A groundbreaking study conducted by researchers at the German Center for Neurodegenerative Diseases (DZNE) and Ludwig-Maximilians-Universität München (LMU) has revealed that the brain’s own immune system plays a decisive role in the loss of smell often associated with the early stages of Alzheimer’s disease. The research, published in the journal Nature Communications, identifies a specific immunological mechanism where microglia—the brain’s resident immune cells—mistakenly attack and destroy nerve fibers essential for processing odors. By combining data from animal models, human post-mortem brain tissue, and advanced Positron Emission Tomography (PET) imaging, the team has provided a physiological explanation for a symptom that has long been recognized but poorly understood. This discovery holds significant implications for the early diagnosis of neurodegenerative conditions and the optimization of emerging amyloid-beta antibody treatments.
The Role of Microglia and the Olfactory Circuit
The sensory decline associated with Alzheimer’s disease typically begins years, or even decades, before the onset of cognitive impairment or memory loss. Specifically, a diminished sense of smell, or hyposmia, is frequently cited as one of the most reliable prodromal indicators of the disease. Until now, the biological "why" behind this sensory erosion remained elusive. The DZNE and LMU study focuses on the intricate connection between two specific regions of the brain: the olfactory bulb and the locus coeruleus.
The olfactory bulb is located in the forebrain and serves as the primary processing station for signals sent from scent receptors in the nasal cavity. The locus coeruleus, situated in the brainstem, acts as a regulatory hub, sending long nerve fibers throughout the brain to modulate various functions, including sleep-wake cycles, cerebral blood flow, and sensory perception. Dr. Lars Paeger, a lead scientist in the study, explains that the locus coeruleus is essential for the "tuning" of the olfactory bulb. When the communication between these two regions is disrupted, the ability to identify and distinguish smells begins to fail.
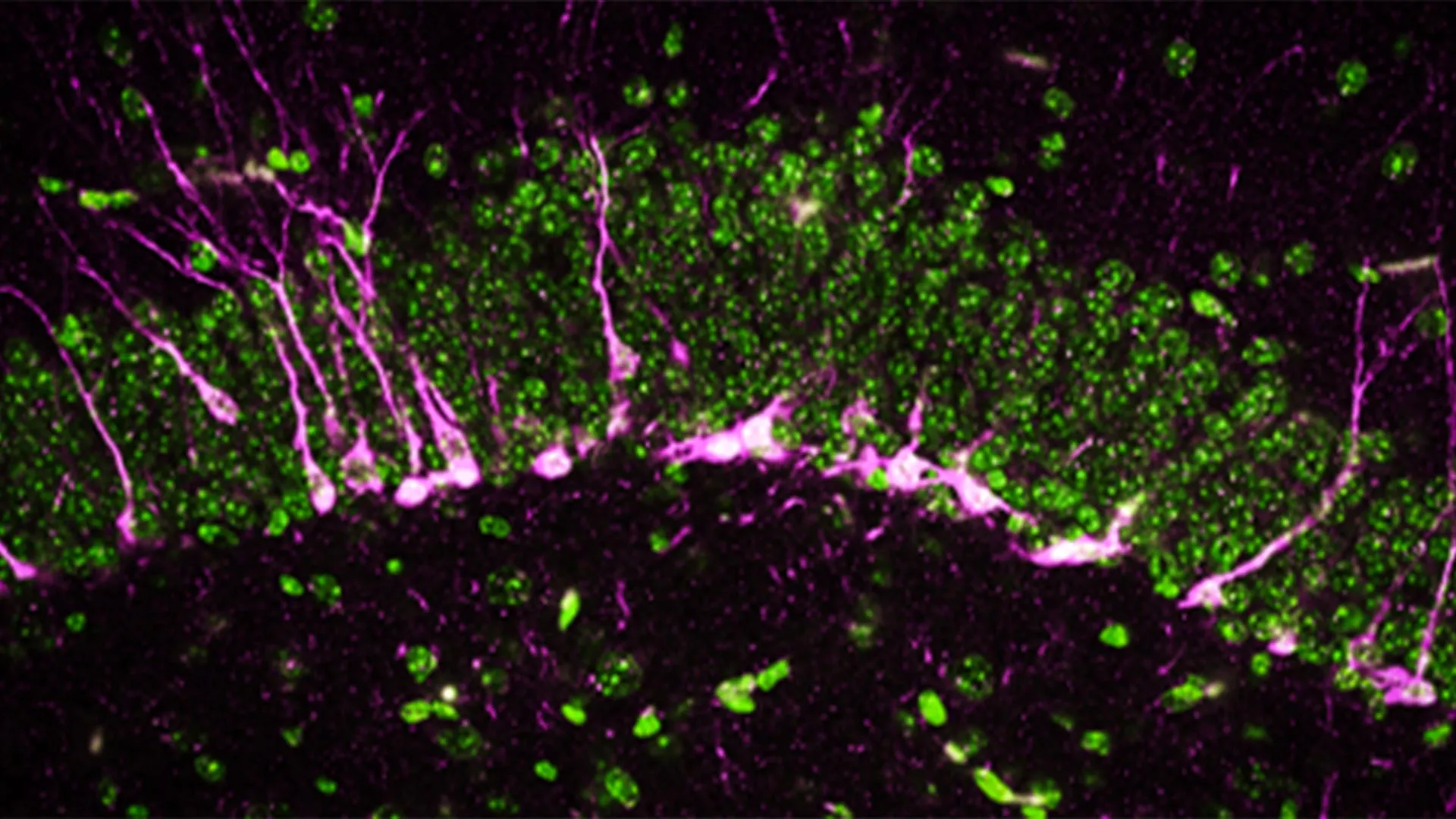
