In a landmark study that bridges the gap between molecular biophysics and reproductive medicine, a Yale-led research team has successfully mapped the structural mechanism by which a specific biological antibody prevents sperm from bonding with an egg. This discovery, centered on a naturally occurring mechanism found in mammals, provides a high-resolution blueprint for understanding how fertilization can be both hindered and facilitated at the molecular level. By identifying the exact spatial configurations that prevent the "lock and key" interaction between sperm and egg, the researchers have opened a dual-purpose gateway for medical science: creating more effective treatments for couples struggling with immuno-infertility and developing a new generation of non-hormonal contraceptives.
The study, published in the Proceedings of the National Academy of Sciences (PNAS), represents a significant leap forward in reproductive biology. For decades, the precise physical interactions that occur at the moment of conception have remained partially shrouded in mystery. While the proteins responsible for sperm-egg recognition were known, the exact way in which the body’s own immune system can sometimes intervene to block this process had never been visualized at such a granular level. The Yale team, in collaboration with researchers from Osaka University, has now provided the first anti-sperm antibody-antigen complex structure, offering a roadmap for future drug development and diagnostic tools.
The Biological Foundation: IZUMO1 and JUNO
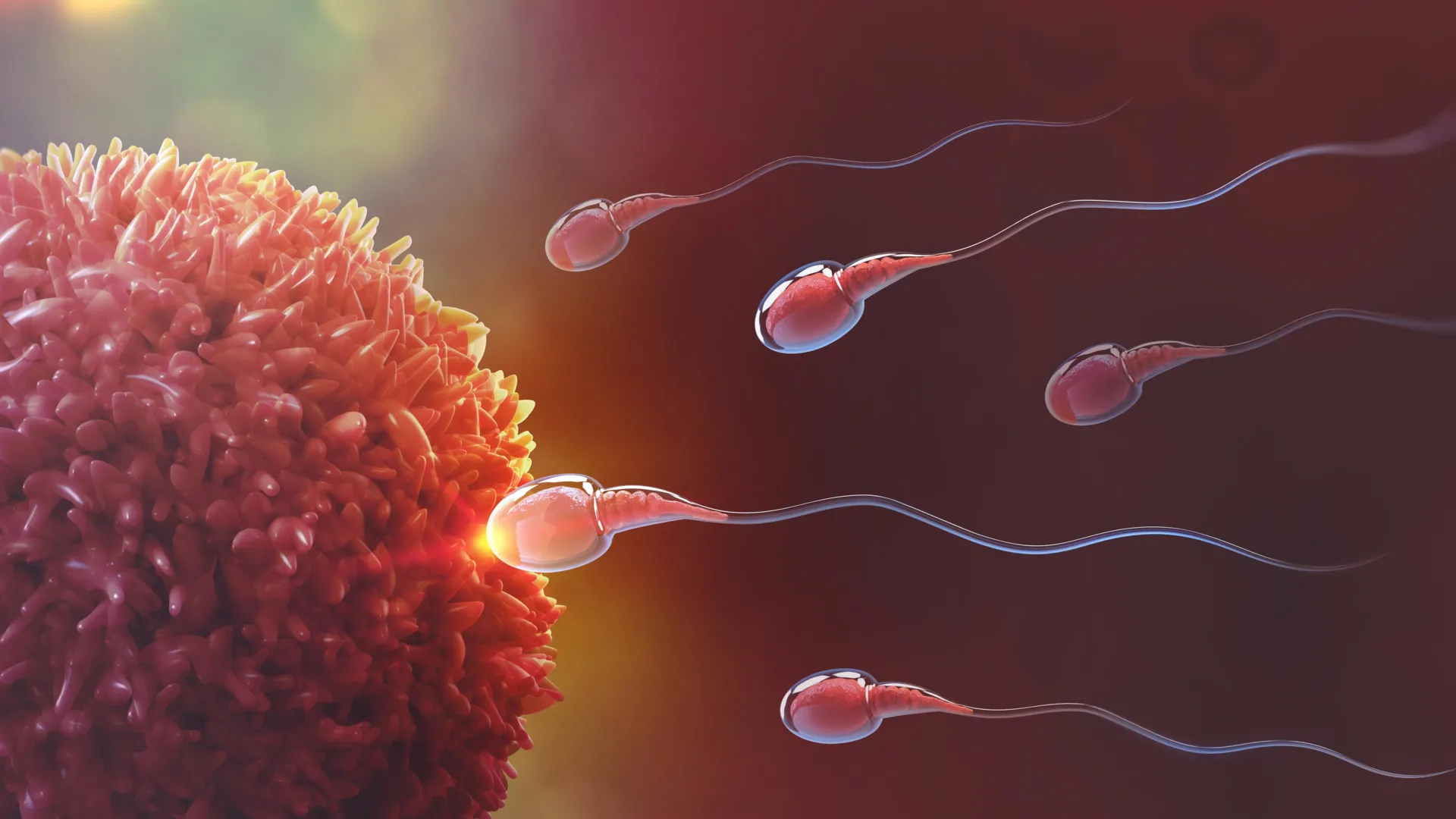
To understand the breakthrough, one must first look at the fundamental proteins involved in mammalian fertilization. The process relies on a sophisticated recognition system involving two primary players: IZUMO1 and JUNO. IZUMO1 is a protein located on the surface of the sperm cell, named after a Japanese shrine dedicated to marriage. Its counterpart, JUNO, is a receptor located on the membrane of the egg (oocyte), named after the Roman goddess of fertility and marriage.
