Detailed three dimensional reconstructions of a key sensory structure in ctenophores reveal far greater structural and functional complexity than scientists previously recognized. The results suggest that a simple brain like system may have existed in some of the earliest animals, offering new insight into how nervous systems evolved. This groundbreaking research, published in the esteemed journal Science Advances, challenges long-held assumptions about the simplicity of early animal nervous systems and suggests that the fundamental building blocks of complex behavior may have emerged far earlier in evolutionary history than previously understood.
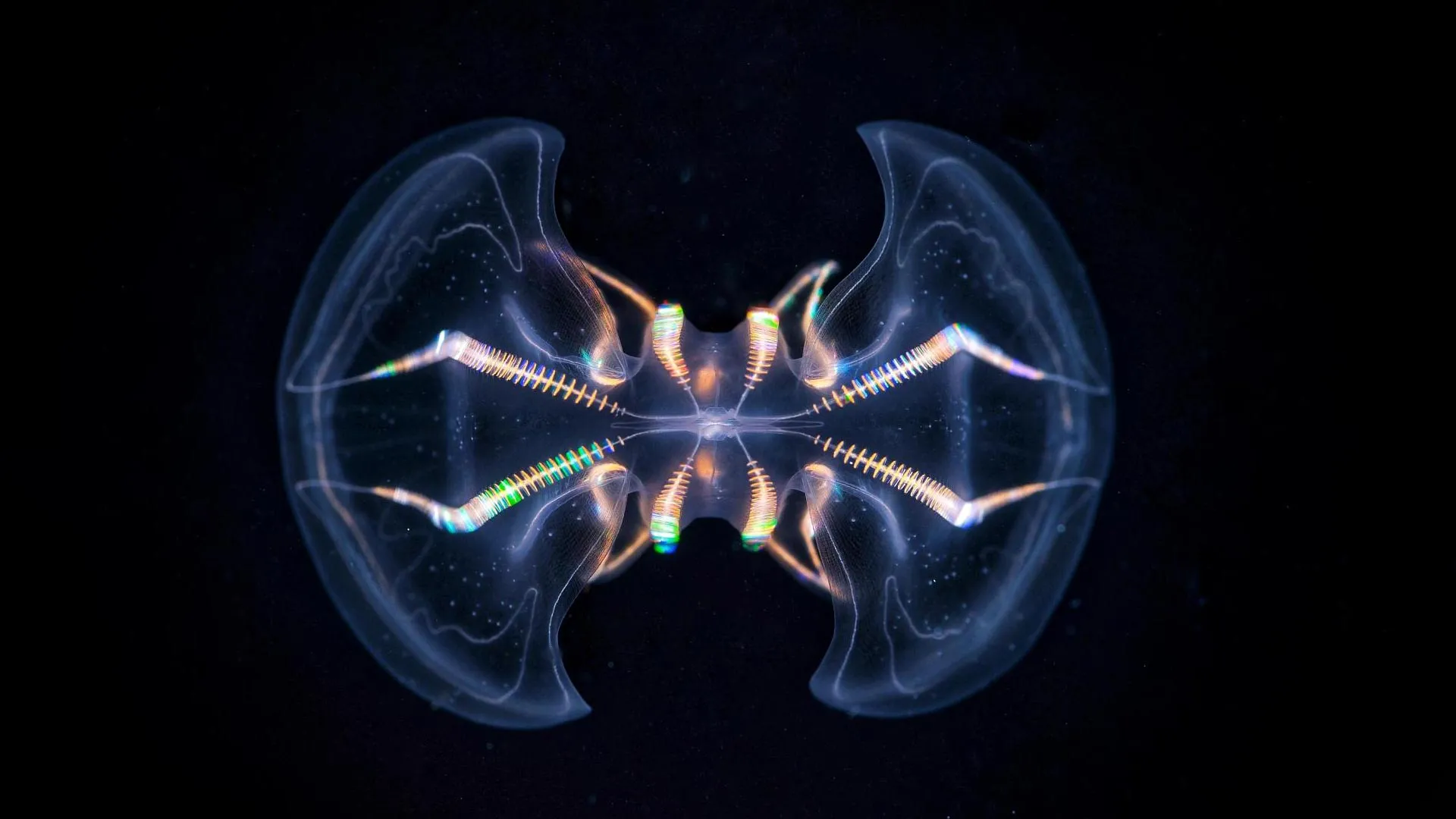
Ctenophores, commonly known as comb jellies, are ethereal, gelatinous marine invertebrates that have graced Earth’s oceans for an estimated 550 million years. Their ancient lineage places them among some of the earliest branching animals, predating many familiar phyla, including cnidarians (like jellyfish and corals) and even some of the earliest bilaterians. These delicate creatures possess a unique and vital sensory apparatus known as the aboral organ (AO). This specialized structure plays a crucial role in their survival, enabling them to perceive and respond to fundamental environmental cues such as gravity, hydrostatic pressure, and light. The precise mechanisms by which these seemingly simple organisms navigate their environment have long been a subject of scientific intrigue.
