The accumulation of tau protein within the neural architecture has long been recognized as a definitive pathological hallmark of Alzheimer’s disease, yet the precise mechanisms governing its buildup and potential clearance have remained partially obscured. In a landmark study published on March 5 in the journal Cell Reports, a publication of Cell Press, an international team of researchers has unveiled a previously unknown biological pathway that could fundamentally alter the scientific understanding of neurodegenerative progression. The study identifies tanycytes—specialized ependymal cells located in the brain’s third ventricle—as vital agents in the transport and elimination of toxic tau proteins. By combining sophisticated animal modeling, cellular assays, and comparative analyses of human brain tissue, the research suggests that the failure of these cells to function as a "shuttle" between the brain and the peripheral circulatory system may be a primary driver of the protein stagnation observed in dementia patients.
The Role of Tau Protein in Neurodegeneration
To understand the significance of this discovery, it is essential to contextualize the role of tau in the healthy and diseased brain. In a normal physiological state, tau proteins are responsible for stabilizing microtubules, the structural scaffolding within neurons that facilitates the transport of nutrients and essential molecules. However, in Alzheimer’s disease and other "tauopathies," these proteins undergo chemical changes that cause them to detach from microtubules and stick to one another, forming insoluble "neurofibrillary tangles." These tangles disrupt neuronal communication and eventually lead to cell death.
For decades, the "amyloid cascade hypothesis"—which posits that the buildup of amyloid-beta plaques is the primary driver of Alzheimer’s—dominated the research landscape. However, the consistent failure of amyloid-targeting drugs in clinical trials has shifted the scientific focus toward tau. Research increasingly indicates that the spread of tau tangles correlates much more closely with cognitive decline and brain atrophy than amyloid plaques do. Consequently, understanding how the brain naturally clears "waste" tau has become a top priority for neuroscientists globally.
Tanycytes: The Brain’s Specialized Regulators
Tanycytes, the focal point of the new study, are a unique population of non-neuronal cells found predominantly in the hypothalamus, lining the floor of the third ventricle. Unlike typical glial cells, tanycytes possess long processes that extend into the hypothalamic parenchyma and contact blood vessels. Historically, these cells were primarily studied for their role in metabolic regulation; they are known to sense nutritional signals in the blood and transport hormones, such as leptin, into the brain to regulate appetite and energy expenditure.