When a sperm cell reaches the egg, IZUMO1 must bind specifically to JUNO to initiate the fusion of the two cells. This connection is the "handshake" that allows the genetic material to combine. If this handshake is weak, improperly aligned, or physically blocked, fertilization cannot occur. Despite the critical nature of this interaction, a variety of factors can disrupt it, leading to what clinicians call "unexplained infertility." According to data from the Centers for Disease Control and Prevention (CDC), approximately 9% of men and 11% of women of reproductive age in the United States face fertility challenges. A significant portion of these cases is attributed to failures in the recognition, adhesion, or fusion stages of the sperm-egg interaction.
The 40-Year Mystery of the OBF13 Antibody
The Yale study focuses on a specific disruptor known as OBF13. This is a naturally occurring monoclonal antibody that was first discovered 40 years ago by researchers at Osaka University in Japan. Early studies established that OBF13 had the unique ability to recognize the IZUMO1 protein on sperm and effectively "neutralize" it, preventing it from binding to the JUNO receptor on the egg.
While OBF13 was known to be a potent inhibitor of fertilization in rodent models, the "how" and "where" of its intervention remained speculative for four decades. Scientists knew the antibody attached to the sperm, but they did not know the exact orientation or the specific molecular sites it occupied. Without this structural data, it was impossible to design medications that could either mimic OBF13 for contraception or block it to treat infertility.
"This will have direct implications for infertility and contraception research, especially immuno-infertility and immuno-contraception," stated Steven Tang, an assistant professor of molecular biophysics and biochemistry in Yale’s Faculty of Arts and Sciences and the corresponding author of the study. Tang’s team sought to visualize this interaction using advanced imaging techniques, finally solving the 40-year-old puzzle.
Methodology: X-Ray Crystallography and the SLAC Accelerator
The research team employed X-ray crystallography to analyze the structure of the IZUMO1 protein as it interacted with the OBF13 antibody. This process involves growing crystals of the protein-antibody complex and then bombarding them with high-intensity X-ray beams. The way the X-rays scatter allows scientists to calculate the exact position of every atom within the molecule.
To achieve the necessary precision, the researchers utilized the SLAC National Accelerator Laboratory in California, a facility supported by the U.S. Department of Energy’s Office of Science. This high-energy environment allowed the team to observe the "high-affinity" variant of OBF13—a version of the antibody that bonds exceptionally tightly to the sperm protein.
The analysis revealed that OBF13 does not just sit on the surface of the sperm; it reconfigures the spatial orientation of the IZUMO1 protein. By attaching itself in a specific geometric pattern, the antibody physically masks the regions of IZUMO1 that are supposed to dock with the JUNO receptor. Essentially, OBF13 acts as a "cap" that prevents the sperm from ever making a successful connection with the egg.
Identifying the Vulnerabilities of JUNO
While the primary focus was on the sperm-side protein (IZUMO1), the researchers also turned their attention to the egg-side receptor, JUNO. In a series of complementary experiments, the team identified key amino acid sites on the JUNO receptor that define its ability to bind with IZUMO1.
