In a significant breakthrough for antibiotic discovery, scientists have elucidated the sophisticated mechanisms by which bacteriophages, viruses that infect bacteria, dismantle a crucial protein essential for bacterial cell wall construction. This research, published in the February 26 issue of Nature, unveils how distinct viral proteins have independently evolved to inhibit MurJ, a key flippase protein responsible for transporting peptidoglycan precursors across the bacterial inner membrane. The remarkable convergence of these viral strategies against MurJ underscores its potential as a highly promising new target for the development of desperately needed antibiotics.
The study was spearheaded by Yancheng Evelyn Li, a graduate student within the laboratory of Professor Bil Clemons at the California Institute of Technology (Caltech). Professor Clemons, the Arthur and Marian Hanisch Memorial Professor of Biochemistry, served as the corresponding author for the groundbreaking research. This investigation into viral attack mechanisms offers a novel perspective on combating the escalating global threat of antibiotic-resistant bacteria.
The Escalating Crisis of Antibiotic Resistance
The relentless adaptability of bacteria poses a profound and growing threat to public health worldwide. As Professor Clemons articulated, "Evolution is powerful, and in bacteria, resistance to antibiotics develops quickly. This means that we now deal with bacteria that are resistant to all the medicines that we have." This evolutionary arms race has led to a scenario where once-treatable infections are becoming increasingly intractable. The statistics are stark: in the United States alone, tens of thousands of lives are lost annually to antibiotic-resistant bacterial infections, a figure that is projected to rise dramatically in the coming years. This alarming trend necessitates an urgent and sustained search for novel therapeutic strategies.
With existing antibiotic classes losing their efficacy at an accelerated pace, the scientific community is actively exploring entirely new vulnerabilities within bacterial biology. This quest involves moving beyond traditional targets and delving into the fundamental processes that govern bacterial survival and proliferation.
Targeting the Bacterial Fortress: The Peptidoglycan Cell Wall
A long-standing and highly attractive area of research in antibiotic development has been the intricate pathway bacteria employ to synthesize peptidoglycan. This rigid, mesh-like polymer forms the essential cell wall, providing structural integrity and protecting bacteria from osmotic lysis. The peptidoglycan biosynthesis pathway is particularly appealing as an antibiotic target because peptidoglycan is a unique and vital component of bacterial cell walls, entirely absent in human cells. "Peptidoglycan is a unique feature of bacteria, and that makes it an attractive antibiotic target," Professor Clemons emphasized.
The therapeutic potential of targeting this pathway is not entirely new. For decades, antibiotics such as penicillin and its derivatives like amoxicillin have successfully disrupted later stages of peptidoglycan production, effectively weakening the bacterial cell wall and leading to cell death. However, the emergence of resistance mechanisms has diminished the effectiveness of these established drugs, spurring the search for earlier and more fundamental points of intervention.
Key Players in Cell Wall Construction: MraY, MurG, and MurJ
Central to the assembly of the peptidoglycan cell wall are three critical proteins that facilitate the transport of essential building blocks across the bacterial inner membrane: MraY, MurG, and MurJ. These proteins act as molecular transporters, ferrying the necessary components to the exterior of the cell where they can be assembled into the robust peptidoglycan layer. The critical nature of these proteins means that any disruption to their function can halt peptidoglycan synthesis, leading to bacterial death. Consequently, these proteins represent highly sought-after drug targets.
While the general roles of MraY, MurG, and MurJ are understood, Professor Clemons noted that "important mechanistic details remain unclear," highlighting the need for deeper investigation into their precise functions and interactions. Currently, no approved drugs directly inhibit these three pivotal proteins. Nevertheless, the potential for developing such inhibitors is considerable. "We do know that we can find small molecules, either derived from nature or synthesized in chemical libraries, that will inhibit these proteins," Professor Clemons stated, expressing optimism about future drug discovery efforts. He also pointed to exciting recent discoveries indicating that bacteriophages have already evolved sophisticated strategies to target this very pathway.
Bacteriophages: Nature’s Bacterial Predators
Bacteriophages, commonly referred to as phages, are viruses that specifically infect bacteria. To complete their life cycle, phages must gain entry into a bacterial cell, replicate themselves, and then egress to infect new bacterial hosts. A critical hurdle for phages exiting a bacterial cell is navigating the formidable peptidoglycan layer. Professor Clemons explained, "Getting back out means that they have to get past the peptidoglycan layer. Because it acts like chainmail, the phages get stuck if they can’t break through it."
