New research emerging from Arizona State University is fundamentally reshaping our understanding of bacterial locomotion, revealing that these single-celled organisms possess a remarkable repertoire of movement strategies that extend far beyond their well-known flagellar propulsion. These groundbreaking discoveries, detailed in two separate studies published in leading scientific journals, indicate that bacteria can effectively spread and colonize diverse environments even when their primary means of movement are disabled, a revelation with profound implications for human health, disease prevention, and biotechnology.
The "Swashing" Phenomenon: Metabolism-Driven Migration
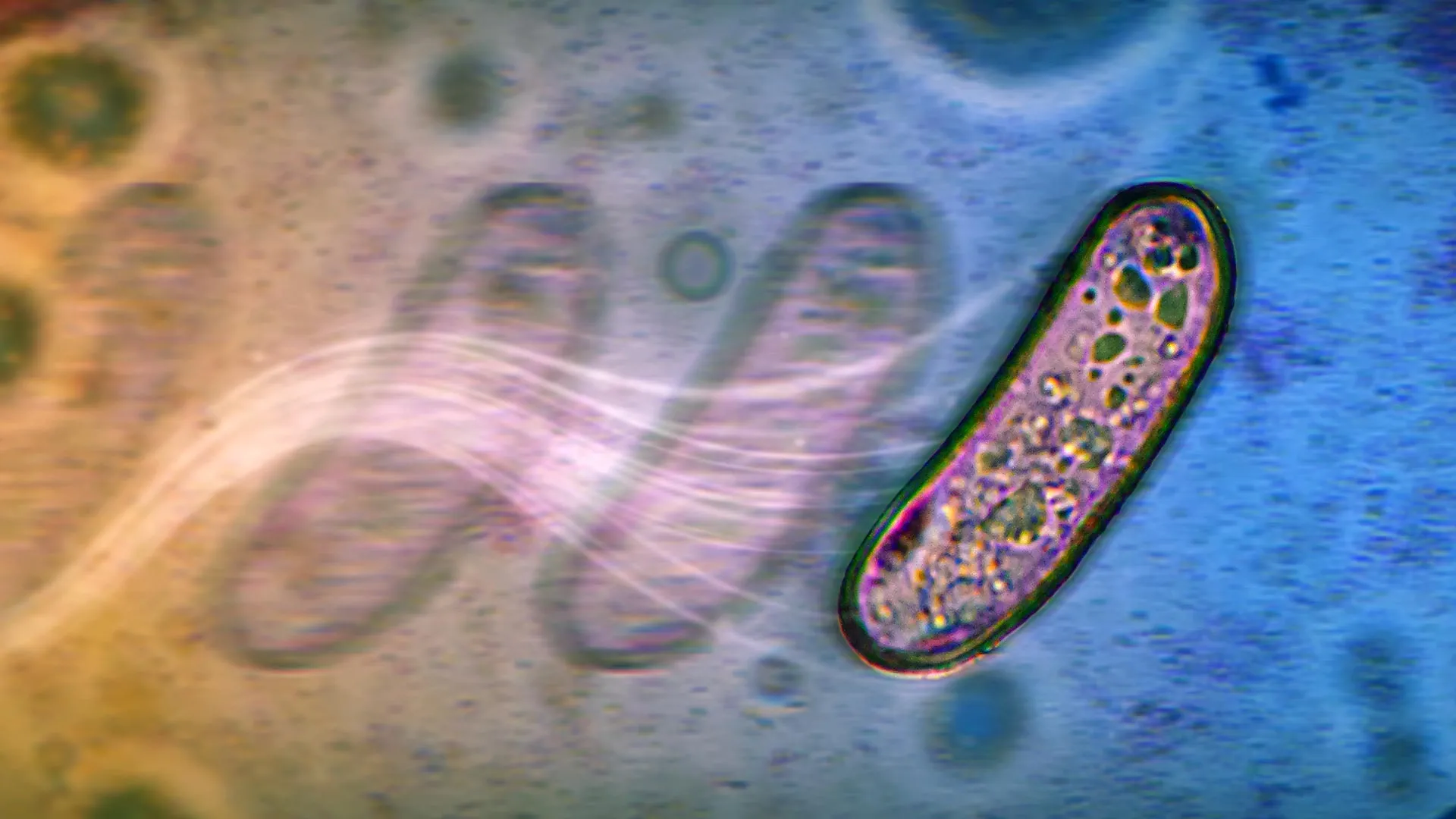
The first study, led by Navish Wadhwa, a researcher at ASU’s Biodesign Center for Mechanisms of Evolution and an assistant professor in the Department of Physics, unveiled a novel mechanism dubbed "swashing." This process allows common bacteria like Salmonella and Escherichia coli (E. coli) to migrate across moist surfaces, not through mechanical propulsion, but via metabolic activity. The research, published in the Journal of Bacteriology and highlighted as an Editor’s Pick, challenges long-held assumptions about bacterial mobility.
