Detailed three dimensional reconstructions of a key sensory structure in ctenophores reveal far greater structural and functional complexity than scientists previously recognized. The results suggest that a simple brain-like system may have existed in some of the earliest animals, offering new insight into how nervous systems evolved.
Ancient Marine Dwellers at the Forefront of Neural Discovery
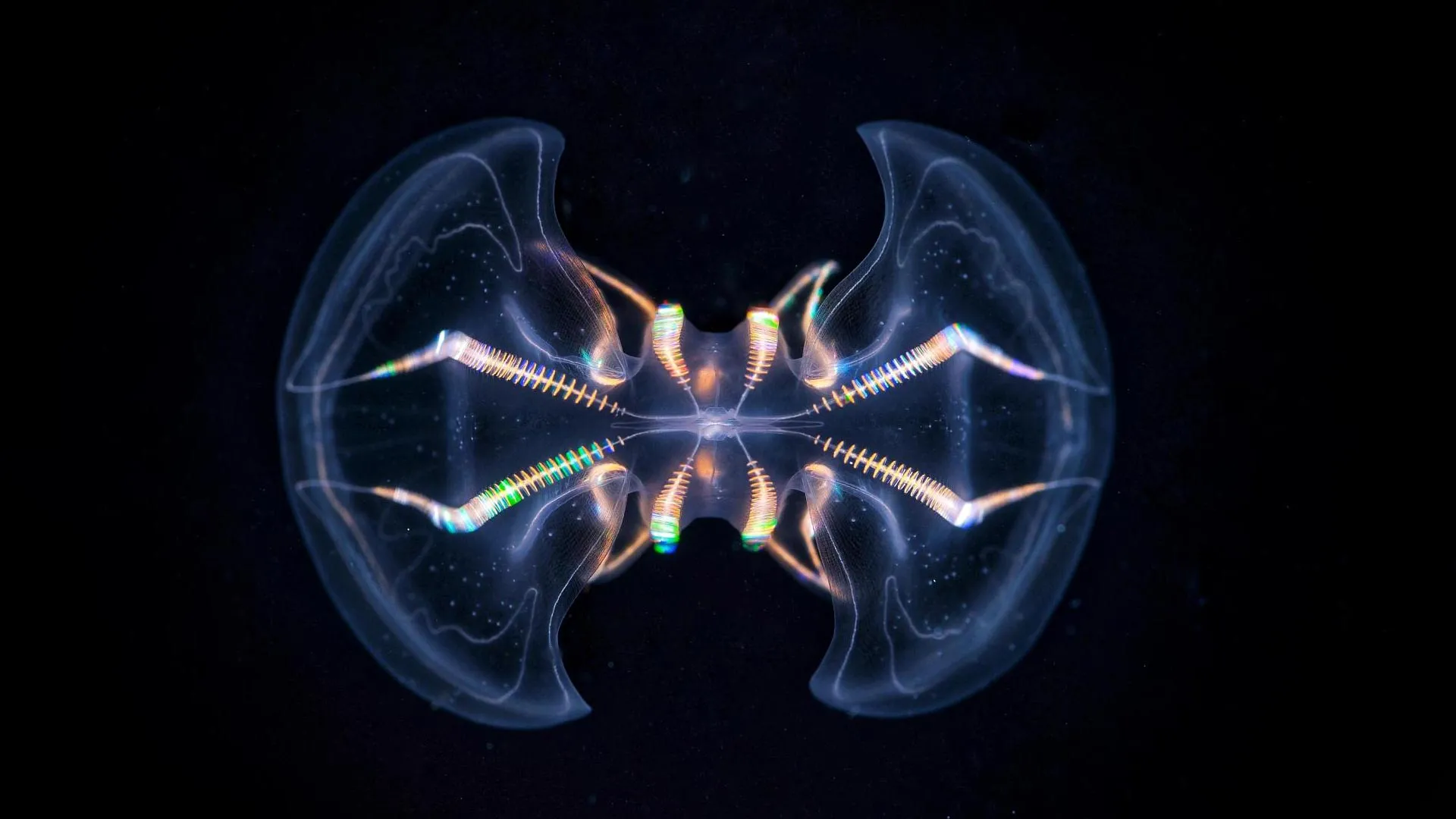
Ctenophores, often referred to as comb jellies, are ethereal, gelatinous inhabitants of Earth’s oceans, with an evolutionary lineage stretching back approximately 550 million years. These ancient creatures possess a remarkable sensory apparatus known as the aboral organ (AO). This specialized structure is instrumental in their ability to perceive crucial environmental cues such as gravity, hydrostatic pressure, and light. A groundbreaking morphological study, recently published in the esteemed journal Science Advances, has unveiled a level of complexity within the aboral organ that significantly surpasses previous scientific understanding.