The researchers discovered that in the early stages of Alzheimer’s, the microglia—cells traditionally tasked with protecting the brain by clearing debris and pathogens—begin to behave pathologically. Instead of maintaining the neural network, they start breaking down the nerve fibers that connect the locus coeruleus to the olfactory bulb. This localized immune overreaction effectively severs the regulatory link required for a functional sense of smell.
The Eat-Me Signal: Membrane Alterations and Hyperactivity
A central finding of the study involves the identification of a molecular "eat-me" signal that triggers the microglial attack. Nerve cells are protected by a membrane composed of various fatty molecules. One of these, phosphatidylserine, is typically sequestered on the inner side of the cell membrane. However, the research team found that in the context of early Alzheimer’s, phosphatidylserine shifts from the interior to the exterior surface of the nerve fibers.
This translocation acts as a biological beacon for microglia. In a healthy developing brain, this process is known as "synaptic pruning," a necessary function that removes redundant or weak connections to streamline neural efficiency. In the Alzheimer’s-afflicted brain, however, this pruning becomes destructive. The researchers hypothesize that the shift in membrane composition is caused by neuronal hyperactivity. In the early phases of Alzheimer’s, neurons often exhibit abnormal firing patterns. This "over-firing" appears to stress the cell membrane, causing the phosphatidylserine to flip outward, which the microglia interpret as a signal that the fiber is defective or no longer needed.
Methodology: From Mice to Human PET Scans
The strength of the study lies in its multi-modal approach, which bridged the gap between laboratory models and clinical human data. The research followed a rigorous three-tiered evidentiary path:
- Animal Models: The team utilized transgenic mice designed to exhibit the hallmarks of Alzheimer’s disease, specifically the accumulation of amyloid-beta plaques. They observed that microglial activity in these mice directly correlated with the degradation of the fibers extending from the locus coeruleus to the olfactory bulb.
