In the wake of The New York Times’ April 2 profile, which painted MEDVi CEO Matthew Gallagher as a "vibe-coded unicorn," the burgeoning telehealth company has found itself embroiled in a swift and escalating wave of scrutiny. Far from the glowing portrayal, subsequent reporting from investigative outlets such as Futurism, Forrester, Drug Discovery & Development, and Business Insider has brought to light a series of troubling revelations. These include a February 2026 FDA warning letter citing misbranding of compounded GLP-1 drugs, multiple lawsuits, a pattern of using Meta advertisements featuring apparently AI-generated "doctor" personas, and significant questions regarding affiliate marketing oversight. Amidst this broader landscape of controversy, one particular product from MEDVi has begun to attract specific attention: QUAD, a compounded erectile dysfunction (ED) therapy that combines a potent cocktail of four active pharmaceutical ingredients, including a drug primarily approved for Parkinson’s disease.
Unpacking QUAD: A Four-Drug Compounded ED Treatment
At the heart of the latest concerns is MEDVi’s QUAD, a compounded sublingual dose product marketed for erectile dysfunction. Unlike FDA-approved medications that undergo rigorous premarket review for safety, effectiveness, and quality, compounded drugs do not receive such scrutiny. This fundamental difference is a critical starting point for understanding the risks associated with QUAD. The formulation is particularly notable for its combination of three well-known PDE5 inhibitors – sildenafil (commonly known as Viagra), tadalafil (Cialis), and vardenafil (Levitra) – with apomorphine. This "stacking" of multiple active ingredients, especially those with similar pharmacological effects and potential side effects, immediately raises red flags within the medical and regulatory communities.
The decision to combine three PDE5 inhibitors is in direct contradiction to established medical guidelines and FDA warnings. The FDA, in its labeling for sildenafil, explicitly states that combinations of PDE5 inhibitors "have not been studied" and "may further lower blood pressure," concluding unequivocally that "the use of such combinations is not recommended." This warning underscores the potential for additive adverse effects, particularly severe hypotension, which can be life-threatening for certain patient populations. Professional drug-interaction databases, widely utilized by U.S. pharmacies and electronic health records, classify the simultaneous prescription of multiple erectile dysfunction agents as a therapeutic duplication, recommending a maximum of one. QUAD, by its very name and composition, contains four such agents, three of which are PDE5 inhibitors.

The Apomorphine Component: A Drug with a Troubled ED History
Perhaps the most contentious ingredient in the QUAD formulation is apomorphine. While apomorphine is FDA-approved in the U.S. for treating motor fluctuations, or "OFF episodes," in adults with advanced Parkinson’s disease, and even used in veterinary medicine to induce vomiting in dogs, it has a distinctly problematic history when it comes to erectile dysfunction.
