Researchers from the Department of Medicine at the School of Clinical Medicine, LKS Faculty of Medicine, University of Hong Kong (HKUMed) have reached a significant milestone in the field of mechanobiology by identifying a specific biological process that explains how physical activity maintains skeletal integrity. The study, published in the prestigious journal Signal Transduction and Targeted Therapy, highlights the discovery of a protein known as Piezo1, which functions as the body’s internal "exercise sensor." This protein allows bone tissue to perceive and respond to physical movement, a finding that could revolutionize the treatment of osteoporosis and age-related bone loss, particularly for patients who are physically unable to engage in traditional exercise.
The research team, led by Professor Xu Aimin, Director of the State Key Laboratory of Pharmaceutical Biotechnology and Chair Professor at HKUMed, has effectively decoded the molecular language through which mechanical stress is translated into bone growth. By identifying Piezo1 and the signaling pathways it governs, the team has opened a pathway for the development of "exercise mimetics"—pharmacological interventions designed to replicate the physiological benefits of physical activity at the cellular level. This discovery offers a potential lifeline for the elderly, bedridden individuals, and those suffering from chronic illnesses that limit mobility, all of whom face a significantly elevated risk of debilitating bone fractures.
The Escalating Global Crisis of Osteoporosis and Bone Fragility
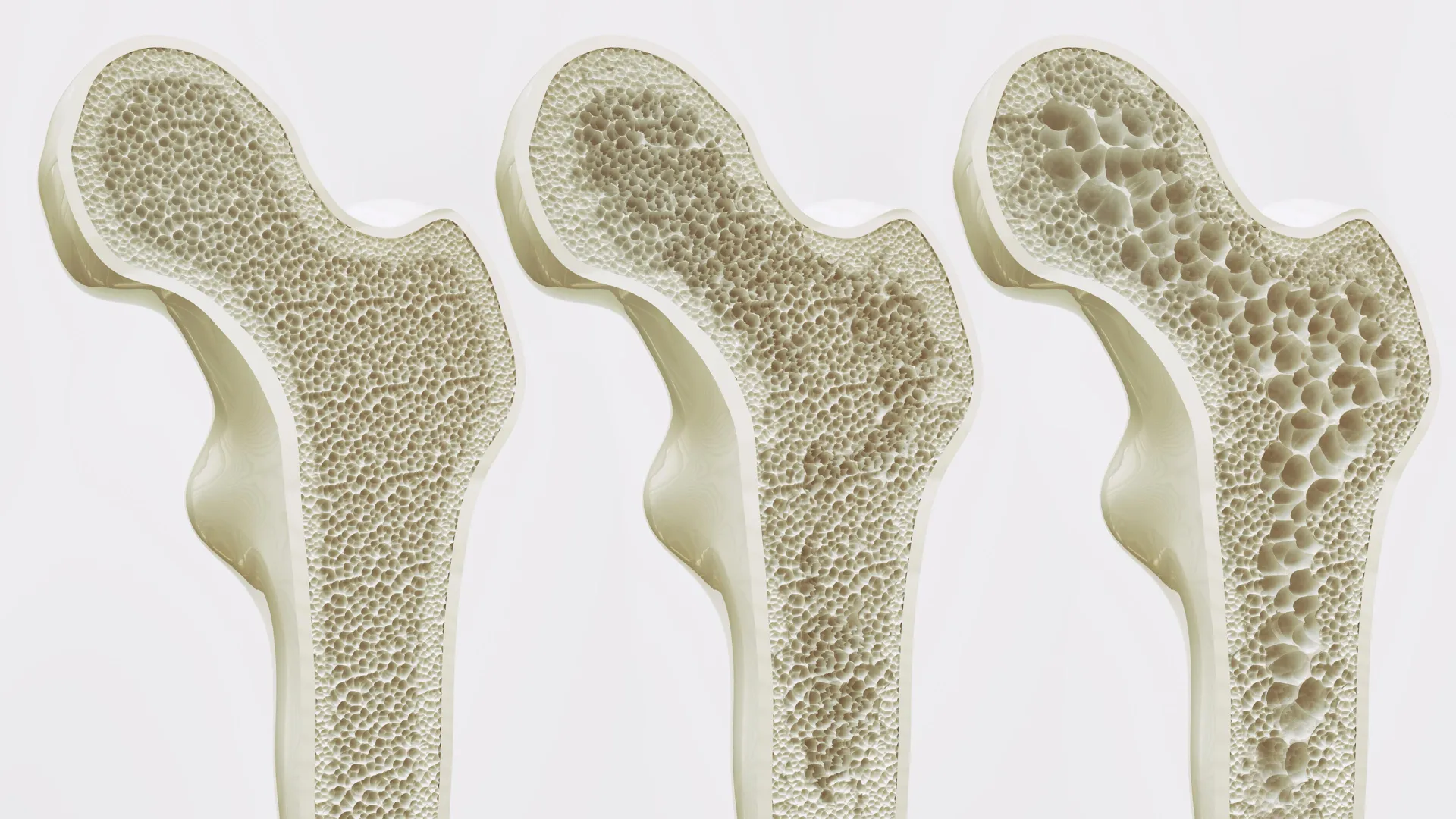
Osteoporosis is often characterized as a "silent epidemic" because bone loss typically occurs without symptoms until a fracture happens. The scale of the problem is immense and is expected to grow as global populations age. According to data from the World Health Organization (WHO), approximately 200 million people worldwide suffer from osteoporosis. The clinical implications are severe: roughly one in three women and one in five men over the age of 50 will experience an osteoporotic fracture in their lifetime.
