In a significant development for AI-driven drug discovery, Terray Therapeutics, a Los Angeles-area startup, has introduced TerraBind, a novel potency prediction model that promises to redefine the landscape of small-molecule binding affinity predictions. The company asserts that TerraBind bypasses a fundamental bottleneck inherent in diffusion-based co-folding models, including the widely recognized Boltz-2, leading to dramatically accelerated inference, improved accuracy, and a critical feature previously absent in comparable models: built-in uncertainty quantification. This launch positions TerraBind as a formidable challenger to established AI methodologies, potentially transforming the efficiency and success rates of early-stage drug development.
The Precedent: Boltz-2 and the Rise of AI in Drug Discovery
The field of AI in drug discovery witnessed a major milestone in June of last year with the release of Boltz-2 by MIT and Recursion Pharmaceuticals. Boltz-2 captivated the scientific community as the first open-source artificial intelligence model capable of jointly predicting protein structure and small-molecule binding affinity. Its predecessor, Boltz-1, had already achieved widespread adoption, and Boltz-2 built upon this momentum by offering accuracy comparable to physics-based free-energy perturbation (FEP) methods—the gold standard for rigorous binding affinity calculations—but at an astonishing speed increase of approximately 1,000-fold. This leap in computational efficiency allowed researchers to screen molecules far more rapidly than traditional simulation techniques, pushing the boundaries of virtual screening and lead optimization.
Boltz-2’s architecture, like many contemporary structure-based potency prediction models from institutions and companies such as Isomorphic Labs, relies on a multi-step process. This typically involves encoding the molecule and protein, followed by a crucial diffusion step that generates a detailed, all-atom 3D pose, illustrating precisely how the small molecule interacts with the protein’s binding site. The potency prediction is then derived from this generated pose. While powerful, this diffusion-based pose generation step has been identified by Terray as a significant computational bottleneck.
TerraBind’s Paradigm Shift: Eliminating the Diffusion Bottleneck
Terray Therapeutics argues that this diffusion-based coordinate generation, despite its prevalence, is the primary impediment to scaling these models for practical use in real-world drug discovery programs. Dr. Jacob Berlin, co-founder and CEO of Terray, likens this approach to "trying to decide the income of people by first looking at pictures of how they dress, instead of directly trying to predict the income." His contention is that the ultimate goal in drug discovery is to understand "how well the molecule works," rather than meticulously detailing its precise atomic pose, especially if that detailed generation consumes the vast majority of computational resources.
TerraBind’s core architectural innovation lies in its complete elimination of this diffusion-based pose generation step. This step, according to Terray’s internal analyses, accounts for approximately 96% of the total computational cost in models like Boltz-2. By removing this computationally intensive component, TerraBind dramatically reduces inference time without, Terray claims, compromising accuracy.
The model achieves this through a sophisticated pipeline that leverages Terray’s proprietary chemical foundation model, COATI (which has its code publicly available on GitHub). COATI is designed to encode molecules and proteins into a shared, high-dimensional representation space. This abstract representation is then directly fed into a Pairformer module. Unlike diffusion models that iteratively denoise a noisy initial state to generate a precise 3D structure, the Pairformer module in TerraBind directly resolves structural features and predicts potency. Crucially, TerraBind-Struct operates at a coarse-grained level, focusing on protein Cβ atoms and ligand heavy atoms only, thereby circumventing the need for iterative denoising and full all-atom coordinate generation.
