Pharmaceutical R&D is at an inflection point, witnessing a profound transformation that challenges the historical dichotomy between small and large molecule therapeutics. This shift is not merely an evolution but a fundamental re-engineering of how new medicines are discovered, developed, and delivered to patients. The industry is moving from a binary choice to a complex, hybrid-driven landscape, where the strengths of diverse modalities are increasingly combined and optimized through the lens of precision medicine and advanced computational intelligence.
The traditional landscape saw small molecules, chemically synthesized compounds typically under 900 Daltons, dominate drug development. Their advantages – oral bioavailability, ease of manufacturing, ambient storage, and established regulatory pathways – made them the bedrock of pharmaceuticals for decades. Large molecules, or biologics, derived from living organisms (proteins, nucleic acids, cells), emerged later, offering unparalleled specificity and the ability to target previously "undruggable" proteins. These typically larger compounds (over 1,000 Daltons) often require injection or infusion and cold storage due to their structural complexity and fragility.
However, recent data presents a compelling paradox. While biologics sales are projected to grow three times faster than their chemical counterparts and are anticipated to overtake small molecule sales by 2027, small molecule approvals have seen a surprising resurgence. In 2025, small molecules accounted for 65% of FDA novel drug approvals, a notable jump from 56% in both 2023 and 2024. This counter-intuitive trend underscores the underlying forces reshaping R&D: technological upgrades, innovative delivery systems, and a data-driven approach that prioritizes target biology over modality dogma.
The Evolving Market Landscape: A Decade of Transformation
The global pharmaceutical market has experienced significant growth and a gradual shift in modality distribution. In 2018, the market was estimated at $828 billion, with small molecules representing 69% of sales and biologics 31%. By 2023, the market had expanded to $1.344 trillion, with biologics capturing a larger share at 42%, while small molecules held 58%. Projections suggest this trend will continue, with many analysts forecasting biologics to surpass small molecule sales within the next few years, potentially as early as 2027. This growth in biologics is fueled by innovation across various categories, including monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), mRNA therapies, and advanced cell and gene therapies like CAR-T.
Despite the rapid growth in biologic sales, the enduring strength of small molecules in FDA approvals highlights their continued relevance and adaptability. The global small molecule drug discovery market, valued at $61.9 billion in 2025, is projected to reach $103 billion by 2031, demonstrating sustained investment and innovation. Similarly, the global biologics market, which stood at $349 billion in 2023, is expected to surge to $1.077 trillion by 2035. This robust growth in both segments confirms that the future of drug discovery is not about one modality replacing another, but rather their synergistic evolution.
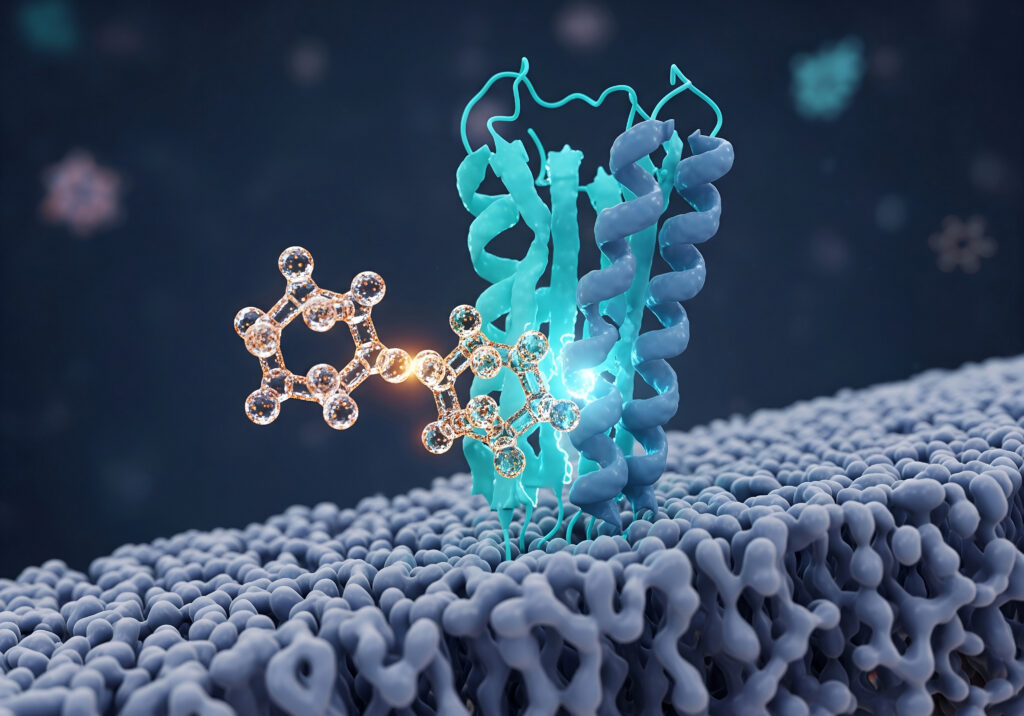
The Rise of Precision Medicine and AI-Driven Discovery
A critical driver of this paradigm shift is the move from broad-spectrum therapies to precision medicine. Advances in genomics, target biology, and artificial intelligence (AI) are empowering researchers to identify and attack diseases with unprecedented specificity. As Andreas Matern, vice president of professional services at Elsevier, aptly puts it, "It’s no longer trying to shotgun blast a whole bunch of things, but instead trying to sniper and be really tight in what we’re attacking."