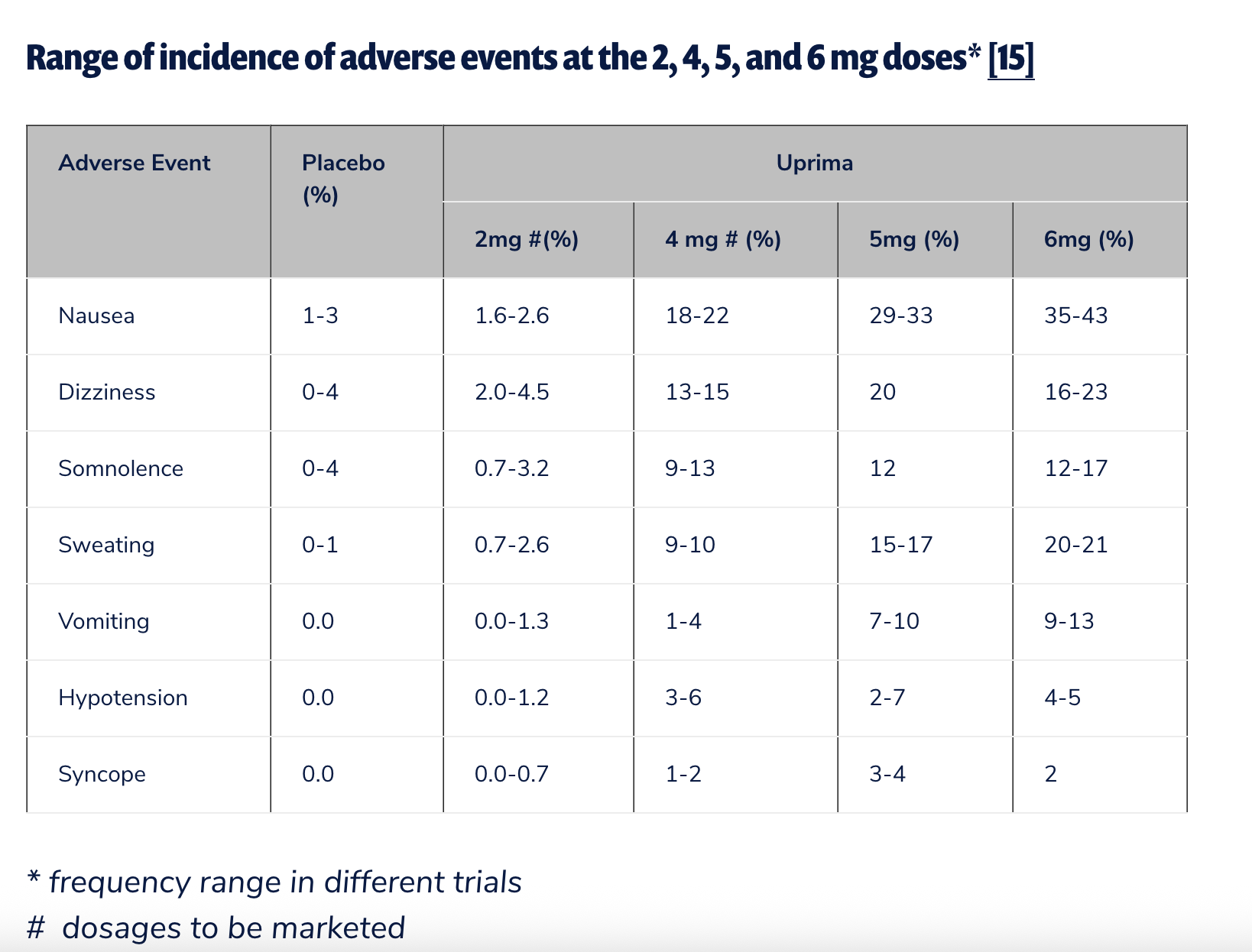
Apomorphine was indeed marketed for ED in Europe, beginning in May 2001 under the brand name Uprima. However, its authorization expired in May 2006, with its then-developer, Abbott Laboratories, choosing not to renew it for commercial reasons. This withdrawal was not without cause; a significant UK study involving over 11,000 patients revealed that two-thirds of men discontinued its use because they felt it simply wasn’t effective. Nausea was also a commonly reported side effect. A 2020 systematic review and meta-analysis published in Archives of Sexual Behavior offered a slightly more positive outlook, suggesting that sublingual apomorphine was generally well tolerated only at lower doses (2 mg and 3 mg), acknowledging that discontinuation rates due to adverse events climbed significantly at higher doses.
In the United States, apomorphine’s journey for ED approval was even shorter and more definitively rejected. In June 2000, a joint venture between Abbott and Takeda, TAP Pharmaceuticals, withdrew its FDA application for sublingual apomorphine (Uprima) after an advisory committee highlighted serious risks, including hypotension and syncope (fainting). Public Citizen, a prominent consumer advocacy organization, played a critical role in opposing its approval. Sidney Wolfe, M.D., then-director of Public Citizen’s Health Research Group, penned a blistering letter to then-CDER director Janet Woodcock, M.D., urging outright rejection. Wolfe’s prescient warning stated, "If the FDA approves this drug, it is certain that it will have to be banned within a short time after marketing begins because of the high probability of further occurrence of the serious injuries it has already been found to cause." FDA approval for ED in the U.S. never materialized, largely due to these safety concerns.
