Researchers at Johns Hopkins Medicine have announced that a newly funded study by the National Institutes of Health is significantly advancing a novel approach to the treatment of Alzheimer’s disease, focusing on a specific brain protein that facilitates the production of a vital signaling gas. The research centers on Cystathionine γ-lyase, an enzyme commonly referred to as CSE, which is the primary catalyst for the internal production of hydrogen sulfide. While hydrogen sulfide is widely recognized by the public for its characteristic "rotten egg" odor and its toxicity in industrial settings, it is increasingly understood by the scientific community as a critical "gasotransmitter" that modulates essential physiological functions within the human brain and cardiovascular system.
According to the study’s lead author, Bindu Paul, M.S., Ph.D., an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, this protein appears to be a cornerstone of memory formation and cognitive preservation. The findings, recently published in the Proceedings of the National Academy of Sciences (PNAS), suggest that the depletion of CSE may be a primary driver of the neurodegeneration and cognitive decline observed in Alzheimer’s patients. By investigating the biochemical pathways of this enzyme, the research team aims to determine whether pharmaceutical interventions designed to boost CSE activity could protect neurons and slow the progression of dementias that currently lack effective long-term treatments.
The Biological Significance of Hydrogen Sulfide in the Brain
Hydrogen sulfide ($H_2S$) belongs to a family of gaseous signaling molecules that also includes nitric oxide and carbon monoxide. In the brain, these gases act as messengers that can pass through cell membranes without the need for traditional receptors, allowing for rapid and widespread cellular communication. However, the therapeutic application of $H_2S$ is fraught with challenges. In high concentrations, the gas is a potent metabolic poison; yet, in the infinitesimal amounts naturally synthesized by the body, it serves as a powerful antioxidant and neuroprotector.
The central challenge for researchers is finding a way to maintain these delicate, naturally occurring levels within the neurons. Earlier studies conducted in murine models indicated that supplemental hydrogen sulfide could shield neurons from the oxidative stress that characterizes aging and disease. Because direct delivery of the gas to the brain is unsafe and impractical for human patients, the Johns Hopkins team is focusing on the "upstream" source: the CSE enzyme itself. By understanding how to regulate the protein that produces the gas, scientists hope to develop a safer, more controlled method of maintaining brain health.
A Timeline of Genetic and Biochemical Discovery
The current breakthrough is the culmination of more than fifteen years of rigorous investigation into the CSE enzyme. The timeline of this research reflects a growing understanding of how systemic proteins influence localized brain function.
In 2008, the CSE protein was first identified as a major regulator of blood vessel function and blood pressure. Researchers discovered that mice lacking the enzyme suffered from significant cardiovascular issues, establishing the protein’s role in maintaining the integrity of the circulatory system. By 2014, a research team led by Solomon Snyder, M.D., D.Sc., D.Phil.—a legendary figure in neuroscience and professor emeritus at Johns Hopkins—reported that CSE played a protective role in models of Huntington’s disease. This was a pivotal moment, as it shifted the focus from the cardiovascular system to the central nervous system.
The connection to Alzheimer’s disease became clearer in 2021. The Johns Hopkins group discovered that CSE function was severely compromised in mice specifically bred to exhibit Alzheimer’s symptoms. They observed that administering micro-doses of hydrogen sulfide helped restore some degree of cognitive function in these mice. However, those earlier studies utilized mice that already possessed multiple genetic mutations linked to neurodegeneration. The latest 2024 study represents a significant leap forward because it isolates the role of CSE. By using mice that are genetically normal except for the absence of the CSE protein, the researchers could definitively attribute cognitive decline to the loss of this single enzyme, rather than a combination of other Alzheimer’s-related mutations.
Analyzing the Impact of CSE Deficiency on Memory
To quantify the impact of the CSE protein on cognitive performance, the research team employed the Barnes maze, a standard behavioral test used to measure spatial learning and memory in rodents. The maze consists of a circular platform with multiple holes around the perimeter; only one hole leads to a dark, safe "escape box," while the others are "false" openings.
