Eli Lilly and Company has announced groundbreaking results from a Phase 3 clinical trial for its investigational triple agonist, retatrutide, revealing an average weight loss of 16.8% in participants with type 2 diabetes. This significant outcome, coupled with superior A1C reduction, positions retatrutide as a potential game-changer in the management of this widespread metabolic condition, particularly in addressing the often-concurrent challenge of obesity. The company confirmed that retatrutide successfully met its primary and key secondary endpoints in the trial, demonstrating its efficacy over placebo at 40 weeks.
Unpacking the Clinical Data: The TRANSCEND-T2D-1 Trial Results
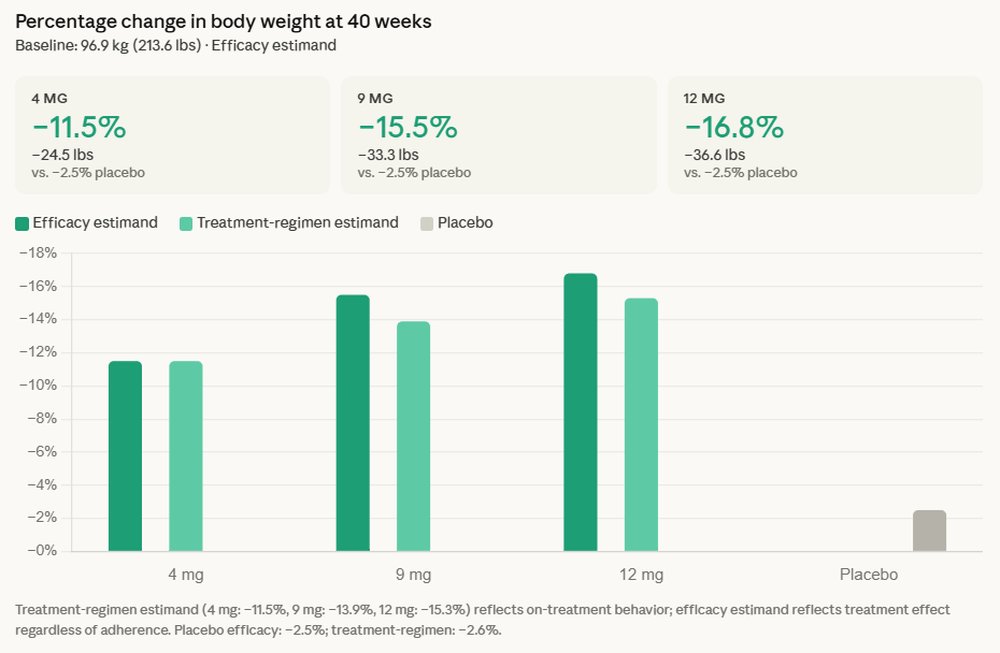
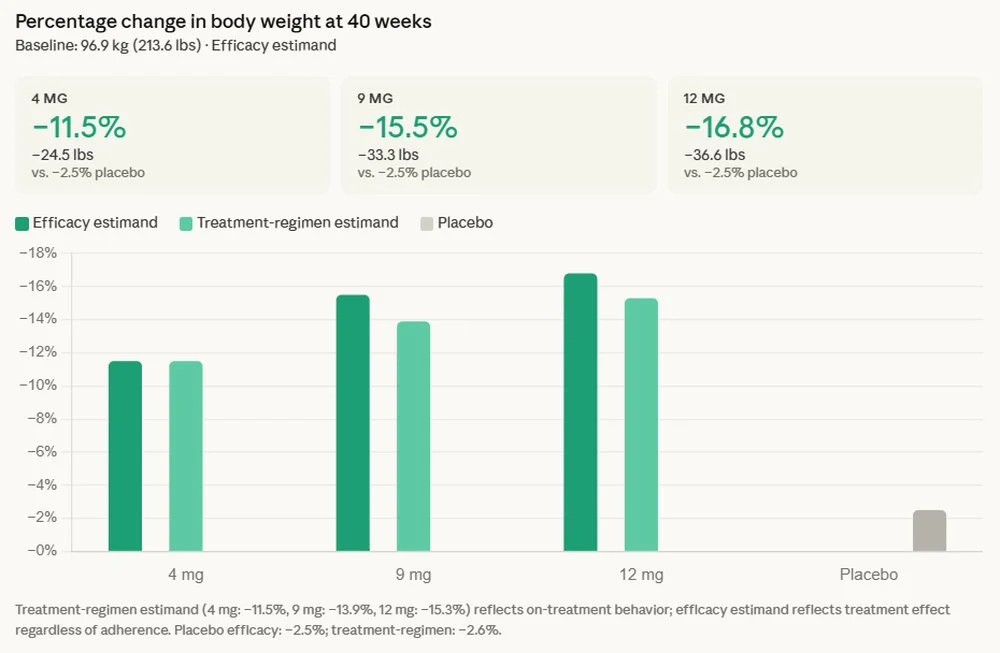
The reported data stems from the TRANSCEND-T2D-1 trial, a crucial component of Lilly’s broader clinical development program for retatrutide. Beyond the impressive 16.8% average body weight reduction, which translates to an average loss of 36.6 pounds for participants, the trial showcased an average A1C reduction of up to 2.0%. A particularly noteworthy observation was the absence of a weight-loss plateau, with participants continuing their trajectory of weight reduction throughout the 40-week study period. This suggests a sustained and potentially deeper impact on metabolic control than observed with some existing therapies.