MEDVi’s marketing, however, paints a different picture, claiming apomorphine "ignites desires" and promising rapid action ("hits your system in 10 minutes") with a prolonged 36-hour response window. These claims, especially when presented alongside a product that has a documented history of efficacy issues and safety concerns for ED, raise serious questions about responsible marketing and patient education.
The Compounding Conundrum and Regulatory Ambiguity
The core of the regulatory challenge surrounding QUAD lies in its status as a compounded drug. Compounded medications are custom-prepared by pharmacists for individual patients based on a specific prescription, often to address unique needs such as allergies to commercial product excipients or a requirement for a different dosage form. However, they bypass the rigorous premarket approval process that brand-name and generic drugs undergo with the FDA to demonstrate safety, efficacy, and consistent quality.
The FDA has been increasingly vocal about the appropriate use of compounding, particularly in situations where compounded drugs closely mimic commercially available products. An April 1, 2026 FDA statement, issued just days before Drug Discovery & Development‘s testing of MEDVi’s systems, clarified policies for compounders, especially as the national GLP-1 supply began to stabilize. This document states that the FDA "intends to consider a compounded drug product to be essentially a copy of a commercially available drug product" if it "contains the same APIs as two or more commercially available drug products in the same, similar or easily substitutable strength." Sildenafil, tadalafil, and vardenafil – three of QUAD’s four active ingredients – are all commercially available in the U.S. as FDA-approved products and their generic equivalents.
The FDA guidance also specifies an exception for cases where a prescriber documents a significant clinical difference for an identified individual patient, and sets a threshold for enforcement: the agency generally does not intend to take action against compounders making "essentially-a-copy" products if they fill "four or fewer prescriptions of that compounded drug product during a calendar month." This threshold is designed to accommodate genuine individualized compounding by local pharmacies, not large-scale distribution of complex, multi-ingredient formulations like QUAD, which appear to be marketed broadly.
Crucially, the April 1 FDA document does not explicitly address the compounding of drugs for uses outside their FDA-approved indications (off-label use). Apomorphine’s only FDA-approved indication in the U.S. is for Parkinson’s disease. This omission leaves a regulatory gray area, but the combination of off-label use with a complex, unstudied multi-drug formulation significantly amplifies patient risk.
MEDVi’s Onboarding Process: Questionable Logic and Safety Gaps
The scrutiny extends beyond the product itself to MEDVi’s patient onboarding and screening processes. Drug Discovery & Development‘s test runs on April 7, 2026, revealed significant flaws in the company’s intake questionnaire, particularly concerning critical safety parameters.
