstudied and potentially dangerous ways. The FDA’s existing guidance and its actions against Medvi for GLP-1 misbranding indicate a growing awareness and intent to regulate this sector more closely, but the scale of the challenge is immense.
Consumer Trust and Industry Responsibility
Ultimately, incidents like the MEDVi QUAD controversy erode public trust in both telehealth and compounding pharmacies. For the telehealth industry to mature responsibly, companies must prioritize patient safety and regulatory compliance over aggressive commercial expansion. This includes robust, medically sound patient screening, transparent communication about drug approval status and risks, and ethical marketing practices. The responsibility extends to affiliate marketers and online platforms, which must also ensure the accuracy and safety of the health information they disseminate. As investigations continue and legal actions unfold, the MEDVi case serves as a stark reminder of the critical importance of regulatory vigilance and unwavering commitment to patient well-being in the evolving landscape of digital healthcare.















Leave a Reply