This strategic shift means pharmaceutical companies are now employing extensive biological intelligence before committing to a specific therapeutic modality. The focus is on a deep understanding of the target’s role in disease, the genetic profile of the patient population, and the optimal mechanism of action, rather than starting with a predetermined drug type. This upfront investment in target validation and biological insight is crucial for increasing success rates in clinical development.
AI and machine learning (ML) are acting as powerful accelerators in this new era. Tools like AlphaFold are revolutionizing protein structure prediction, enabling more accurate target identification and rational drug design. Generative chemistry models are rapidly synthesizing novel molecular structures, vastly expanding the chemical space explored. Large language models (LLMs) are being layered on top of proprietary and public datasets, extracting new insights from vast amounts of scientific literature, clinical trial data, and real-world evidence. This computational prowess is compressing timelines, generating novel hypotheses, and, crucially, informing modality selection with greater precision than ever before.
Daphne Koller, CEO and founder of Insitro, emphasized this at the Danaher Summit: "Most programs fail in the clinic because of the early stages of the decision-making process. The focus of the next few years needs to be on increasing the success rate (the yield), rather than just efficiency. AI must help us select the right therapeutic hypotheses and the right patient populations from the start." This highlights a fundamental change: AI isn’t just about faster experimentation, but smarter decision-making at the earliest, most critical stages of R&D.
Defining the Modalities: A Deeper Dive
To understand the convergence, it’s essential to delineate the characteristics and evolving capabilities of each modality.
Small Molecules: A Renaissance Underway
Traditionally, small molecules were valued for their ability to penetrate cell membranes, accessing intracellular targets. Their oral bioavailability, stability, and cost-effective, scalable manufacturing made them the preferred choice for many conditions. For years, the narrative suggested biologics would gradually displace them. However, a "technological upgrade," as described by Murray Aitken of IQVIA, has led to a small molecule renaissance.
Key innovations driving this resurgence include:
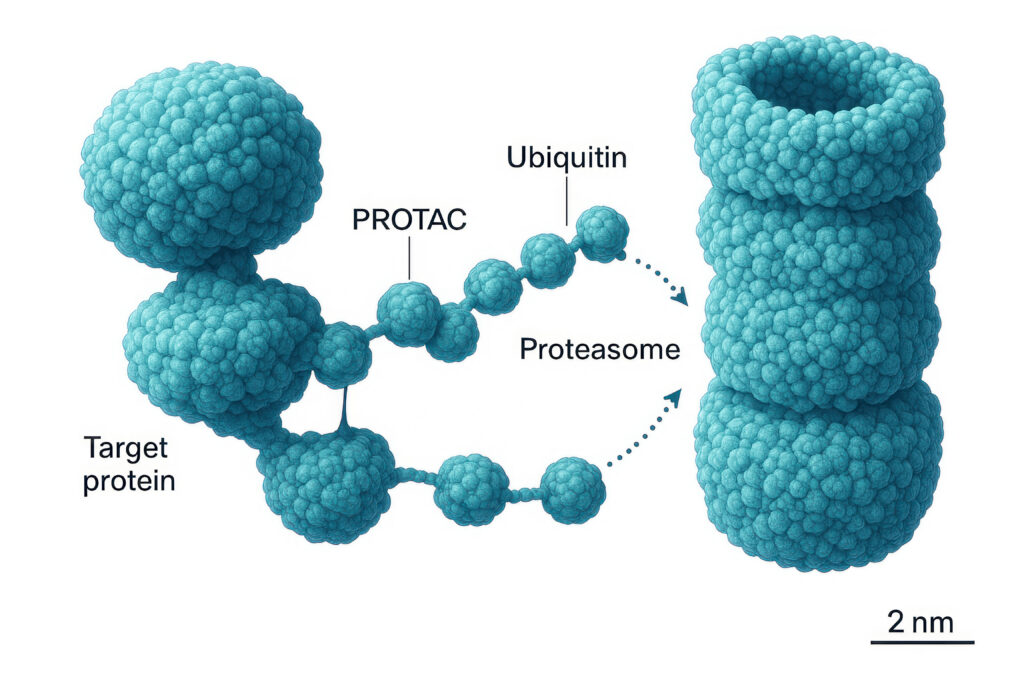
- Targeted Protein Degradation (TPD): This groundbreaking approach, exemplified by proteolysis-targeting chimeras (PROTACs) and molecular glues, hijacks the cell’s natural protein disposal machinery (ubiquitin-proteasome system) to eliminate disease-causing proteins rather than merely inhibiting their function. TPD dramatically expands the "druggable proteome," making previously inaccessible targets (e.g., those lacking conventional binding pockets or being too large/flexible) amenable to small molecule intervention. Nello Mainolfi, CEO of Kymera Therapeutics, noted, "Targeted protein degradation allows us to reach broader patient populations compared to injectable biologics. We are developing oral drugs with biologics-like activity, reaching critical signaling nodes that have been historically ‘undruggable’ with traditional inhibitors."