The discovery of these specific sites is crucial for overcoming infertility. The researchers found that even when OBF13 or its variants were present, certain modifications or access points on the JUNO receptor could still facilitate a successful bond. By understanding these "binding pockets," scientists may be able to develop therapies that help sperm bypass immune-system interference. This is particularly relevant for patients suffering from immuno-infertility, a condition where the body produces antibodies that mistakenly attack reproductive cells as if they were foreign pathogens.
Chronology of Reproductive Breakthroughs
The Yale-led study is the latest in a long timeline of milestones in the study of fertilization:
- 1980s: Researchers at Osaka University identify the OBF13 antibody and observe its ability to block fertilization in mice.
- 2005: The IZUMO1 protein is identified as a critical factor in sperm-egg fusion.
- 2014: The JUNO receptor is discovered on the egg, completing the "lock and key" model.
- 2016–2020: Studies begin to explore the structural biology of these proteins, but high-resolution complexes with antibodies remain elusive.
- 2024: The Yale and Osaka team publishes the first high-resolution structure of the anti-sperm antibody-antigen complex, detailing the OBF13-IZUMO1 interaction.
Implications for Non-Hormonal Contraception
One of the most promising applications of this research is in the field of male contraception. Currently, the majority of contraceptive options are female-oriented and often involve hormonal manipulation, which can carry side effects ranging from mood swings to increased risks of blood clots. The search for a "male pill" or a non-hormonal female option has been a "holy grail" of reproductive science for years.
The Yale study provides a direct path toward this goal. By understanding how OBF13 potently blocks fertilization, pharmaceutical researchers can design small-molecule inhibitors or synthetic antibodies that mimic OBF13. These drugs would target the IZUMO1-JUNO interaction specifically, leaving the rest of the body’s hormonal systems untouched.
"We provide high-resolution information that will open avenues for discovering IZUMO1 regulators, guide antibody and small-molecule inhibitor design, and support drug screening for contraceptive development," Tang said. This approach offers a highly targeted "precision medicine" model for birth control, potentially reducing side effects and increasing efficacy.
Addressing the Crisis of Immuno-Infertility
On the opposite side of the spectrum, the research offers hope to the millions of people worldwide who experience infertility. Immuno-infertility occurs when either the male or female body produces anti-sperm antibodies (ASAs). In men, these antibodies can cause sperm to clump together, reducing motility. In women, ASAs can prevent sperm from penetrating the cervical mucus or, as demonstrated in this study, block the sperm from binding to the egg.
Currently, treatments for immuno-infertility are limited and often involve invasive procedures like In Vitro Fertilization (IVF) or Intracytoplasmic Sperm Injection (ICSI). By identifying the specific amino acid sites on JUNO and the reconfiguration caused by OBF13, scientists can now look for ways to neutralize these harmful antibodies or develop "decoy" molecules that allow the sperm to reach the egg unimpeded.
Global Collaboration and Institutional Support
The success of this research highlights the importance of international scientific cooperation. The study’s first author, Yonggang Lu, and co-author Masahito Ikawa are both from Osaka University, the institution where OBF13 was first discovered. This multi-generational, cross-continental effort combined decades of biological observation with cutting-edge American biophysical technology.
The work was supported by a diverse array of prestigious organizations, reflecting the high stakes of the research. Funding was provided by the National Institutes of Health (NIH), the David Sokal Innovation Award of the Male Contraception Initiative, the Japan Society for the Promotion of Science, the Japan Agency for Medical Research and Development, and the Takeda Science Foundation.
A New Frontier in Reproductive Science
The mapping of the IZUMO1-OBF13 complex marks the beginning of a new era in molecular reproductive medicine. While the study was conducted using rodent models—a standard practice in early-stage biological research—the IZUMO1 and JUNO proteins are highly conserved across mammalian species, including humans. This means the structural insights gained are highly likely to translate to human physiology.
As the scientific community digests these findings, the next steps will involve high-throughput drug screening to find molecules that can either mimic or inhibit the OBF13 effect. For the 1 in 10 couples facing fertility issues, and for the millions seeking safer contraceptive alternatives, the resolution of this 40-year-old molecular mystery provides a clear path forward. The "handshake" between sperm and egg is no longer a hidden event; it is a visible, understandable, and ultimately, a manageable biological process.












Leave a Reply