Glioblastoma multiforme remains one of the most formidable challenges in modern oncology, characterized by its rapid progression, high rate of recurrence, and a prognosis that has seen little improvement despite decades of research. Classified as a Grade IV astrocytoma by the World Health Organization, these tumors are notoriously resistant to conventional therapies, including surgical resection, radiotherapy, and chemotherapy with temozolomide. For many patients, even with the most aggressive standard-of-care treatments, the average life expectancy remains under two years. However, a groundbreaking study from the German Cancer Consortium (DKTK) at the West German Tumor Center Essen has unveiled a previously unknown dimension of the body’s immune response to these tumors. Researchers have discovered that the skullcap, long viewed merely as a protective casing for the brain, houses "islands" of highly potent immune cells that play a decisive role in the fight against glioblastoma. This discovery not only challenges fundamental assumptions about the lymphatic and immune systems but also forces a critical re-evaluation of how neurosurgical procedures are conducted.
A Paradigm Shift in Immunological Understanding
For decades, the prevailing view in immunology was that the body’s defense system operated as a centralized, holistic network. It was believed that immune cells were produced in primary lymphoid organs like the bone marrow of the long bones or the thymus, and were then circulated through the blood and lymphatic systems to wherever they were needed. When a tumor appeared in the brain, it was assumed that the immune response was recruited from these systemic reserves.
The findings from the Essen-based research team, led by Professor Björn Scheffler, suggest a much more localized and specialized arrangement. "What we have found is surprising and fundamentally new," Scheffler explains. The data indicates that in the case of glioblastomas, the body does not rely solely on systemic recruitment. Instead, it organizes a "forward operating base" within the regional bone marrow niches of the skull, directly adjacent to the site of the malignancy. These niches act as concentrated reservoirs of anti-tumor activity, gathering and maturing immune cells specifically tasked with infiltrating the brain to combat the tumor.
This localized organization is a biological revelation. It suggests that the immune system possesses a level of spatial sophistication previously unrecognized in neuro-oncology. The presence of these highly potent immune cells in the skull’s marrow niches implies that the "battleground" for brain cancer extends beyond the blood-brain barrier and into the very bone structure surrounding the central nervous system.
From Animal Models to Human Validation: The Research Chronology
The journey to this discovery began with observations in animal models. Preliminary experiments on mice suggested that there might be a direct communication pathway between the bone marrow of the skull and the meninges, the protective membranes covering the brain. These animal studies indicated that immune cells could migrate through microscopic channels from the bone to the brain surface, bypassing the traditional systemic circulation.
To determine if this phenomenon existed in humans, the team at the West German Tumor Center Essen had to develop entirely new protocols for tissue sampling. First author Celia Dobersalske emphasized the technical hurdles involved in obtaining viable bone marrow samples from the skullcap of untreated glioblastoma patients. Unlike traditional bone marrow biopsies, which are typically taken from the iliac crest (hip bone), sampling the skull marrow during the early stages of a neurosurgical intervention requires extreme precision to avoid contamination and preserve the cellular architecture.