- RNA-Targeting Small Molecules: An emerging frontier involves small molecules designed to bind and modulate RNA directly, upstream of protein expression. Historically, RNA was considered undruggable by small molecules. However, advances in structural biology and AI are enabling the design of molecules that can target transcription factors, splice sites, and other RNA structures, opening up entirely new therapeutic avenues inaccessible to traditional small molecules and most biologics. A significant commercial validation of this approach came last year with Merck Group’s $2 billion partnership with Skyhawk Therapeutics for RNA-targeting small molecules in neurological disorders.
- Oral Delivery Innovation: One of the most commercially significant trends is the push to make complex molecules, including peptides and other structures historically requiring injection, orally available. The GLP-1 story, with semaglutide evolving from an injectable peptide to an oral formulation and next-generation oral small molecule GLP-1 agonists in development, is a prime example. This innovation promises to combine the targeting precision of biologics with the convenience and scalability of small molecules, expanding access globally by eliminating cold chain requirements and injection barriers.
Large Molecules: Expanding the Therapeutic Frontier
Biologics are undergoing a full-scale expansion, with pipelines becoming broader, more diverse, and technically ambitious. What began with monoclonal antibodies in the 1980s has blossomed into a rich ecosystem of modalities, each with unique mechanisms and target classes.
Key large molecule innovations include:
- Monoclonal Antibodies (mAbs) and Bispecific Antibodies: mAbs remain the backbone, increasingly engineered for improved half-life, reduced immunogenicity, and enhanced effector functions. Bispecific antibodies, designed to engage two targets simultaneously (e.g., redirecting T cells to tumor cells), represent a powerful evolution, enabling novel therapeutic mechanisms, particularly in oncology.
- Antibody-Drug Conjugates (ADCs): These hybrid molecules combine the precise targeting of an antibody with the cytotoxic potency of a small molecule payload. ADCs deliver chemotherapy directly to cancer cells, minimizing systemic toxicity and revolutionizing cancer treatment. They have seen a wave of approvals and late-stage pipeline activity, with over 100 ADCs in clinical development globally.
- mRNA Therapies: Rapidly validated by COVID-19 vaccines, mRNA platforms are now being diversified into oncology vaccines, rare disease treatments, and other infectious disease applications. These therapies instruct the body’s own cells to produce therapeutic proteins.
- RNA Interference (RNAi) and Antisense Oligonucleotides (ASOs): Modalities like siRNA and ASOs silence disease-causing genes at the mRNA level, offering a gene-specific approach. They have gained regulatory traction and approvals in rare diseases and cardiovascular indications.
- Cell and Gene Therapies (e.g., CAR-T, CRISPR): CAR-T cell therapies involve engineering a patient’s own T cells to recognize and destroy cancer cells, transforming the treatment of hematologic malignancies. CRISPR and other gene editing technologies, with the first approved CRISPR therapy (Casgevy) reaching patients in 2023, promise to correct genetic defects at their source, offering curative potential for a range of genetic disorders.