Traditionally, bacteria are understood to move using flagella, whip-like appendages that rotate to propel cells forward. This active swimming is crucial for bacteria to seek out nutrients, evade harmful conditions, and form complex communities. However, Wadhwa’s team observed that flagella-less strains of Salmonella and E. coli were surprisingly adept at spreading across wet surfaces. "We were amazed by the ability of these bacteria to migrate across surfaces without functional flagella," Wadhwa stated. "In fact, our collaborators originally designed this experiment as a ‘negative control,’ meaning that we expected, once rendered flagella-less, the cells to not move. But the bacteria migrated with abandon, as if nothing were amiss, setting us off on a multiyear quest to understand how they were doing it."
The team discovered that the key to this unexpected movement lies in bacterial metabolism. When these microbes ferment sugars, they produce acidic byproducts such as acetate and formate. These compounds, in turn, influence the surrounding water layer on the surface. The acidic byproducts create a localized osmotic gradient, drawing water towards the bacterial colony. This influx of water generates tiny, outward-flowing currents across the moist surface. These currents then passively carry the bacterial cells along, akin to leaves drifting on a gentle stream. This "swashing" effect enables the colony to expand its reach, even without the active swimming capabilities of flagella.
The significance of this finding is amplified by the ubiquitous presence of fermentable sugars in various biological and environmental niches. Sugars like glucose, maltose, and xylose are common energy sources for many bacteria. Environments within the human body, such as mucus lining the gut, respiratory tract, or urinary tract, are rich in these sugars. This suggests that bacteria can establish and spread infections through swashing, even if their flagella are compromised by antibiotics or the host’s immune system. Similarly, the food processing industry, where sugar-rich materials are often present, could be vulnerable to bacterial spread via this mechanism.
The researchers further demonstrated that this metabolic motility is distinct from flagellar-driven "swarming," another form of surface motility where bacteria move collectively in coordinated patterns. When surfactants, similar to detergent molecules, were introduced, they completely halted swashing. Crucially, these same surfactants did not impede swarming, providing strong evidence that swashing and swarming are governed by entirely separate physical principles. This distinction opens avenues for developing targeted interventions: treatments designed to inhibit swashing might not affect swarming, and vice versa, allowing for more precise control strategies.
The implications for healthcare are particularly stark. Many medical devices, including catheters, implants, and even hospital surfaces, can become contaminated by bacterial biofilms. These biofilms, slimy communities of bacteria, are notoriously difficult to eradicate. If bacteria can colonize these surfaces through swashing, then simply targeting flagella might be insufficient to prevent infection. Understanding and disrupting the metabolic pathways that drive swashing could become a critical component of infection control protocols. Moreover, in food safety, recognizing that E. coli and Salmonella can spread via these passive fluid flows could lead to improved sanitation practices in food processing facilities by focusing on controlling sugar availability and surface pH.
The "Molecular Gearbox": Precision Control in Flavobacteria
In parallel, a second ASU study delved into the intricate world of flavobacteria, a group of bacteria that, unlike E. coli, do not possess flagella for swimming. Instead, these microbes navigate surfaces using a sophisticated molecular machine known as the type 9 secretion system (T9SS). This system functions as a cellular engine, powering a dynamic conveyor belt that moves along the bacterial cell’s exterior, enabling them to glide across surfaces. The findings of this research, published in the journal mBio, were also recognized for their significant contribution, with the lead researcher, Chiranjib Shrivastava, expressing enthusiasm for the discovery of a "dual-role nanogear system."