The experiment compared normal mice with those engineered to lack the CSE protein. At the age of two months, both groups of mice performed with equal proficiency, successfully locating the escape route within a three-minute window. This indicated that the absence of the protein did not cause immediate birth defects or developmental delays in basic navigation. However, as the mice aged, the disparity became stark. By the age of six months, the CSE-deficient mice showed a profound inability to remember the location of the shelter. While the control mice continued to navigate the maze with ease, the deficient mice struggled, exhibiting a progressive loss of spatial memory.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute to CSE loss," noted Suwarna Chakraborty, a researcher in Paul’s lab and the study’s first author. This age-related decline closely mirrors the clinical progression of Alzheimer’s disease in humans, where symptoms often remain dormant until middle or late age.
Cellular Erosion and the Breakdown of the Blood-Brain Barrier
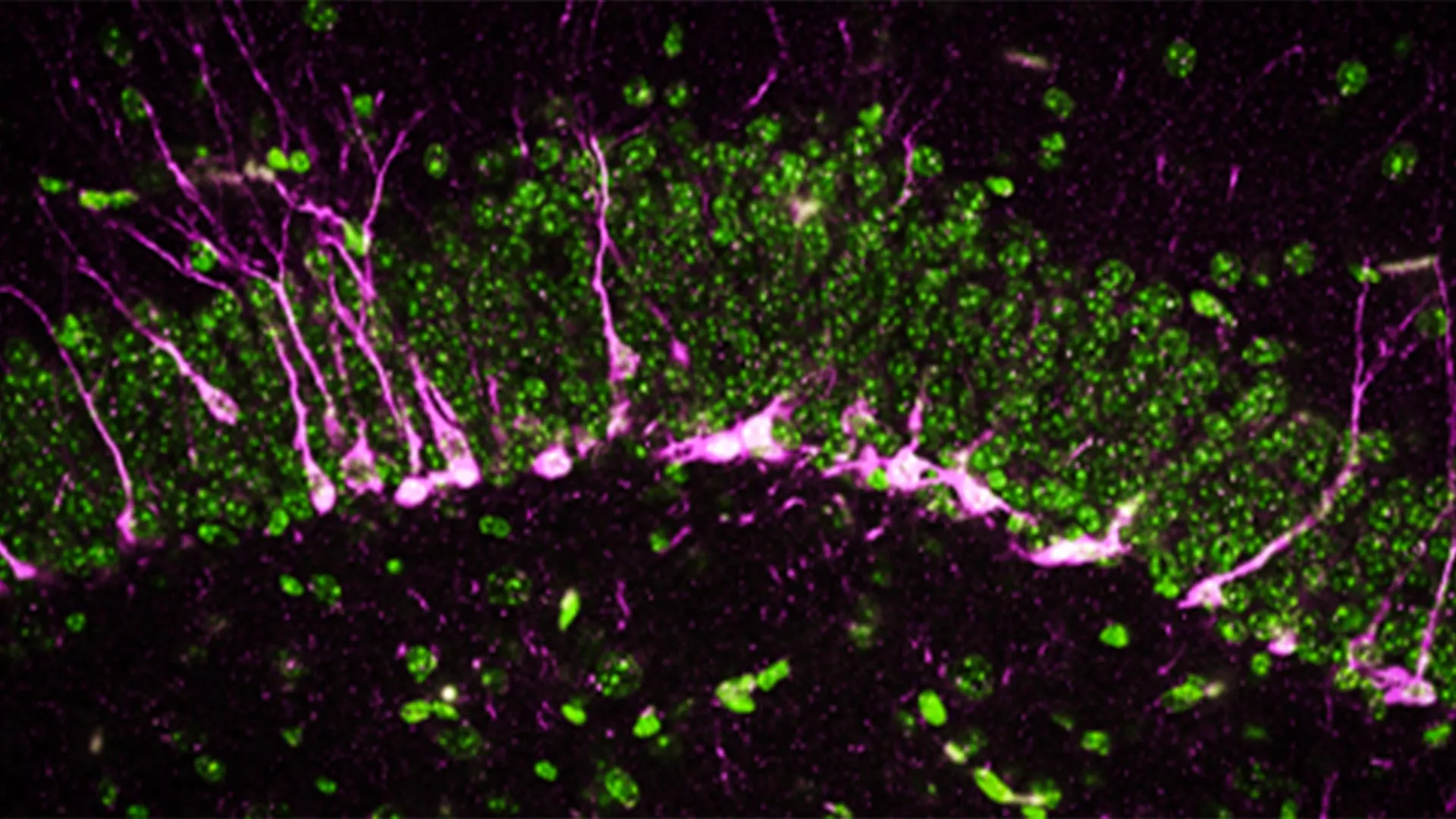
Beyond behavioral changes, the study provided a deep dive into the structural and cellular damage caused by the absence of CSE. Using high-powered electron microscopy and biochemical analysis, the team examined the hippocampus—the region of the brain responsible for converting short-term memories into long-term ones.