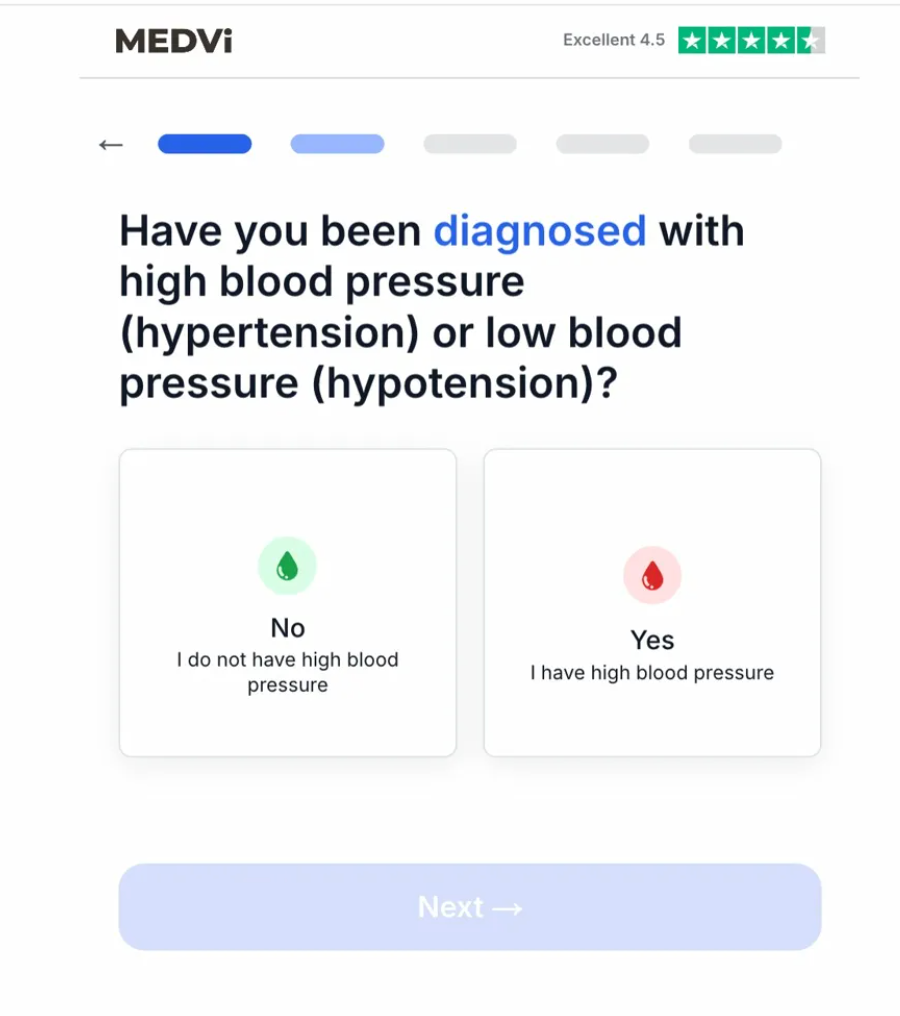
The PDE5 inhibitors (sildenafil, tadalafil, vardenafil) are known vasodilators and can lead to dangerous hypotension in some patients, especially when combined with nitrates or certain blood pressure medications. They carry explicit contraindications with nitrates and warnings regarding interactions with alpha-blockers and other blood pressure medications. Given the additive blood-pressure-lowering effects of combining these drugs, the screening for hypotension is paramount.
However, during the test runs, MEDVi’s intake system presented a question asking whether the user had been "diagnosed with high blood pressure (hypertension) or low blood pressure (hypotension)." Disturbingly, the system offered only two answer cards, both referring to high blood pressure. Hypotension, explicitly mentioned in the question, had no corresponding answer option. When "Yes, I have high blood pressure" was selected, the intake halted with an "Eligibility Status" message stating, "Based on your last answer, we cannot complete your assessment," prompting a review of the answer. This gating logic only fired on hypertension, failing to provide an off-ramp for patients suffering from or prone to hypotension, a condition that could be severely exacerbated by QUAD.