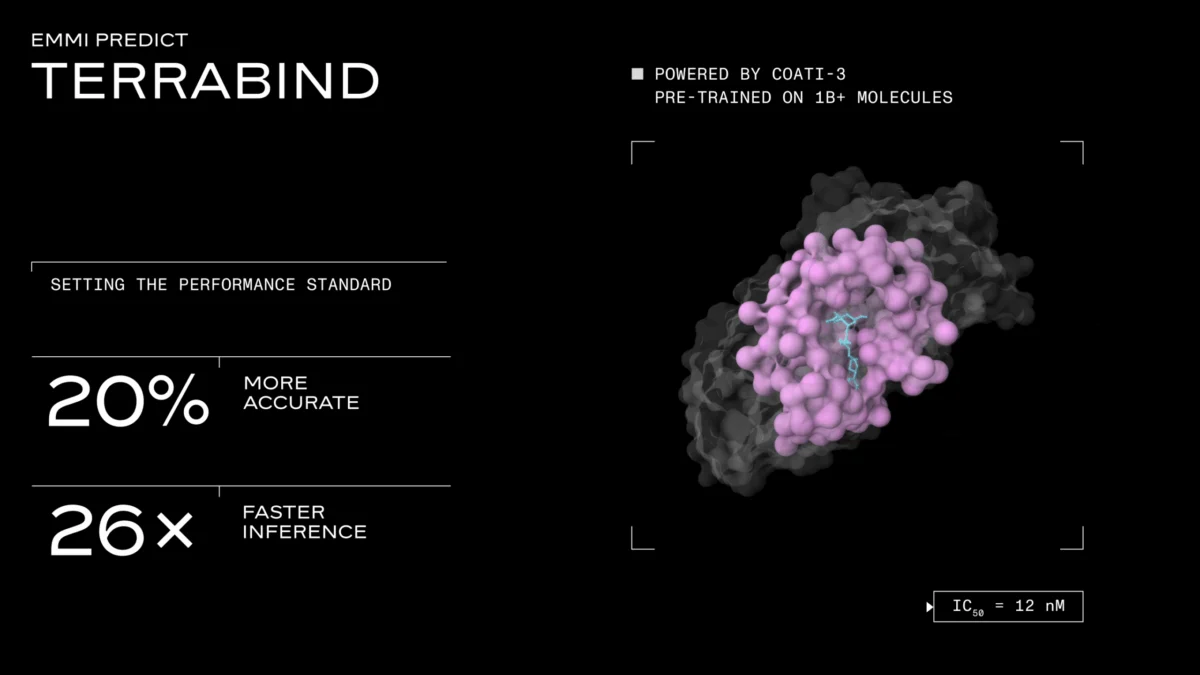
This architectural reimagining translates into staggering performance gains. In end-to-end benchmarks conducted on a single A6000 GPU (using 196 tokens and 10 samples per complex), TerraBind reduced inference time from 27.8 seconds per complex for Boltz-2 to a mere 1.04 seconds. This represents a 26-fold increase in speed, allowing for vastly more rapid evaluation of potential drug candidates.
Unprecedented Speed and Scalability for Drug Discovery
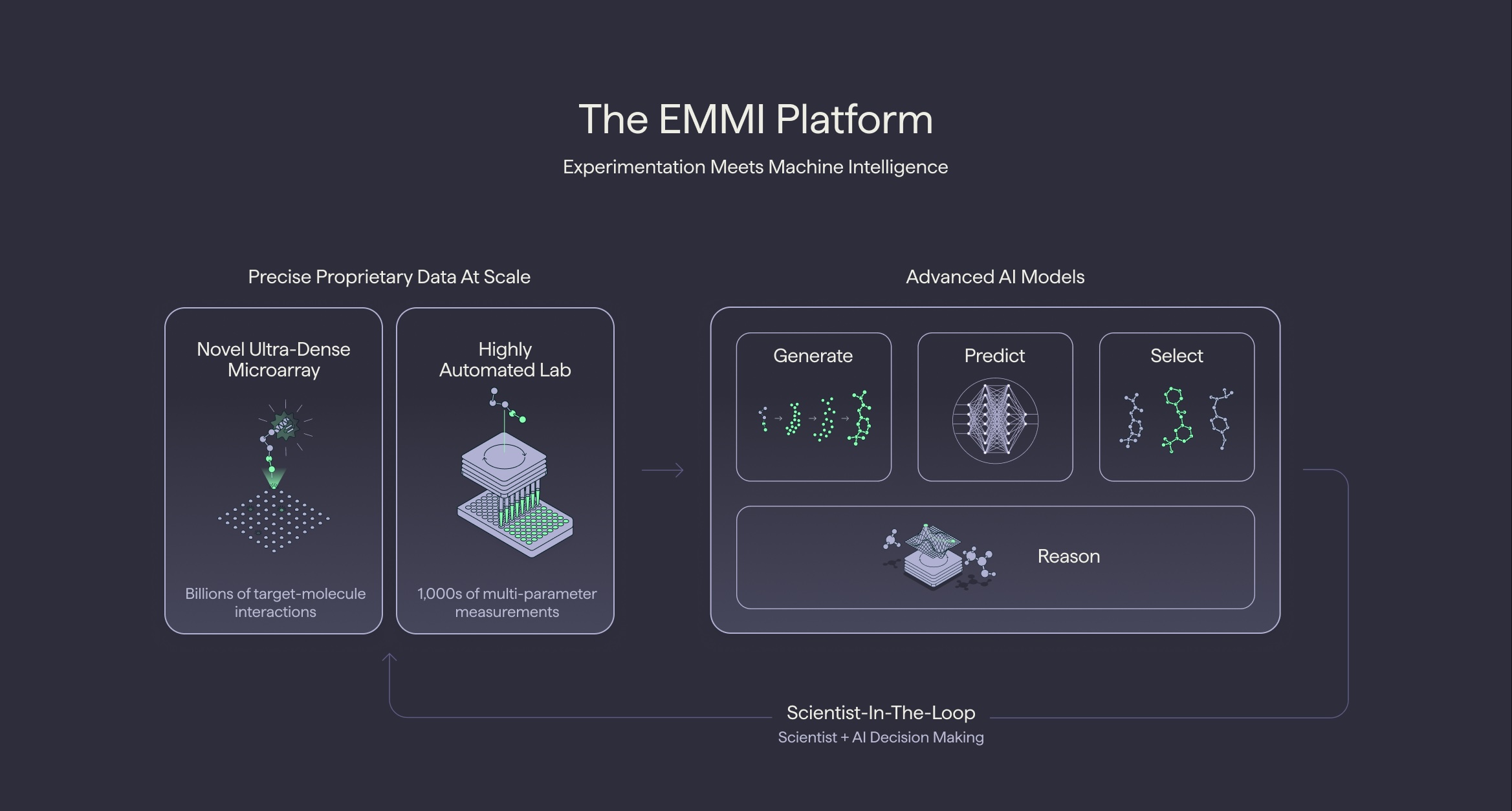
The practical implications of this speed advantage for drug discovery are profound. Terray has integrated TerraBind into a two-tiered screening pipeline within its EMMI (Experimental and Machine Learning for Molecular Innovation) platform. The initial tier employs an ultra-fast sequence-only model, TerraBind-Seq, which is an astounding 20,000 times cheaper than public alternatives. This initial screening quickly sifts through vast libraries of molecules, yielding a manageable subset of promising candidates. This subset then undergoes a more rigorous, structure-based evaluation using TerraBind-Struct. Even at this second tier, TerraBind-Struct is still 20 times cheaper and faster than Boltz-2.
The net effect of this tiered approach is the ability to perform million-scale virtual screening, a significant departure from the industry standard, which has typically been limited to thousands of molecules due to computational constraints. This exponential increase in screening capacity allows drug developers to explore a much broader chemical space, significantly increasing the likelihood of discovering novel and potent therapeutic compounds. Terray’s EMMI platform further streamlines this process by integrating proprietary experimental hardware, including an ultra-dense microarray capable of generating billions of target-molecule interactions, with AI models that generate, predict, and select molecules in a scientist-in-the-loop workflow.