"We demonstrate that the aboral organ is a sophisticated and functionally distinctive sensory system," stated Pawel Burkhardt, the group leader at the Michael Sars Centre, University of Bergen. "Our research profoundly deepens our comprehension of how behavioral coordination evolved across the animal kingdom."
Unraveling the Cellular Tapestry of an Ancient Sensory Hub
To meticulously map the internal organization of the aboral organ, researchers embarked on a collaborative effort with Maike Kittelmann at Oxford Brookes University. Their methodology leveraged cutting-edge volume electron microscopy, a powerful technique that enabled the creation of exceptionally detailed three-dimensional reconstructions of the organ’s intricate architecture.
The exhaustive analysis yielded the identification of seventeen distinct cell types residing within the aboral organ. Of particular note were eleven secretory and ciliated cell types that had remained undiscovered in prior investigations. This remarkable cellular diversity serves as compelling evidence that the aboral organ functions as a highly integrated, multimodal sensory processing center.
"The morphological diversity of the aboral organ’s cells was immediately astonishing," remarked Anna Ferraioli, a postdoctoral researcher at the Michael Sars Centre and the lead author of the study. "Working with volume electron microscopy data feels akin to uncovering exciting new insights on a daily basis. When contrasted with the apical organs of cnidarians and bilaterians, the aboral organ exhibits a striking and unique complexity."
A Hybrid System: Bridging Synaptic and Non-Synaptic Communication
Beyond its impressive cellular heterogeneity, the aboral organ exhibits a profound and intricate connection with the comb jelly’s broader nervous system. Ctenophores are characterized by a diffuse nerve net composed of fused neurons, forming a continuous network that pervades their entire bodies.
The research team discovered that this nerve net establishes direct synaptic connections with cells within the aboral organ, thereby facilitating a dynamic, two-way communication pathway. Concurrently, a significant proportion of cells within the aboral organ were observed to contain a multitude of vesicles. This observation strongly suggests that these cells are capable of releasing widespread chemical signals through a process known as volume transmission. The synergistic interplay of these mechanisms indicates that the aboral organ relies on both direct, point-to-point synaptic signaling and more diffuse, non-synaptic forms of chemical communication.
"I believe our work offers a vital perspective on the extent of knowledge we can glean from the study of morphology," Ferraioli elaborated. "While the aboral organ is certainly not analogous to the human brain, it can be accurately described as the organ that ctenophores utilize as their ‘brain’ for processing sensory information and coordinating responses."
Evolutionary Echoes: Illuminating the Genesis of Brains
The research team also delved into the expression patterns of specific developmental genes within ctenophores. Many genes that play critical roles in body organization in other animal groups are indeed present in these organisms. However, their expression patterns in ctenophores diverge significantly, hinting at alternative evolutionary trajectories.
This observed divergence suggests that the aboral organ may not be a direct homologue of the centralized brains found in other animal phyla. "In essence," Burkhardt added, "it appears that evolution has independently conceived of centralized nervous systems on multiple occasions throughout the history of life."
Connecting Neural Architecture to Behavioral Output
Further corroboration for these groundbreaking findings emerges from an independent but related study. Led by Kei Jokura at the National Institute for Basic Biology in Japan, in conjunction with Professor Gaspar Jekely from Heidelberg University, this research focused on reconstructing the complete neural wiring of the comb jelly’s gravity-sensing organ. This parallel investigation, which also involved Burkhardt, significantly bolsters the overall conclusions.