The research team at Caltech, led by Professor Clemons, focuses on studying small bacteriophages that utilize single-stranded DNA or RNA genomes. These viruses, characterized by their compact genetic material, employ elegant and efficient strategies to overcome bacterial defenses and achieve replication. In a notable prior publication in Science in 2023, the Clemons lab reported on their work with the well-studied phage φX174, a virus with a long history of research at Caltech.
Viral Proteins That Precisely Target and Disable MurJ
The strategy employed by these small phages often involves the production of specialized protein antibiotics known as single-gene lysis proteins, or Sgls. These Sgls are key to the phage’s ability to break free from the host bacterium. Li and Clemons’ recent work has zeroed in on Sgls that specifically target MurJ, one of the essential cell wall proteins.
MurJ functions as a flippase, a type of transporter that facilitates the movement of molecules across a membrane. In the context of peptidoglycan biosynthesis, MurJ is responsible for translocating peptidoglycan precursors from the inner side of the bacterial membrane to the outer side, where they can be incorporated into the growing cell wall. Previous research from collaborating institutions had already established that two distinct and unrelated Sgls, known as SglM and SglPP7, achieve bacterial lysis by inhibiting MurJ.
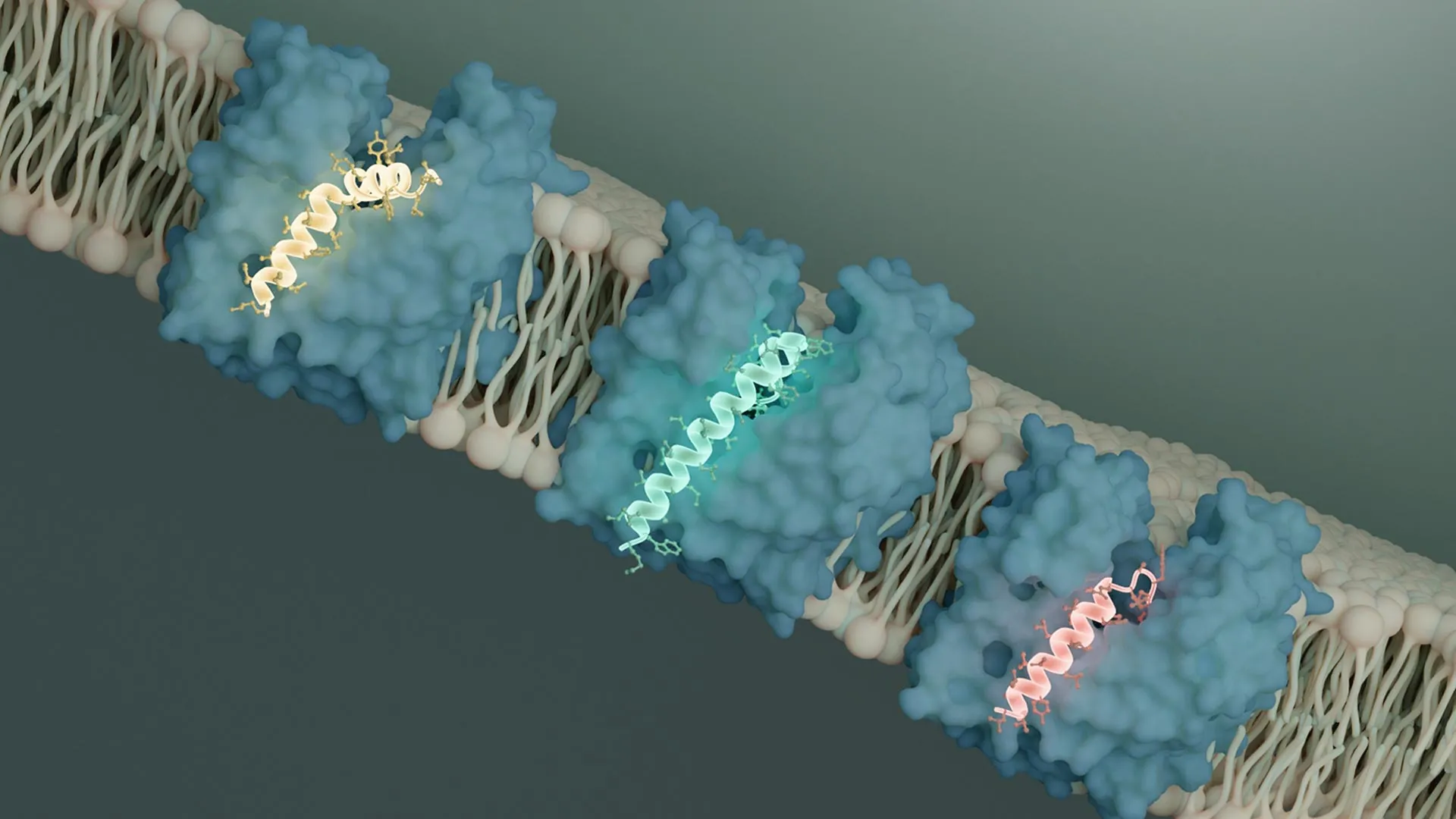
To unravel the molecular details of this inhibition, Li employed cryo-electron microscopy (cryo-EM) at Caltech’s Beckman Institute Biological and Cryogenic Transmission Electron Microscopy (Cryo-EM) Resource Center. Cryo-EM allows scientists to visualize the three-dimensional structures of proteins and their complexes at near-atomic resolution. Flippases like MurJ operate by undergoing conformational changes, essentially "flipping" to expose the transported molecule to either side of the membrane without creating a permanent pore. When MurJ binds its peptidoglycan precursor cargo on the inside of the cell, it undergoes a structural rearrangement to release the molecule on the outside.