Accuracy and the Unexpected Benefits of Architectural Simplicity
A natural concern with stripping out a major architectural component like diffusion-based pose generation is a potential compromise in predictive accuracy. However, Terray’s internal benchmarks suggest the opposite. Dr. Berlin stated, "You would worry that you’re trading speed and scale for quality, but we don’t. The quality is best in class: 20% better than Boltz, roughly on par with what Isomorphic put out, but it does 26 times more and quantifies the uncertainties."
TerraBind demonstrated a 16-20% improvement in Pearson correlation relative to Boltz-2 on two independent held-out test sets: the public CASP16 binding affinity challenge and a proprietary dataset comprising over 25,000 assay readouts spanning 18 distinct targets. On the proprietary dataset, TerraBind outperformed Boltz-2 on 15 out of 18 targets. Terray attributes these unexpected accuracy gains to the architectural change itself. By removing the diffusion head, the Pairformer module is "forced" to fully resolve structural features and directly learn the relationship between input representations and potency, rather than offloading that complex work to a downstream generative step. This direct learning path appears to lead to a more robust and accurate potency prediction.
While TerraBind produces coarse-grained geometry rather than the high-resolution, all-atom poses generated by diffusion models, Terray argues that this level of structural detail is entirely sufficient for reliable pose prediction and affinity ranking. The emphasis shifts from generating visually appealing, publication-quality molecular snapshots to providing actionable, accurate potency predictions. It’s important to note that while the CASP16 results are publicly verifiable, the stronger accuracy claims derived from Terray’s proprietary dataset are not currently available for independent verification, a common limitation in competitive pharmaceutical R&D.
The Game-Changing Power of Uncertainty Quantification
Beyond speed and accuracy, Terray emphasizes a feature that Dr. Berlin believes is paramount for practical drug discovery: uncertainty quantification (UQ). Most existing AI models provide a point prediction for potency but offer no insight into the model’s confidence in that prediction. In a field where experimental validation is costly and time-consuming, this lack of confidence estimation can lead to inefficient resource allocation.
The standard practice in drug discovery involves a "greedy" selection process, where researchers simply choose the top-ranked molecules for synthesis and testing. However, as Terray highlights in its EMMI platform documentation, model uncertainty is often correlated. If a model overvalues a specific structural motif, and the top-ranked molecules share this motif, a greedy selection could result in an entire batch of compounds either succeeding or failing together. While enforcing structural diversity can mitigate some risks, it doesn’t account for shared uncertainties among structurally distinct molecules.
TerraBind addresses this critical gap with TerraBind-Epinet, a variant that incorporates epistemic neural networks to estimate the model’s uncertainty for every prediction. This uncertainty information is then fed into an acquisition function called Expected Maximum (EMAX). EMAX allows Terray to build "hedged baskets" of molecules for synthesis, strategically balancing potential potency with predictive confidence.
Dr. Berlin explained the rationale: "The math will tell you: if this molecule is what I think it is, this other molecule is unlikely to be what I think it is, and vice versa. So you can build hedged baskets of molecules where if number one works, number ten won’t, which is fine, you got a winner, but if number two doesn’t, number nine probably will. You get rid of the ‘whole set’ type outcome that’s very common in drug discovery, where you make a bunch of molecules and you don’t move your program far enough along." Terray’s internal benchmarking demonstrates that this EMAX-driven approach yields a 6-fold improvement in selection performance over traditional greedy methods, translating to approximately 3-fold savings in time and cost in retrospective studies. This ability to make more informed, risk-aware decisions about which molecules to pursue could significantly de-risk early-stage drug development.
Navigating the Path to Clinical Impact
While TerraBind’s technical achievements are impressive, Dr. Berlin remains pragmatic about the journey from model advantage to actual drug development outcomes. He cautions against the common misconception that winning benchmarks automatically solves the problem, emphasizing the necessity of deeply embedding these AI capabilities into internal workflows and connecting them across the entire drug discovery pipeline.