In Hong Kong, the situation is particularly acute due to the city’s rapidly aging demographic. Current statistics indicate that osteoporosis affects 45% of women and 13% of men aged 65 and older. These fractures, particularly of the hip and spine, are not merely orthopedic issues; they are major drivers of mortality and morbidity. Hip fractures, in particular, are associated with a 20% mortality rate within the first year and often lead to a permanent loss of independence for survivors. Beyond the personal toll, the economic burden on healthcare systems is staggering, involving long-term hospitalization, surgery, and extensive rehabilitation services.
The primary challenge in managing this crisis is that the most effective non-pharmacological intervention—weight-bearing exercise—is often inaccessible to the very populations who need it most. Frailty, sarcopenia (muscle loss), and neurological conditions frequently prevent high-risk patients from performing the movements necessary to stimulate bone density. This creates a therapeutic gap that the HKUMed research aims to bridge.
Decoding the Cellular Switch: The Competition Between Bone and Fat
To understand the significance of the Piezo1 discovery, it is necessary to examine the internal environment of the bone marrow. Within the marrow reside mesenchymal stem cells (MSCs), which are versatile "progenitor" cells capable of differentiating into various types of tissue. In a healthy, active individual, these stem cells are regularly signaled to become osteoblasts—the cells responsible for building new bone.
However, the aging process and physical inactivity disrupt this delicate balance. As the body ages or remains sedentary, the "fate" of these mesenchymal stem cells shifts. Instead of becoming bone-forming cells, they increasingly differentiate into adipocytes, or fat cells. This shift leads to the accumulation of marrow adipose tissue (MAT). This process is doubly destructive: not only is less new bone being created, but the expanding fat cells crowd out healthy tissue and create a pro-inflammatory environment that further weakens the bone structure. This "fatty bone" phenomenon makes the skeletal system more porous and brittle, significantly increasing the likelihood of fractures even from minor falls.
Piezo1: The Molecular Gateway to Physical Benefits
The HKUMed study utilized a combination of sophisticated mouse models and human stem cell analysis to isolate the mechanism that prevents this fatty degeneration. The researchers identified Piezo1, a mechanosensitive ion channel located on the surface of mesenchymal stem cells. Piezo1 acts as a transducer, converting the physical pressure of movement into chemical signals within the cell.
Through a series of experiments, the team demonstrated that when Piezo1 is activated—either through physical movement or through targeted stimulation—it sends signals that effectively "lock" the stem cells into a bone-building pathway. In mouse models where Piezo1 was specifically deleted from bone marrow stem cells, the results were dramatic. Even with regular movement, these mice showed a rapid increase in bone marrow fat and a significant decrease in bone density, mimicking the effects of advanced osteoporosis.
The study further revealed that the absence of Piezo1 triggers the release of specific inflammatory signals, namely Ccl2 and lipocalin-2. These molecules act as chemical messengers that accelerate the conversion of stem cells into fat and actively inhibit the production of new bone. Crucially, the researchers found that by blocking these inflammatory signals, they could partially restore the healthy balance of bone over fat, suggesting multiple points for potential medical intervention.
Chronology of the Research and Collaborative Efforts
The discovery of Piezo1’s role in bone health is the culmination of years of multidisciplinary research. The project involved a high level of international cooperation, reflecting the global importance of the findings.