A recent, comprehensive morphological study has significantly advanced our understanding of this enigmatic organ. Through the application of cutting-edge imaging technologies, researchers have unveiled a level of complexity within the aboral organ that surpasses what was discernible through earlier, less advanced investigative methods. "We show that the AO is a complex and functionally unique sensory system," stated Pawel Burkhardt, group leader at the Michael Sars Centre, University of Bergen, and a key figure in the research. "Our study profoundly enhances our understanding of the evolution of behavioral coordination in animals." This sentiment underscores the transformative nature of the findings, suggesting a reevaluation of the cognitive capabilities of early animal life.
Unraveling the Cellular Architecture of an Ancient Sensory Hub
To meticulously dissect the intricate internal organization of the aboral organ, the research team, in collaboration with Maike Kittelmann at Oxford Brookes University, employed state-of-the-art volume electron microscopy. This advanced technique provides an unparalleled level of detail, allowing for the generation of extraordinarily precise three-dimensional reconstructions of biological structures. Unlike conventional microscopy, which offers a two-dimensional glimpse, volume electron microscopy permits scientists to virtually slice through an entire organ, cell by cell, and reconstruct its complete architecture in three dimensions.
The exhaustive analysis yielded a remarkable discovery: the aboral organ houses an astonishing diversity of cell types. Researchers identified a total of 17 distinct cell types within this single sensory structure. Crucially, this included 11 previously unidentified secretory and ciliated cell types. The sheer variety of specialized cells within the AO strongly indicates its role as a sophisticated, multimodal sensory organ, capable of integrating information from multiple environmental sources simultaneously.
Anna Ferraioli, a postdoctoral researcher at the Michael Sars Centre and the first author of the study, expressed her astonishment at the findings. "I was amazed almost immediately by the morphological diversity of the aboral organ cells. Working with volume EM data feels like discovering new exciting things every day," she remarked. Ferraioli further emphasized the organ’s distinctiveness: "The AO has a striking complexity when compared to apical organs of cnidarian and bilaterian. It is so unique!" This comparison highlights how the aboral organ stands apart even from the sensory structures of closely related or similarly ancient animal groups, suggesting an independent evolutionary trajectory for sensory processing.
A Hybrid Neural Communication System Emerges
Beyond its remarkable cellular diversity, the aboral organ exhibits a profound and intimate connection with the ctenophore’s broader nervous system. Ctenophores possess a nerve net, a decentralized network of interconnected neurons that forms a continuous, diffuse structure throughout their entire bodies. This represents a fundamental difference from the centralized nervous systems found in many more complex animals, such as vertebrates.
The research revealed that this nerve net establishes direct synaptic connections with cells situated within the aboral organ. Synapses are specialized junctions where neurons transmit signals to other neurons or to effector cells, such as muscles or glands. These direct connections create a dedicated pathway for bidirectional communication between the central nerve net and the sensory organ. Signals can be transmitted from the environment, processed by the AO, and then relayed to the rest of the body for coordinated action.
However, the communication mechanisms extend beyond these direct synaptic links. Many cells within the aboral organ were observed to contain a substantial number of vesicles. Vesicles are small sacs that store and release chemical messengers, known as neurotransmitters or neuromodulators. The presence of numerous vesicles suggests that these cells can release widespread chemical signals through a process termed volume transmission. This form of signaling involves the diffusion of chemicals across extracellular spaces, influencing a broader range of target cells than is typically achieved through targeted synaptic transmission.