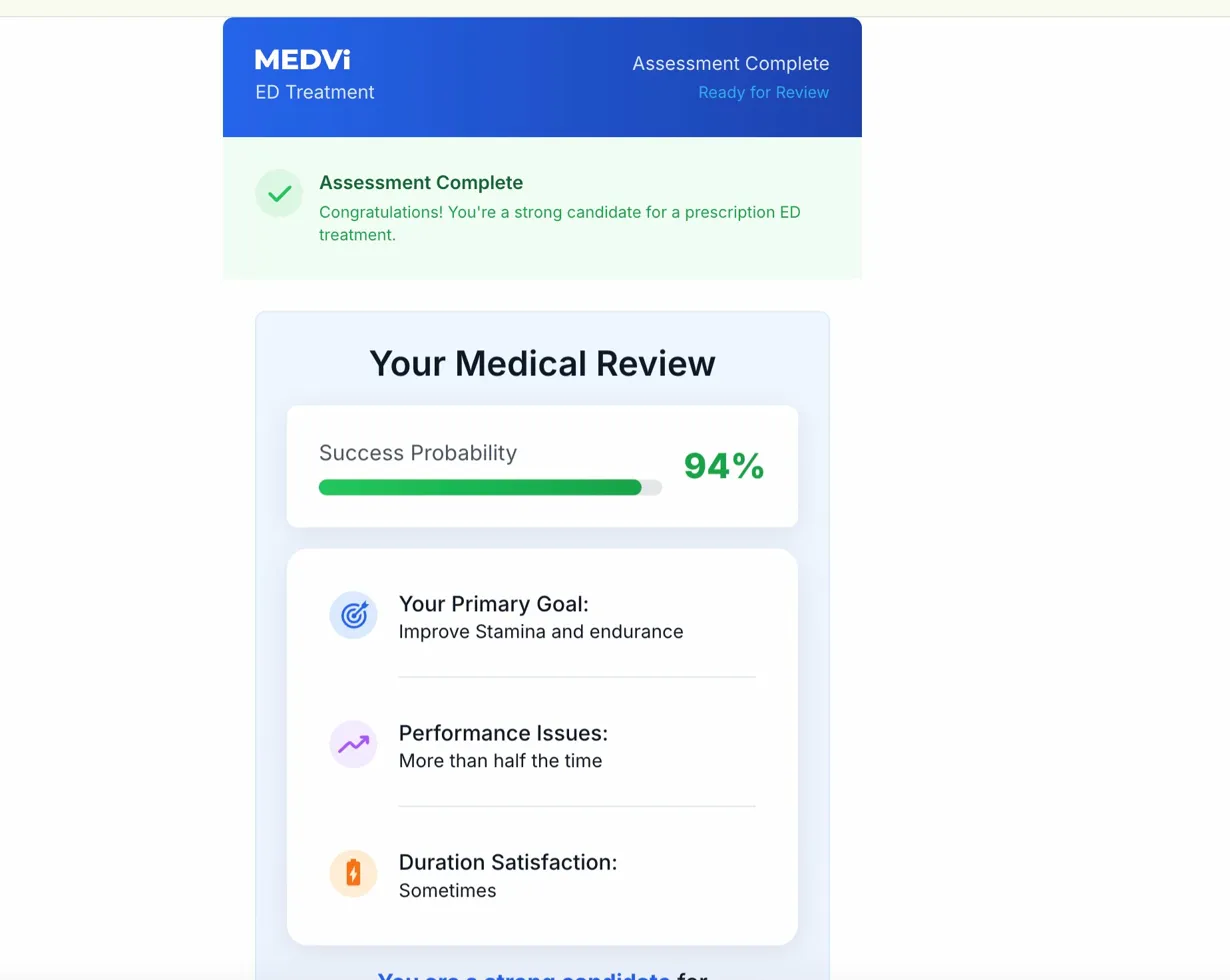
Furthermore, the consistency of a "94% Success Probability" displayed on the "Your Medical Review" screen for QUAD, irrespective of varied answer combinations during testing, raises concerns. This identical 94% figure has also been observed in MEDVi’s GLP-1 weight-loss intake, as documented in a screen recording filed as an exhibit in the federal class action Day v. OpenLoop Health. Such a static, seemingly pre-determined success rate, rather than one tailored to individual patient profiles, undermines the credibility of the medical review process and could create misleading expectations for patients.
The Broader Context of MEDVi’s Legal and Ethical Challenges
The controversy surrounding QUAD is not an isolated incident but part of a larger pattern of regulatory and ethical challenges facing MEDVi. The company’s sudden rise to prominence, amplified by the New York Times profile, has quickly been overshadowed by a cascade of critical reporting.