The research led by Vincent Prevot, a director of research at INSERM (the French National Institute of Health and Medical Research), reveals that tanycytes possess a dual functionality. Beyond their metabolic duties, they appear to serve as a critical drainage system. "Our findings reveal a previously underappreciated, disease-relevant role for tanycytes in neurodegeneration," Prevot stated. He emphasized that these cells act as a bridge between the cerebrospinal fluid (CSF), which bathes the brain, and the bloodstream, allowing the brain to export toxic metabolic byproducts.
Chronology of the Research and Experimental Design
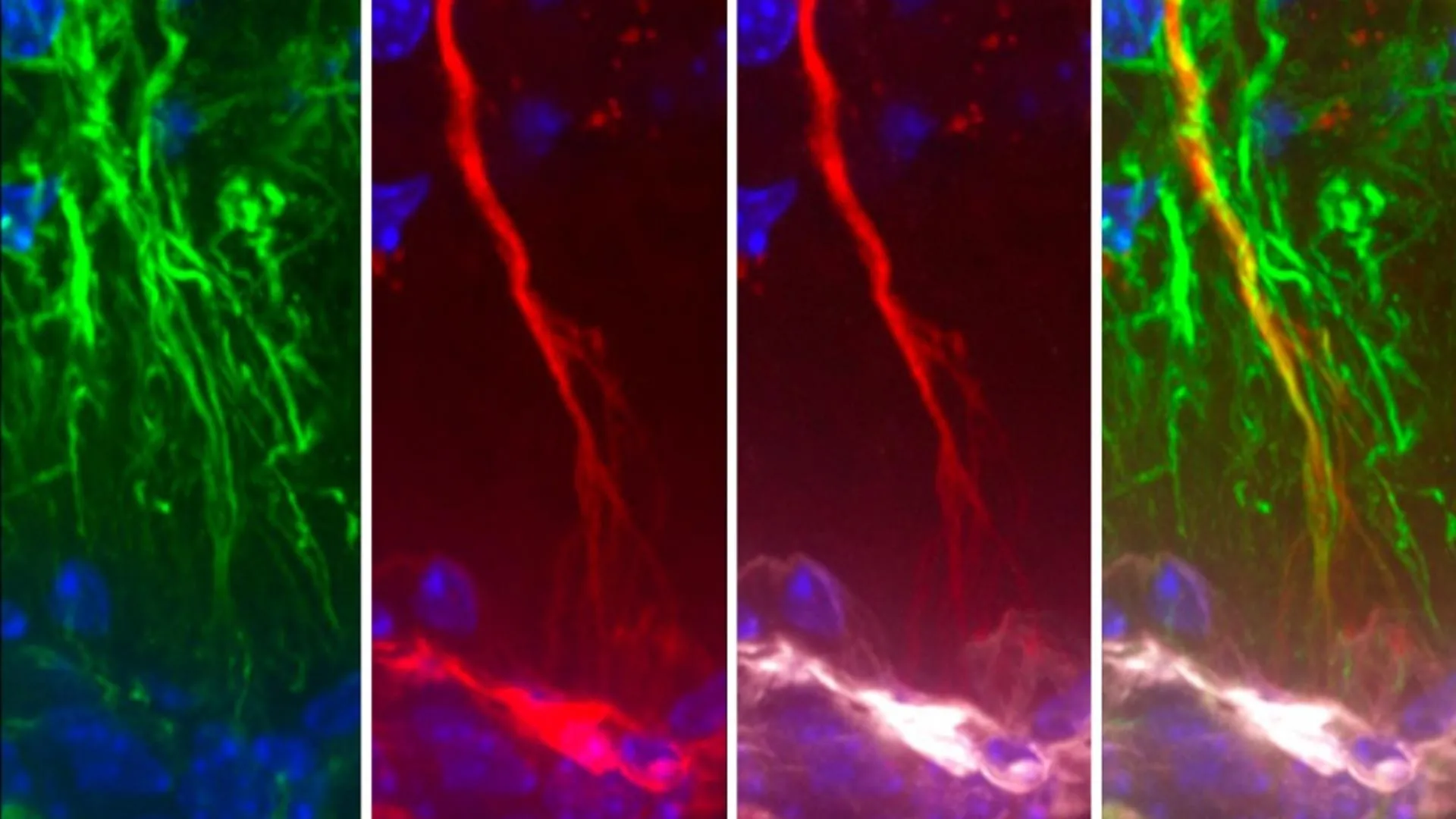
The investigation proceeded through several distinct phases, beginning with the observation of how tau moves within the central nervous system. The research team initially utilized rodent models to track the movement of fluorescently labeled tau proteins. By injecting these proteins into the CSF of mice, the scientists were able to observe the molecules migrating toward the third ventricle, where they were actively taken up by tanycytes.
Following the animal experiments, the team moved to cellular models to isolate the biochemical mechanisms at play. They discovered that tanycytes utilize specific receptors to "grab" tau proteins from the CSF and transport them through the cell body to be released into the surrounding blood vessels. This process, known as transcytosis, effectively removes the protein from the brain’s internal environment and places it into the systemic circulation, where it can be degraded by the liver or cleared by the kidneys.
The final and most critical phase involved the analysis of human brain tissue. The researchers compared the hypothalamic regions of deceased individuals who had been diagnosed with Alzheimer’s disease against those of a healthy control group. The results were striking: in the Alzheimer’s patients, the tanycytes were significantly fragmented. Furthermore, genetic analysis revealed that the genes responsible for the "shuttle" function were downregulated, suggesting that in the diseased state, the brain’s drainage system is structurally and functionally compromised.
Supporting Data and Quantitative Insights
The study’s data highlights a significant correlation between tanycyte health and the severity of tau pathology. In rodent models where tanycyte function was artificially inhibited, the researchers recorded a 40% to 60% increase in tau retention within the brain parenchyma within a relatively short timeframe. Conversely, in models where tanycyte activity was stimulated, the clearance rate of tau into the blood increased significantly.