Li’s cryo-EM analyses revealed a striking similarity in how both SglM and SglPP7 interact with MurJ. The viral proteins were found to bind to a specific groove on the MurJ protein. This binding event effectively prevents MurJ from undergoing the necessary conformational change required for its transport function.
"It is clear that both of these Sgls bind to MurJ in an outward-facing conformation, locking it into this position," Li stated. This observation holds significant implications for drug development. The outward-facing conformation of MurJ is exposed to the extracellular environment, making it potentially more accessible to therapeutic molecules than an inward-facing conformation that would be shielded by the membrane.
Convergent Evolution: A Testament to MurJ’s Vulnerability
The researchers were not only intrigued by the mechanism of inhibition but also by the evolutionary implications of their findings. Professor Clemons expressed his surprise at the degree of convergence: "These peptides, which have no evolutionary links to each other, have both figured out how to target MurJ in a very similar way. These are two examples of convergent evolution, in which different evolutionary paths arrive at the same solution. We were surprised!"
Convergent evolution is a powerful biological phenomenon where unrelated organisms independently evolve similar traits or solutions to environmental challenges. In this case, different bacteriophage lineages, facing the same obstacle of the bacterial cell wall, have independently evolved distinct viral proteins that achieve the same outcome by binding to the same critical site on MurJ. This suggests that MurJ is a particularly vulnerable and effective target for phages seeking to lyse bacteria.
Given the rapid evolutionary rate of viruses, the research team posits that numerous other phages likely harbor similar Sgls capable of targeting MurJ. The relative ease of isolating bacteriophages and sequencing their genomes offers a rich opportunity for discovering additional biological insights and, crucially, identifying new antibiotic targets.
Expanding the Discovery: A Third Viral Inhibitor
Furthering their investigation, the researchers analyzed the genome of another bacteriophage with the assistance of a collaborator. This analysis led to the identification of a novel Sgl, designated SglCJ3, originating from a phage genome predicted to be named Changjiang3. Using cryo-EM, Li proceeded to determine the structure of SglCJ3 in complex with MurJ. The results were consistent with their previous findings: SglCJ3 also binds to MurJ and locks it into the same outward-facing conformation, thereby inhibiting its function.
"This is a third genome that evolved a distinct peptide to inhibit the same target in a similar way," Professor Clemons remarked. "It is the first strong evidence that evolution identifies MurJ as a great target for killing bacteria, which means we should follow evolution’s lead and develop therapeutics that target MurJ. This demonstrates the power of basic biology to help us solve problems in medicine. Our path is set on leveraging Sgl discovery, and we hope to continue to be supported to turn these concepts into realities."
This repeated evolutionary solution highlights MurJ as a highly conserved and essential bacterial protein, making it an exceptionally promising target for broad-spectrum antibiotic development. The implications of this discovery are far-reaching, offering a potential new avenue for combating the growing threat of multidrug-resistant infections.
Authorship and Funding Sources
The research paper detailing these findings is titled "Convergent MurJ flippase inhibition by phage lysis proteins." Beyond the primary authors, Yancheng Evelyn Li and Bil Clemons, the research team included Caltech graduate student Grace F. Baron and contributions from Francesca S. Antillon, Karthik Chamakura, and Ry Young of Texas A&M University. This critical research was made possible through the generous support of several prominent organizations, including the Chan Zuckerberg Initiative, the National Institutes of Health, the G. Harold and Leila Y. Mathers Foundation, and the Center for Phage Technology at Texas A&M, which is jointly sponsored by Texas A&M AgriLife. The collaborative nature of this research underscores the global effort required to address complex scientific challenges like antibiotic resistance.















Leave a Reply