Oncology remains the single largest driver of biologic R&D investment, with ambitious large molecule science deployed across ADCs, immuno-oncology (checkpoint inhibitors, CAR-T, bispecifics), expanding from hematologic cancers into solid tumors.
The GLP-1 Story: A Template for Modality Evolution
The success of GLP-1 receptor agonists provides a compelling case study in the commercial and scientific power of large molecule R&D, and crucially, how it can drive small molecule innovation. Drugs like semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) have become among the fastest-selling pharmaceuticals in history, validating peptide engineering on an unprecedented scale for metabolic disorders and obesity.
Andreas Matern explains, "GLP-1 is really showing how that balance is evolving. They are a peptide-based modality – technically in the large molecule category – but the commercial success is now driving innovation across small molecules: oral delivery and combination therapy." This illustrates a pattern: therapies often start as injectables due to the technical simplicity of delivering complex molecules. However, once the biological validation is established and commercial demand is clear, there’s a strong incentive to invest in more convenient formats, including oral small molecules. This arc – from injectable biologic to oral small molecule agonist – is likely to become a template for future modality evolution across various therapeutic areas, bridging the perceived gap between the two classes.
Investment Landscape and Strategic Imperatives
The dynamic interplay between small and large molecules, coupled with the rise of hybrid modalities and AI, profoundly impacts the investment landscape and strategic goals of pharmaceutical companies.
- Large Pharma: For established pharmaceutical giants, the imperative is portfolio diversification across modalities. They leverage their extensive infrastructure and financial capacity to absorb risk across therapeutic areas and scale manufacturing for diverse drug types. Strategic acquisition or licensing of platform biotechs becomes crucial for accessing novel modalities without the time and cost of building from scratch. This strategy mitigates risk and rapidly integrates cutting-edge innovation.
- Biotech Companies: Biotechs typically adopt a platform-driven approach, specializing in a specific modality or technology (e.g., a particular gene editing platform, a novel ADC linker chemistry, or an RNA-targeting approach). While this carries higher risk concentration, it allows for faster, more focused innovation. The most fundable and partnerable biotechs are those that combine a truly novel modality with a validated target and a clear path to convenient delivery, especially oral, to maximize market potential.
- Investors: Smart investors are keenly observing advancements in delivery technology, recognizing that solving the "last-mile problem" – safely and conveniently getting complex molecules to their targets – is key to unlocking broader market access and patient benefit. This includes advancements in lipid nanoparticles for RNA therapies, oral peptide technologies, and innovative ADC linker chemistry.
The Indispensable Role of Data in Modern R&D
For researchers and R&D teams, the data foundation is no longer merely supportive; it is the new lab bench. Investing in robust data infrastructure from day one is paramount. This includes electronic laboratory notebooks (ELNs) for structured data capture, ontologies and knowledge graphs for organizing complex biological and chemical information, and adherence to FAIR (Findable, Accessible, Interoperable, Reusable) data principles. High-quality, well-governed, and integrated data is the fuel for AI-powered drug discovery, enabling predictive modeling, hypothesis generation, and informed decision-making.
The shift from trial-and-error experimentation to data-driven prediction represents the most consequential change in drug discovery in a generation. Organizations that embrace data quality, governance, and integration will be best positioned to leverage AI effectively, translating scientific insights into successful therapies.
Conclusion: A Spectrum, Not a Competition
The binary view of small versus large molecules is increasingly a false dichotomy. As the gray area between these modalities continues to expand with hybrid approaches like ADCs, PROTACs, and RNA-small molecule conjugates, the modality question transforms into a spectrum of choices, each tailored to specific biological challenges.
Andreas Matern summarizes this collaborative future: "I don’t see small and large molecules competing for dominance. I think there’s going to be a continued balance… small molecules will remain at the foundation because they’re scalable, they’re orally delivered, they’re stable. And biologics will probably be where we continue to expand, where things like precision and durability matter."
Ultimately, scientific insight alone is not enough. The enduring challenge of drug discovery remains getting the right drug to the right target in the right patient. This necessitates ongoing innovation in delivery technologies – from lipid nanoparticles to oral peptide formulations and advanced formulation science. The expansion of the therapeutic modality toolkit is unequivocally good news for patients, science, and the pharmaceutical industry alike. More tools mean more addressable targets, a wider range of treatable diseases, and broader patient access. In this evolving landscape, the true competition is not between modalities themselves, but between organizations that embrace this complexity and integration versus those that fail to adapt to this new, hybrid reality of pharmaceutical innovation.














Leave a Reply