Data from human tissue samples further reinforced these findings. Quantitative imaging showed that tanycytes in Alzheimer’s patients exhibited a 30% reduction in "process length"—the extensions that reach out to blood vessels—compared to age-matched controls. This structural degradation directly limits the cells’ ability to interface with the circulatory system. Moreover, the study identified specific mRNA markers associated with tau transport that were nearly absent in the advanced stages of the disease, providing a potential molecular target for future diagnostics.
Broader Context: The Brain’s Plumbing System
The discovery of the tanycyte-mediated clearance pathway adds a new layer to our understanding of the "glymphatic system," a term coined by Dr. Maiken Nedergaard in 2012 to describe the brain’s waste-clearance pathway. While the glymphatic system primarily describes how CSF and interstitial fluid exchange to flush out waste during sleep, the role of tanycytes suggests a more localized and specialized "export gate" in the hypothalamus.
The hypothalamus is a region of the brain that controls vital functions such as sleep, temperature, and hunger. The fact that tau clearance is mediated through this region may explain why patients with Alzheimer’s often suffer from metabolic disturbances and disrupted sleep patterns long before cognitive symptoms appear. If tanycytes are failing, it creates a "bottleneck" effect where waste builds up, leading to a toxic environment that accelerates the death of nearby neurons.
Official Responses and Scientific Reactions
The scientific community has reacted to the findings with cautious optimism. While Vincent Prevot and his team at INSERM are leading the charge, the work was a collaborative effort supported by the European Research Council (ERC) and the National Institutes of Health (NIH).
"Focusing on tanycyte health could be a way to improve tau clearance and limit disease progression," Prevot noted in his summary of the implications. By shifting the therapeutic focus from "how to stop tau from forming" to "how to help the brain get rid of it," the research opens a new frontier in drug development.
Independent experts in neurobiology have noted that this study provides the first evidence for structural and functional alterations in these little-known brain cells in the context of human disease. However, they also echo Prevot’s caution regarding the transition from bench to bedside. The complexity of the blood-brain barrier and the difficulty of targeting specific non-neuronal cells without affecting the hypothalamus’s other critical functions remain significant hurdles.
Implications for Future Treatment and Challenges
The potential implications for Alzheimer’s treatment are profound. If tanycyte health can be preserved or restored through pharmacological intervention, it may be possible to slow or even halt the progression of tau accumulation. This "maintenance of internal balance" could serve as a prophylactic approach for individuals at high genetic risk for the disease.
However, the path to a viable treatment is fraught with challenges. One primary obstacle is the current limitation of animal models. Mice do not naturally develop Alzheimer’s disease; researchers must use transgenic models that overexpress certain proteins, which may not perfectly replicate the slow, decades-long progression of the disease in humans.
Furthermore, the study emphasizes the need for longitudinal research. "We need larger patient groups and long-term studies to determine cause and effect," the researchers noted in their discussion. It remains unclear whether tanycyte dysfunction is a primary cause of tau buildup or a secondary consequence of the toxic environment created by the disease. Clarifying this "chicken or egg" scenario is vital for determining when a tanycyte-focused intervention should be administered.
Conclusion and Future Outlook
The identification of tanycytes as key players in tau clearance represents a paradigm shift in neurodegeneration research. By highlighting the importance of the brain’s interface with the rest of the body, the study moves away from a neuron-centric view of Alzheimer’s and toward a more holistic, systems-biology approach.
As the global population ages, the prevalence of Alzheimer’s is expected to rise dramatically, with some estimates suggesting that 150 million people could be living with dementia by 2050. The work supported by the European Research Council, the Fondation pour la Recherche Médicale, and the Fondation NRJ for Neuroscience provides a necessary foundation for a new generation of therapies. While the road to a cure remains long, the discovery of the tanycyte "shuttle" provides a clear and promising new direction for the scientific community to explore in the fight against this devastating condition.














Leave a Reply