The T9SS operates by propelling an adhesive-coated belt around the cell, effectively pulling the bacterium forward in a motion that researchers liken to a microscopic snowmobile. The ASU team’s breakthrough was the identification of a key protein within this system, GldJ, which acts as a molecular "gear shifter." This protein controls the direction of the motor’s rotation. By making a minor modification, specifically removing a small segment of GldJ, the researchers were able to reverse the motor’s rotation from counterclockwise to clockwise. This reversal directly alters the bacterium’s direction of travel, demonstrating a remarkable level of directional control.
This discovery offers a detailed glimpse into a molecular gearbox that allows flavobacteria to precisely adjust their movement in response to complex and dynamic environmental cues. Such adaptability is crucial for their survival and colonization, providing them with an evolutionary advantage in navigating diverse terrains. The research illuminates how bacteria can fine-tune their motility and secretion processes, adapting to the subtle shifts in their surroundings.
The implications of the T9SS system extend beyond mere motility and have a complex relationship with human health. In the oral microbiome, flavobacteria employing the T9SS have been implicated in gum disease. The proteins they secrete can trigger inflammation, and emerging research suggests potential links to systemic conditions such as heart disease and Alzheimer’s.
Conversely, in the gut microbiome, T9SS activity can be beneficial. Proteins released through this system can protect antibodies from degradation, thereby bolstering the immune system and potentially enhancing the efficacy of oral vaccines. This duality underscores the importance of understanding these systems for both therapeutic and prophylactic purposes.
Shrivastava articulated the future directions of this research: "Building on this breakthrough, we now aim to determine high-resolution structures of this remarkable molecular conveyor to visualize, at atomic precision, how its moving parts interlock, transmit force and respond to mechanical feedback. Unraveling this intricate design will not only deepen our understanding of microbial evolution but also inspire the development of next-generation bioengineered nanomachines and therapeutic technologies."
Broader Implications: A Multifaceted Approach to Bacterial Control
Taken together, these two distinct discoveries from Arizona State University paint a compelling picture of bacterial ingenuity. The "swashing" mechanism reveals an elegant, metabolism-driven strategy for surface migration, while the "molecular gearbox" of the T9SS demonstrates sophisticated, environmentally responsive locomotion. Both highlight that bacteria possess multiple, often overlooked, pathways for movement and dissemination.
This understanding is critical because the more ways bacteria can move and spread, the more challenging they become to control. Traditional approaches to combating bacterial infections have often focused on disabling flagella, a strategy that may prove less effective against bacteria employing swashing. The ASU research strongly suggests that a more comprehensive approach is needed, one that considers the diverse mechanisms bacteria employ.
The findings underscore the importance of environmental control in managing bacterial populations. Factors such as sugar availability, pH levels, and surface chemistry, which influence swashing, could become key targets for intervention. For example, altering the sugar content or acidity of surfaces in food processing plants or medical settings could significantly impede bacterial spread.
Furthermore, the identification of the T9SS’s molecular gearbox opens up new avenues for developing highly targeted therapies. Interfering with these intricate molecular machines could not only prevent bacterial movement but also inhibit the secretion of harmful proteins associated with disease. This could lead to novel strategies for preventing biofilm formation, a persistent challenge in healthcare.
The broader impact of this research extends to the burgeoning field of microbiome research. By understanding how beneficial bacteria navigate and interact within complex environments like the gut, scientists may be able to harness these mechanisms to design more effective microbiome-based therapies. Conversely, a deeper knowledge of how pathogenic bacteria utilize these systems will enable the development of more precise interventions to mitigate their detrimental effects.
Ultimately, the work from Arizona State University serves as a powerful reminder of the complex and often surprising adaptability of microbial life. As we continue to unravel these intricate biological mechanisms, the potential for innovative solutions in medicine, public health, and biotechnology grows exponentially. The era of viewing bacterial movement solely through the lens of flagella is drawing to a close, replaced by a more nuanced appreciation for the multifaceted strategies that allow these microscopic organisms to thrive and spread.















Leave a Reply