The trial also illuminated retatrutide’s positive influence on key cardiovascular risk factors, which are often elevated in individuals with type 2 diabetes and obesity. Participants experienced clinically meaningful improvements from baseline across several parameters, including non-HDL cholesterol, triglycerides, and systolic blood pressure. These improvements are critical, as cardiovascular disease remains the leading cause of morbidity and mortality among people with type 2 diabetes. The reduction in these risk markers suggests a broader cardiometabolic benefit that extends beyond glycemic control and weight management, potentially translating into improved long-term cardiovascular outcomes for patients.
Regarding safety and tolerability, the common adverse effects reported were nausea, diarrhea, and vomiting. These are consistent with the known profiles of other incretin-based therapies, such as GLP-1 receptor agonists and dual GIP/GLP-1 receptor agonists. The management of these gastrointestinal side effects often involves careful dose titration, and their consistency with established treatments suggests a predictable safety profile within the class.
The Science Behind the Breakthrough: A Triple Agonist’s Mechanism
Retatrutide is an investigational, first-in-class GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon triple hormone receptor agonist. This unique pharmacological profile distinguishes it from existing treatments, which typically target one (like semaglutide, a GLP-1 agonist) or two (like tirzepatepatide, a GIP/GLP-1 dual agonist) of these incretin hormones.
Each of these hormones plays a distinct yet synergistic role in metabolic regulation:
- GLP-1: Primarily known for stimulating insulin secretion in a glucose-dependent manner, suppressing glucagon secretion, slowing gastric emptying, and promoting satiety in the brain. This contributes to improved glycemic control and weight loss.
- GIP: Also a glucose-dependent insulin secretagogue, GIP has been found to play a role in fat deposition and energy balance. Its synergistic action with GLP-1 is thought to enhance both glucose lowering and weight loss effects.
- Glucagon: Traditionally viewed as a hormone that raises blood glucose, recent research has highlighted that glucagon receptor agonism, when combined with GLP-1 and GIP agonism, can lead to increased energy expenditure and direct lipolytic effects (fat breakdown) in adipose tissue. This multifaceted action on metabolism is hypothesized to be the driving force behind retatrutide’s profound impact on weight loss.
By simultaneously engaging all three receptors, retatrutide aims to leverage the full spectrum of their metabolic effects, offering a more comprehensive approach to managing glucose homeostasis, energy balance, and appetite regulation. This triple action is believed to be key to achieving superior outcomes compared to agents targeting fewer pathways.
A New Horizon in Diabetes and Obesity Treatment
Type 2 diabetes affects hundreds of millions globally, and its prevalence continues to rise in tandem with the global obesity epidemic. The intricate link between obesity and insulin resistance makes effective weight management a critical, yet historically challenging, component of diabetes care. For many individuals with type 2 diabetes, achieving both robust A1C control and substantial weight loss has been an uphill battle, often requiring complex treatment regimens and significant lifestyle changes that are difficult to sustain.
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, underscored this challenge in his statement: “For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes. With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes.” His remarks highlight the unmet need that retatrutide aims to address, offering a single therapeutic agent with the potential to tackle multiple facets of cardiometabolic disease simultaneously.
The results from TRANSCEND-T2D-1 suggest a potential paradigm shift, offering a level of efficacy that could redefine treatment expectations for type 2 diabetes and its associated comorbidities. The ability to achieve significant glycemic control alongside substantial and sustained weight loss within a single medication represents a major step forward for patients and clinicians alike.
Lilly’s Expanding Footprint in Cardiometabolic Health
Eli Lilly has been a formidable player in the diabetes space for decades, and its recent advancements have solidified its position at the forefront of cardiometabolic drug development. The company’s dual GIP/GLP-1 receptor agonist, tirzepatide, marketed as Mounjaro for type 2 diabetes and Zepbound for chronic weight management, has already set new benchmarks in both glycemic control and weight loss. In trials for type 2 diabetes, tirzepatide has demonstrated A1C reductions of up to 2.5% and weight loss of around 15-16%. For obesity without diabetes, Zepbound has shown average weight loss exceeding 20% in its pivotal trials.