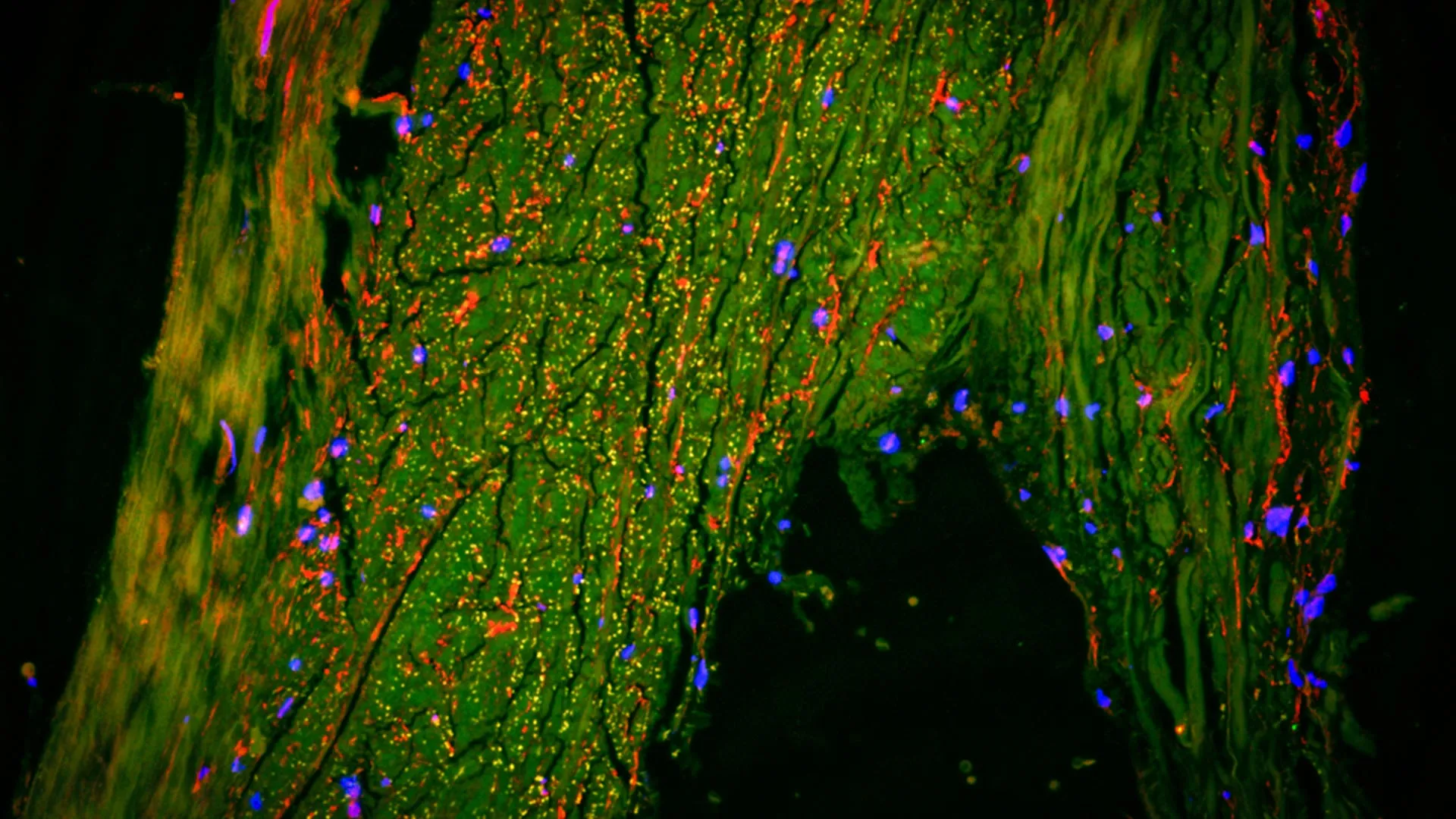
- Human Brain Tissue: To validate the mouse findings, researchers examined post-mortem brain tissue from individuals who had been diagnosed with Alzheimer’s. The analysis confirmed a significant reduction in the density of nerve fibers in the olfactory bulb and a corresponding increase in microglial markers, mirroring the results seen in the animal models.
- PET Imaging: In a critical step for clinical application, the researchers analyzed Positron Emission Tomography (PET) scans from living patients. These patients ranged from those with mild cognitive impairment (MCI) to those with confirmed Alzheimer’s. The PET scans allowed the team to visualize the activity of the locus coeruleus and the integrity of the olfactory system in real-time, providing a "living" confirmation of the immune-mediated damage.
Prof. Dr. Jochen Herms, a co-author of the study and a research group leader at DZNE and LMU, noted that while damage to these nerves had been observed in the past, the immunological cause was the missing piece of the puzzle. The study establishes that these immune events occur very early, often before the widespread accumulation of the amyloid-beta plaques that define the later stages of the disease.