The February 2026 FDA warning letter for misbranding compounded GLP-1 drugs for weight loss, a different product line, signals a pattern of non-compliance with federal regulations concerning compounded medications. The use of apparently AI-generated "doctor" personas in Meta ads further erodes trust, suggesting a deceptive marketing strategy that blurs the lines between genuine medical advice and promotional content. Several lawsuits against the company and its partners also point to systemic issues, potentially related to misleading advertising, product safety, or patient care.
Affiliate marketing practices also come under scrutiny. An affiliate page titled "MEDVi QUAD Reviews: Is This Fast 4-in-1 ED Formula Worth the Hype?" explicitly states that users can "check your eligibility for MEDVi QUAD in minutes" while simultaneously admitting the therapy "may not be suitable for individuals with certain medical conditions or those taking specific medications. A clinician review is required to determine safety." Another affiliate page, posted on MedicalFoundationofNC.org, acknowledges the FDA warning letter concerned a different product line but then advises readers to "ask hard questions before enrolling." This page, which also discloses potential affiliate commissions, explicitly states it is "not medical advice," "not a medical practice, pharmacy, or licensed healthcare provider," and "not affiliated with The Medical Foundation of North Carolina, Inc., UNC Health Foundation, UNC Health Care, the UNC School of Medicine, or the University of North Carolina at Chapel Hill." The complexity and disclaimers inherent in these affiliate relationships highlight a fragmented and potentially problematic marketing ecosystem.
Official Responses and the Call for Accountability

In response to inquiries about QUAD, an HHS public affairs spokesperson stated that "the FDA generally does not discuss compliance matters except with the company involved" and that "when violations occur, the agency takes action as appropriate." The spokesperson referred Drug Discovery & Development to the aforementioned April 1 FDA statement on compounders. This official stance, while standard, underscores the agency’s watchful role without providing specific commentary on MEDVi’s current practices regarding QUAD.
MEDVi and OpenLoop Health, mentioned in the context of the Day v. OpenLoop Health lawsuit, were contacted for comment but did not immediately provide a response. Nicholas Chimicles, lead counsel for the plaintiffs in the class action, declined to comment further on the litigation but confirmed awareness of the recent New York Times profile of MEDVi. The silence from the implicated companies leaves many questions unanswered, particularly regarding their rationale for combining these potent drugs and their response to documented safety concerns.
Implications for Patient Safety and the Future of Telehealth
The case of MEDVi’s QUAD raises profound implications for patient safety, regulatory oversight of compounded drugs, and the ethical boundaries of telehealth marketing. The combination of three PDE5 inhibitors with apomorphine, a drug previously deemed unsuitable for ED due to efficacy and safety concerns, creates a pharmacological "stack" with an unknown safety profile. The potential for severe adverse events, particularly dangerous hypotension, is significantly heightened by this untested combination, especially given the known contraindications of PDE5 inhibitors.
The apparent flaws in MEDVi’s patient intake process, specifically the failure to adequately screen for hypotension, suggest a critical lapse in patient protection. The standardized "94% success probability" further indicates a potential oversimplification or misrepresentation of treatment outcomes. As telehealth platforms rapidly expand, the onus on companies to ensure robust, evidence-based medical screening and transparent communication of risks and benefits becomes paramount.
The ongoing scrutiny of MEDVi, encompassing its GLP-1 products, marketing tactics, and now its ED therapies, serves as a cautionary tale. It highlights the challenges regulators face in keeping pace with rapidly evolving digital health models and the critical need for vigilance from consumers, medical professionals, and investigative journalism to ensure that innovation does not come at the expense of patient safety and ethical practice. The questions surrounding QUAD demand thorough investigation and underscore the fundamental principle that medical treatments, especially complex drug combinations, must prioritize proven safety and efficacy above all else.















Leave a Reply