The researchers identified several "hallmarks" of Alzheimer’s disease in the brains of the CSE-deficient mice:
- Reduced Neurogenesis: The birth of new neurons is essential for maintaining cognitive flexibility. In the absence of CSE, the proteins required for neurogenesis were either significantly reduced or entirely absent.
- DNA Damage and Oxidative Stress: Without the antioxidant properties of the hydrogen sulfide produced by CSE, the brain cells were left vulnerable to free radicals, leading to extensive DNA fragmentation.
- Blood-Brain Barrier Failure: Perhaps most significantly, the researchers observed large breaks in the blood vessels within the brain. The blood-brain barrier (BBB) is a selective filter that prevents toxins and pathogens in the blood from entering brain tissue. The failure of this barrier is a known precursor to the inflammatory cycles seen in Alzheimer’s.
Co-first author Sunil Jamuna Tripathi emphasized that the mice lacking CSE were compromised at multiple physiological levels. The damage was not confined to a single pathway but represented a systemic failure of the brain’s protective and regenerative mechanisms.
The Broader Impact on Alzheimer’s Research and Treatment
The implications of this research are substantial given the current state of Alzheimer’s care. According to the U.S. Centers for Disease Control and Prevention (CDC), more than 6 million Americans are currently living with Alzheimer’s, a number projected to nearly triple by 2060. Despite billions of dollars in research funding, the medical community has struggled to produce treatments that do more than temporarily manage symptoms.
Most current Alzheimer’s research focuses on the "amyloid hypothesis"—the idea that the buildup of amyloid-beta plaques is the primary cause of the disease. However, the limited clinical success of amyloid-targeting drugs has led many researchers to look for alternative "upstream" targets. The Johns Hopkins study suggests that the CSE/hydrogen sulfide pathway could be one such target. If the loss of CSE precedes the formation of plaques and tangles, then stabilizing this enzyme could potentially prevent the neurodegenerative process from ever gaining momentum.
"This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," stated Solomon Snyder, the study’s co-corresponding author. Snyder, who retired from the Johns Hopkins faculty in 2023 but continues to contribute to the field, noted that identifying a single protein with such a profound impact on multiple disease markers is a rare and promising development.
Conclusion and Future Directions
The research led by Bindu Paul and her colleagues provides a compelling case for the development of CSE-targeted therapies. The next phase of research will likely involve screening for small molecules or compounds that can enhance the expression or activity of the CSE enzyme without crossing the threshold into toxicity.
The study was a massive collaborative effort involving institutions such as Case Western Reserve University, the Leibniz Institute for Analytical Sciences in Germany, the Medical University of South Carolina, and West Virginia University. Funding was provided by a wide array of organizations, including the National Institutes of Health, the Department of Defense, and the American Heart Association, highlighting the cross-disciplinary interest in this pathway.
As the global population ages, the urgency for a breakthrough in neurodegenerative treatment grows. While the transition from mouse models to human clinical trials is a long and complex process, the identification of the CSE protein offers a new and scientifically rigorous path forward. By focusing on the fundamental chemistry of brain signaling and the protective power of hydrogen sulfide, researchers at Johns Hopkins are opening a new chapter in the fight against Alzheimer’s disease.












Leave a Reply