The combined evidence of both synaptic and non-synaptic signaling pathways points to a sophisticated and flexible communication system within the aboral organ. This hybrid approach allows for both rapid, precise signaling and broader, modulatory effects. "I think our work provides an important perspective on how much we can learn from studying morphology," Ferraioli explained. Reflecting on the functional significance of the AO, she offered a compelling analogy: "I would say that the AO is definitely not like our brain, but it could be defined as the organ that ctenophores use as a brain." This statement, while acknowledging the anatomical differences, emphasizes the functional role of the AO as a central processing unit for sensory information and behavioral coordination in ctenophores.
Rethinking the Origins of Nervous Systems and Brains
The implications of this research extend significantly to our understanding of the evolutionary trajectory of nervous systems and the emergence of centralized brains. The team also delved into the expression patterns of key developmental genes within ctenophores. These genes are crucial for orchestrating the complex processes of embryonic development and body organization in virtually all multicellular animals.
While many of these developmental genes are conserved across diverse animal lineages, their specific patterns of expression within ctenophores were found to differ substantially from those observed in other animal groups, particularly cnidarians and bilaterians. This divergence in gene expression suggests that the aboral organ, despite its complex sensory functions, may not be directly homologous to the brains or even the apical organs found in other animal lineages. Instead, it points towards an independent evolutionary pathway for developing sophisticated sensory processing and coordination mechanisms.
"In other words," Burkhardt elaborated, "evolution seems to have invented centralized nervous systems more than once." This assertion is a powerful statement, suggesting that the evolutionary pressures favoring centralized information processing may have arisen independently in different lineages, leading to convergent solutions for complex behavior. This challenges the traditional view of a single origin for complex nervous systems and opens the possibility of multiple, independent evolutionary experiments in neural organization.
Linking Neural Structure to Coordinated Behavior
Further corroboration for these groundbreaking findings comes from a related, parallel study. Led by Kei Jokura at the National Institute for Basic Biology in Japan, and involving Prof. Gaspar Jekely from Heidelberg University, this separate research effort also included Pawel Burkhardt. In this complementary investigation, scientists focused on reconstructing the complete neural wiring of the ctenophore’s gravity-sensing organ, a crucial component of the aboral organ system.
By integrating high-speed imaging techniques with detailed three-dimensional reconstructions of over 1,000 individual neurons, this study elucidated how networks of fused neurons within the gravity-sensing apparatus coordinate the beating of cilia. Cilia are hair-like appendages that ctenophores use for locomotion and orientation. The precise coordination of these cilia across different regions of the animal’s body, orchestrated by the neural circuits, enables comb jellies to maintain their stability and orientation as they navigate the water column.
"The similarities to neural circuits in other marine organisms suggest that comparable solutions to gravity sensing may have evolved independently in distant animal lineages," Jokura remarked. This observation further reinforces the idea of convergent evolution in sensory systems, where different animal groups, facing similar environmental challenges, arrive at analogous biological solutions through independent evolutionary pathways. The detailed mapping of these neural circuits provides a concrete example of how ancient, decentralized nervous systems could achieve complex behavioral control.
A Paradigm Shift in Our Understanding of Early Nervous Systems
Taken together, these interwoven studies present a compelling case for a reevaluation of the origins and early evolution of nervous systems. The research strongly suggests that early nervous systems, rather than being uniformly simple and decentralized, may have harbored a greater degree of centralization and functional sophistication than previously assumed, even in lineages that diverged very early in animal history.
According to Ferraioli, the next critical phase of research will involve a deeper exploration of the molecular underpinnings of these discoveries. Future work will aim to identify the specific molecular characteristics of the newly discovered cell types within the aboral organ. Furthermore, researchers plan to investigate the extent to which the aboral organ influences and dictates comb jelly behavior. Understanding these molecular mechanisms will provide crucial insights into the functional capabilities of these ancient sensory systems and their contribution to the evolutionary success of ctenophores.
The implications of this research are far-reaching. By revealing the unexpected complexity of a sensory organ in an ancient animal group, these studies challenge existing models of neural evolution. They suggest that the fundamental principles of sensory integration, information processing, and behavioral coordination may have emerged much earlier in the history of life than previously thought. This could necessitate a revision of evolutionary timelines and a deeper appreciation for the diverse strategies employed by early animals to interact with their environment. The aboral organ of the ctenophore, once considered a relatively simple sensory structure, now stands as a testament to the remarkable ingenuity of evolutionary processes and offers a unique window into the dawn of nervous system complexity.















Leave a Reply