By integrating high-speed imaging techniques with detailed three-dimensional reconstructions of over 1,000 cells, the researchers elucidated how interconnected networks of fused neurons orchestrate the coordinated beating of cilia across different regions of the comb jelly’s body. This precise coordination is essential for the organism to maintain its orientation and navigate effectively within its aquatic environment.
"The parallels observed with neural circuits in other marine organisms suggest that similar evolutionary solutions for gravity sensing may have independently arisen in distinct animal lineages," Jokura commented, underscoring the convergent nature of evolutionary adaptations.
A Paradigm Shift in Understanding Nervous System Origins
Collectively, these complementary studies present a compelling case that early nervous systems may have been more centralized and sophisticated than previously hypothesized by the scientific community. According to Ferraioli, the immediate next steps for this research will involve identifying the precise molecular characteristics of the newly discovered cell types within the aboral organ. Furthermore, researchers aim to investigate the extent to which the aboral organ influences the overall behavior of comb jellies, thereby deepening our understanding of its functional significance.
Broader Implications for Evolutionary Biology
The revelations stemming from the study of ctenophore aboral organs carry significant implications for our understanding of the evolutionary trajectory of nervous systems. For decades, the prevailing view posited that cnidarians, with their simpler nerve nets, represented an earlier stage in nervous system evolution, with bilaterians developing more complex, centralized brains. The discovery of a sophisticated, potentially brain-like sensory organ in ctenophores, which diverged from other animal lineages very early on, challenges this linear progression.
This finding suggests that the evolution of centralized nervous systems may not have been a singular event but rather a recurring theme in animal evolution. Different animal groups, facing similar environmental pressures and requiring sophisticated sensory processing and behavioral coordination, may have independently evolved analogous structures. This concept of convergent evolution, where unrelated organisms develop similar traits, appears to be a potent force in shaping the diversity of life.
The identification of 17 distinct cell types, including novel secretory and ciliated cells, points to a remarkable degree of specialization within the aboral organ. This level of cellular differentiation is often associated with complex sensory processing and integration. The dual mode of communication—synaptic and volume transmission—further highlights the organ’s sophistication, allowing for both rapid, targeted responses and broader, modulatory signaling.
The comparison of gene expression patterns with other animals, revealing differences in the deployment of conserved developmental genes, underscores that evolutionary innovation often involves repurposing existing genetic toolkits. The same genes that build brains in vertebrates might be employed in novel ways to construct sensory organs in invertebrates like ctenophores.
Future Research Directions and Methodological Advancements
The path forward for research in this area is multifaceted. The identification of novel cell types necessitates further investigation into their specific functions and molecular signatures. Techniques such as single-cell RNA sequencing and immunofluorescence microscopy will be crucial in characterizing these newly discovered cellular components.
Understanding the precise neural circuits connecting the aboral organ to other parts of the ctenophore body will also be a priority. While the current studies have mapped neural connections, further functional studies are needed to decipher how information flows and is processed. Electrophysiological recordings and genetic manipulation techniques, where applicable, could provide deeper insights into the dynamic neural activity.
Moreover, comparative studies across different ctenophore species and other early-diverging animal groups (like sponges and cnidarians) will be vital in tracing the evolutionary history of sensory structures and the emergence of nervous systems. By examining the variations and commonalities in these organs, scientists can build a more robust phylogenetic framework for understanding neural evolution.
The methodological advancements that enabled these discoveries, particularly volume electron microscopy, are themselves transformative. This technology allows for an unprecedented level of detail in reconstructing biological structures, opening up new avenues of research across various fields of biology, from developmental biology to neuroscience. The ability to visualize and analyze cellular architecture in three dimensions at such high resolution is a testament to the rapid progress in scientific instrumentation.
The implications of these findings extend beyond the realm of evolutionary biology. They prompt a re-evaluation of what constitutes a "nervous system" and a "brain." The existence of a complex, integrated sensory processing center in an ancient lineage like ctenophores suggests that the evolutionary pressures driving the development of sophisticated nervous systems may have been present much earlier in animal history than previously assumed. This research contributes to a richer, more nuanced understanding of the incredible diversity and ingenuity of life’s evolutionary journey.















Leave a Reply