The emergence of retatrutide as a triple agonist, building upon the success of tirzepatide, illustrates Lilly’s strategic commitment to pushing the boundaries of incretin-based therapies. While direct head-to-head comparisons are still needed, retatrutide’s 16.8% weight loss in type 2 diabetes participants places it in a highly competitive bracket, potentially surpassing the weight loss observed with GLP-1 agonists like semaglutide (Ozempic/Wegovy, which typically achieves 5-10% weight loss in T2D and up to 15% in obesity). This sequential innovation from Lilly underscores a clear vision to develop increasingly potent and comprehensive solutions for metabolic diseases.
The competitive landscape is intense, with Novo Nordisk’s semaglutide (Ozempic for T2D, Wegovy for weight management) currently dominating market share. However, Lilly’s rapid advancements with tirzepatide and now retatrutide indicate a fierce race to develop the most effective treatments, which ultimately benefits patients by driving innovation and improving therapeutic options.
Broader Therapeutic Potential: Beyond Type 2 Diabetes
The impressive results in type 2 diabetes are just one piece of the puzzle for retatrutide. Lilly is actively exploring the drug’s potential across a wide spectrum of cardiometabolic and related conditions through an extensive Phase 3 clinical trial program. This comprehensive investigation highlights the profound systemic impact that a potent metabolic modulator like retatrutide could have.
Ongoing Phase 3 trials are evaluating retatrutide for:
- Obesity and Overweight with at least one weight-related medical problem: Given the substantial weight loss observed in the T2D trial, its efficacy in a broader obesity population is highly anticipated.
- Knee Osteoarthritis: Obesity is a major risk factor for osteoarthritis, particularly in weight-bearing joints. Weight loss could significantly alleviate symptoms and slow disease progression.
- Moderate-to-Severe Obstructive Sleep Apnea: Obesity is the primary driver of obstructive sleep apnea. Reducing body weight can dramatically improve or resolve this condition, which itself carries significant cardiovascular risks.
- Chronic Low Back Pain: Similar to osteoarthritis, excessive body weight can exacerbate chronic low back pain.
- Cardiovascular and Renal Outcomes: The observed improvements in cardiovascular risk factors suggest a potential for reducing major adverse cardiovascular events (MACE) and protecting kidney function, critical considerations for diabetes patients.
- Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD, formerly NAFLD): This condition, characterized by fat accumulation in the liver, is closely linked to obesity and insulin resistance. Weight loss is the cornerstone of MASLD treatment, making retatrutide a promising candidate.
This ambitious development program reflects a growing understanding that metabolic dysfunction is often at the root of a constellation of chronic health problems. A single agent capable of addressing this underlying dysfunction could offer a holistic treatment approach, simplifying care and improving outcomes across multiple organ systems.
Market Implications and Patient Access
The potential market implications of retatrutide are substantial. If approved, it would enter a rapidly expanding market for anti-obesity and diabetes medications, potentially commanding a significant share due to its superior efficacy. Financial analysts and industry observers will be closely watching its further development and regulatory progress, anticipating its impact on Lilly’s revenue projections and the competitive dynamics within the pharmaceutical sector. The "first-in-class" designation also grants it a unique position.
However, the success of such innovative therapies also brings questions of patient access and affordability. High-efficacy drugs often come with premium price tags, and ensuring broad insurance coverage and equitable access for all eligible patients will be a critical challenge. Patient advocacy groups will likely emphasize the need for these transformative treatments to be accessible to the millions who could benefit from them, given the immense personal and societal burden of type 2 diabetes and obesity. Policymakers and healthcare systems will need to grapple with the economic implications of widespread adoption of such potent, next-generation therapies.
Expert Reactions and Future Outlook
While formal statements from independent experts are pending full data publication, the medical community is likely to react with considerable enthusiasm to these preliminary results. Endocrinologists and cardiologists would view retatrutide as a powerful new tool in their arsenal, particularly for patients struggling with the dual burden of poorly controlled type 2 diabetes and significant obesity. The prospect of achieving an average 16.8% weight loss in a diabetes population is exceptionally high and could set a new benchmark for therapeutic efficacy.
Researchers will be keen to see the full data set, including detailed safety analyses, long-term efficacy, and specific subgroup analyses. The sustained weight loss trajectory at 40 weeks will prompt questions about even longer-term potential. Future studies will also be crucial in determining retatrutide’s cardiovascular outcomes and its exact positioning relative to existing dual agonists like tirzepatide.
The development of retatrutide underscores a significant evolution in our understanding and treatment of metabolic diseases. From single-target GLP-1 agonists to dual GIP/GLP-1 agonists, and now to a triple agonist, the incremental gains in efficacy are profound. This progression offers a beacon of hope for individuals living with these challenging conditions, promising not just symptom management but a more comprehensive approach to disease modification and potentially improved quality of life and longevity. The next steps for retatrutide will involve the completion of its expansive Phase 3 program and subsequent regulatory filings, which could usher in a new era of metabolic medicine.














Leave a Reply