Terray Therapeutics, founded in 2019, has an internal pipeline focused on immunology, but its programs have not yet reached the clinical stage. Therefore, the ultimate real-world impact of the EMMI platform and TerraBind remains preclinical. However, the company has reported significant milestones through its partnerships with major pharmaceutical entities like Bristol Myers Squibb, Gilead, and Calico, indicating confidence from industry leaders in Terray’s technological approach.
The Strategic "Data Moat" and Workflow Integration
Terray acknowledges that its architectural innovations, once publicized, may be rapidly adopted by competitors. Dr. Berlin anticipates that "should everybody have an awakening upon seeing our stuff that our architectural choices are vastly preferred, I expect people will work on it… I think it’ll probably sweep through many of the models very quickly." However, he firmly believes that Terray’s durable competitive advantage rests on two crucial pillars: proprietary data and deep workflow integration.
Firstly, Terray claims to possess the largest global database of small-molecule binding data, currently exceeding 14 billion unique target-molecule interactions and growing at an astonishing rate of approximately 1 billion interactions per quarter. This colossal dataset is generated through the company’s proprietary ultra-dense microarray hardware, a sophisticated experimental platform capable of measuring interactions between 32 million molecules and targets of interest within minutes. This technology allows Terray to generate high-quality measurements in vast, unexplored regions of chemical space where no public datasets currently exist. This proprietary data edge is critical, as, in Berlin’s words, "Almost everybody in the AI space except us has focused their work in areas of known chemical space, because they don’t have proprietary hardware and data at the scale we do." This enables Terray to discover genuinely novel chemical entities rather than simply optimizing existing ones, addressing a common criticism that "many current ‘AI drugs’ are strikingly similar to previous patents."
Secondly, Terray’s strength lies in its comprehensive workflow integration. The COATI chemical foundation model, which powers TerraBind’s speed, has been in continuous development for years and has been trained on over a billion molecules. It is not merely a prediction engine but underpins Terray’s entire generate-predict-select cycle. Scientists at Terray are reportedly transitioning to interacting with the EMMI platform through an AI agent, allowing them to specify a desired outcome and let the AI plan the optimal workflow, rather than manually sequencing individual models. This level of seamless integration maximizes the impact of each technological component.
Investment, Collaboration, and the Long View of AI in Drug Discovery
Terray Therapeutics has garnered significant financial backing, raising over $200 million. Its Series B funding round was co-led by prominent investors Nvidia and Bedford Ridge, underscoring the confidence placed in its innovative approach. Nvidia, in particular, is not only an investor but also a technology development partner, and the TerraBind announcement is being co-promoted with the graphics processing unit (GPU) giant, highlighting the synergy between Terray’s computational demands and Nvidia’s advanced hardware.
Founded in 2019, predating both the COVID-19 pandemic and the widespread ChatGPT-fueled AI wave, Terray has witnessed the broader industry gradually converge on the foundational thesis it was already building around. Dr. Berlin offers a philosophical perspective on the ongoing debate between AI hype and reality in drug discovery, seeing it as an inherent aspect of the industry’s extended timeline. He envisions a future where the current skepticism will give way to clear evidence of transformative change: "In 15 years, when you look back, it’ll be like, ‘Whoa, the drug discovery process changed, the success rate changed, the time to get through all this changed.’ But as you go through it, right up against the frontier, you’ll have people arguing all the way through because one drug will fail, two will succeed, and you’ll have the whole cycle again."
TerraBind represents a bold step forward in the quest to harness AI for more efficient and successful drug discovery. By challenging conventional architectural approaches and prioritizing practical utility through speed, accuracy, and uncertainty quantification, Terray Therapeutics is charting a course that could significantly accelerate the delivery of new therapies to patients, moving beyond theoretical benchmarks to tangible impact within the demanding realities of pharmaceutical research and development.














Leave a Reply