A Timeline of Alzheimer’s Progression
Alzheimer’s disease is now understood as a decades-long process rather than a sudden onset of dementia. The timeline of the disease is generally categorized into three phases: the preclinical (asymptomatic) stage, the mild cognitive impairment (MCI) stage, and finally, the dementia stage.
The research suggests that the microglial attack on the olfactory system occurs during the transition from the preclinical stage to MCI. This is a critical window where the brain is undergoing significant biochemical changes, but the patient may still appear cognitively healthy. By identifying olfactory dysfunction as a direct result of immune-mediated nerve damage, the study provides a biological marker that could potentially be screened for during routine physical exams long before a patient experiences memory loss.
Supporting Data and the Global Context
The implications of this research are underscored by the rising global burden of Alzheimer’s disease. According to the World Health Organization (WHO), more than 55 million people worldwide are currently living with dementia, a figure expected to rise to 139 million by 2050. Olfactory dysfunction is estimated to affect between 85% and 90% of all Alzheimer’s patients, making it one of the most common—yet underutilized—diagnostic markers.
In the United States, the Alzheimer’s Association reports that one in three seniors dies with Alzheimer’s or another form of dementia. The economic impact is equally staggering, with global costs estimated at over $1.3 trillion annually. Research that facilitates early detection is viewed by economists and healthcare providers as the most effective way to reduce the long-term societal costs of the disease, as early intervention can delay the need for intensive institutional care.
Official Responses and Clinical Implications
The scientific community has reacted with cautious optimism to the findings. Members of the Munich-based "SyNergy" Cluster of Excellence, a collaborative research initiative focused on the role of inflammation in neurological diseases, have highlighted the study as a prime example of how "neuro-immunology" is redefining our understanding of brain health.
The clinical implications are particularly relevant given the recent FDA approval of amyloid-beta antibodies, such as lecanemab (Leqembi) and donanemab. These therapies work by clearing amyloid plaques from the brain, but clinical trials have consistently shown that they are most effective when administered during the earliest stages of the disease. Once significant neuronal death has occurred, the benefits of these drugs diminish.
"Our findings could pave the way for the early identification of patients at risk of developing Alzheimer’s," says Prof. Herms. He suggests that if a patient exhibits a declining sense of smell, they could be fast-tracked for comprehensive diagnostic testing, such as PET scans or cerebrospinal fluid analysis. This would allow for the administration of amyloid-beta antibodies at a point in the disease progression where they have the highest probability of slowing cognitive decline.
Analysis of Broader Impacts
This research represents a shift in the "amyloid hypothesis" that has dominated Alzheimer’s research for decades. While amyloid-beta remains a central player, this study emphasizes that the brain’s immune response and the health of the locus coeruleus are equally vital factors. It suggests that Alzheimer’s is not just a disease of "protein misfolding," but also a disease of "immune dysregulation."
Furthermore, the study highlights the importance of the locus coeruleus as a "vulnerability bottleneck" in the brain. Because this small region in the brainstem influences so many different systems, its early degradation has a cascading effect. If clinicians can protect the integrity of the locus coeruleus or modulate the microglial response in the olfactory bulb, they may be able to preserve sensory and cognitive functions for much longer periods.
Future Research and Diagnostic Tools
Moving forward, the research team at DZNE and LMU intends to investigate whether similar immune-mediated mechanisms are responsible for other early sensory symptoms of Alzheimer’s, such as changes in visual processing or auditory sensitivity. There is also interest in developing non-invasive "smell tests" that are sensitive enough to distinguish between normal age-related olfactory decline and the specific type of loss caused by microglial pruning.
In the long term, this could lead to the development of "immunomodulators"—drugs designed to prevent microglia from attacking healthy nerve fibers without compromising their ability to clear harmful pathogens. As the field of neurology moves toward personalized medicine, understanding the specific immunological profile of each patient’s brain will be essential for selecting the right combination of treatments.
The study in Nature Communications serves as a reminder that the most complex challenges in medicine often require looking at the smallest components—in this case, the misplaced molecules on a nerve membrane and the overzealous immune cells that respond to them. By identifying the cause of early olfactory loss, science has moved one step closer to catching Alzheimer’s disease before it begins its most destructive phase.











Leave a Reply