- Phase I: Identification and Hypothesis: The research began with the observation that physical inactivity leads to rapid bone loss in both humans (such as astronauts or bedridden patients) and animal models. Researchers focused on identifying the specific "sensor" that detects mechanical loading.
- Phase II: Laboratory Experimentation: Using CRISPR-Cas9 technology and other genetic tools, the team developed "knockout" mice lacking the Piezo1 protein in specific cell lines. This allowed them to isolate the protein’s function with high precision.
- Phase III: Signaling Pathway Mapping: Once Piezo1 was identified, the team mapped the downstream effects, discovering the roles of Ccl2 and lipocalin-2. This provided a comprehensive map of the "exercise-to-bone" communication channel.
- Phase IV: Peer Review and Publication: The findings were subjected to rigorous peer review before being published in Signal Transduction and Targeted Therapy, a leading journal in the field of molecular medicine.
The study was co-led by Professor Xu Aimin and Dr. Wang Baile, Research Assistant Professor at HKUMed. Significant contributions were also made by Professor Eric Honoré of the Institute of Molecular and Cellular Pharmacology in France, bringing together expertise from the French National Centre for Scientific Research (CNRS) and the University of Hong Kong.
Official Responses and Expert Perspectives
The research team has expressed high levels of optimism regarding the clinical translation of these findings. Professor Xu Aimin emphasized that the study addresses a fundamental limitation in current geriatric care.
"Current treatments for osteoporosis rely heavily on physical activity, which many patients simply cannot perform," Professor Xu stated. "We need to understand how our bones get stronger when we move or exercise before we can find a way to replicate the benefits of exercise at the molecular level. This study is a critical step towards that goal."
Dr. Wang Baile highlighted the specific benefits for vulnerable populations, noting that the discovery of "exercise mimetics" could preserve the independence of elderly patients. "Our findings open the door to developing drugs that chemically activate the Piezo1 pathway to help maintain bone mass. This is especially meaningful for patients who are frail or recovering from serious injury."
Professor Eric Honoré added that the implications of the study extend beyond traditional physical therapy. "In the future, we could potentially provide the biological benefits of exercise through targeted treatments, thereby slowing bone loss in groups such as bedridden patients or those with limited mobility, and substantially reducing their risk of fractures."
Broader Impact and Future Implications
The identification of Piezo1 as an exercise sensor has implications that reach far beyond the treatment of osteoporosis. It contributes to the growing field of "mechanomedicine," which seeks to treat diseases by targeting the way cells respond to physical forces.
1. Aerospace and Space Medicine:
One of the most significant challenges for long-term space travel is the rapid loss of bone density (spaceflight-induced osteoporosis) due to the lack of gravity. Astronauts currently spend hours each day on specialized exercise equipment to mitigate this effect. Piezo1-targeted therapies could potentially provide a pharmacological supplement to protect the skeletal health of explorers on long-duration missions to Mars and beyond.
2. Rehabilitation and Critical Care:
Patients in intensive care units (ICU) or those suffering from spinal cord injuries often experience rapid skeletal and muscular atrophy. By "tricking" the body into thinking it is exercising, clinicians may be able to maintain the bone health of these patients during their recovery, shortening rehabilitation times and improving long-term outcomes.
3. Economic Impact on Healthcare:
By reducing the incidence of hip and spinal fractures, Piezo1-based treatments could save billions of dollars in healthcare costs globally. The reduction in the need for surgical interventions and long-term nursing care would alleviate the pressure on public health systems in aging societies.
Funding and Institutional Support
This groundbreaking research was made possible through extensive support from several major scientific and governmental bodies. In Hong Kong, the project received funding from the Research Grants Council’s Areas of Excellence Scheme and the General Research Fund, as well as the Health and Medical Research Fund under the Health Bureau.
International support was equally robust, including contributions from the National Key R&D Program of China, the National Natural Science Foundation of China, and the Human Frontier Science Program. Additional funding was provided by French institutions, including the French National Research Agency and various foundations for medical research, as well as the Macau Science and Technology Development Fund.
As the HKUMed team moves toward the clinical application phase, their work stands as a testament to the power of collaborative, molecular-level research in solving some of the most pressing public health challenges of the 21st century. The transition from "understanding" to "mimicking" the benefits of exercise marks a new era in the fight against age-related degeneration.















Leave a Reply