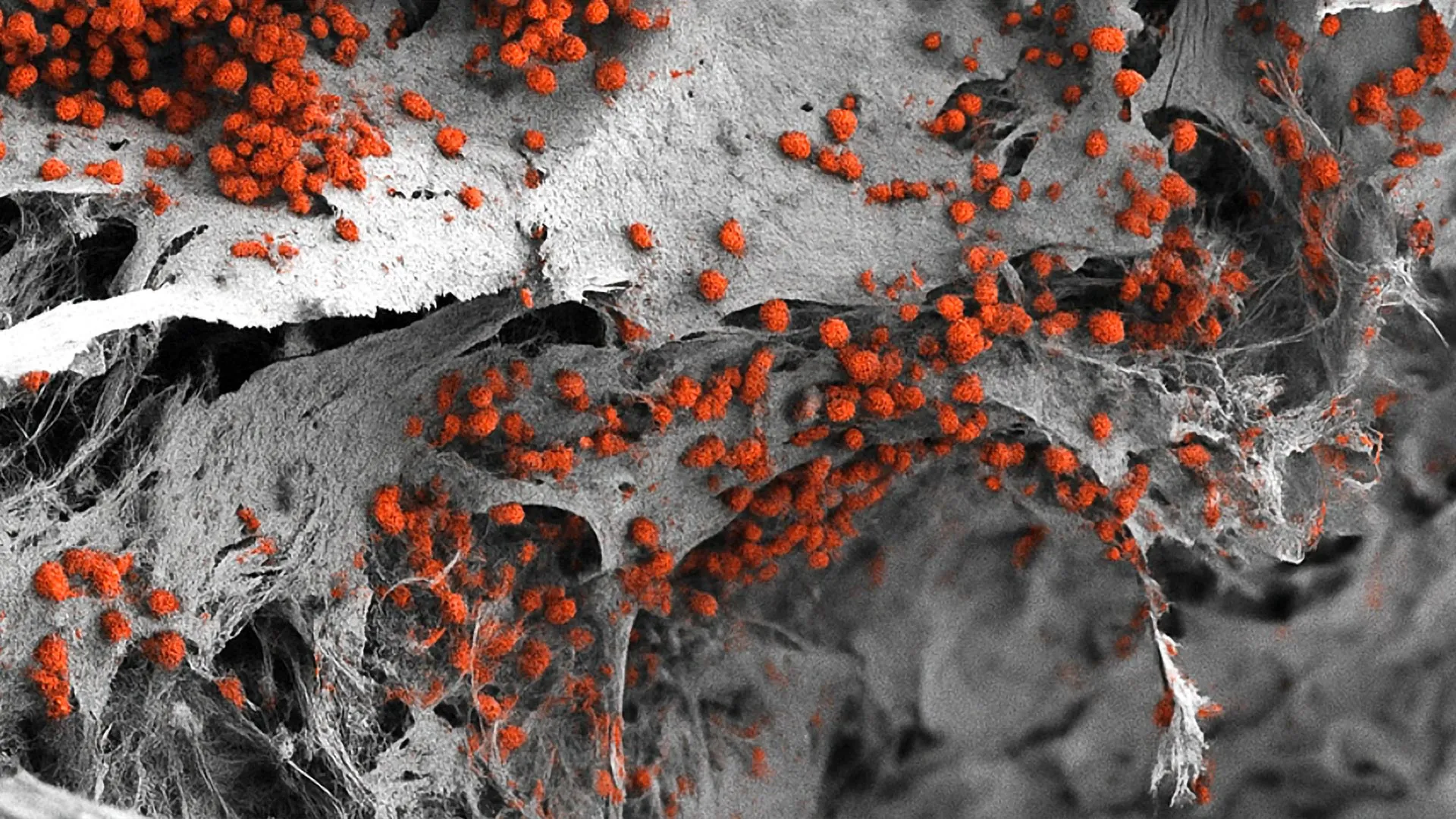
Once the methods were established, the results were definitive. The researchers identified bone marrow niches in the skull that were densely populated with active lymphoid stem cells. These are the "parent" cells that eventually differentiate into various types of white blood cells. More importantly, the researchers found a significant concentration of mature cytotoxic T lymphocytes, specifically CD8+ cells, within these skull-based niches.
The Role of CD8+ Cells and Clonal Evidence
CD8+ T cells are the "assassin" cells of the immune system. Their primary function is to recognize specific antigens on the surface of malignant or infected cells and initiate a process that leads to the destruction of those cells. In the context of glioblastoma, the presence of these cells is a critical indicator of the body’s attempt to mount an immune response.
The Essen team’s discovery went beyond merely finding these cells; they were able to prove their direct involvement in the fight against the tumor. By utilizing advanced genetic sequencing and cellular mapping, the researchers identified that the CD8+ cells in the bone marrow niches and the CD8+ cells found infiltrating the glioblastoma tissue were descendants of the same cell clones. A "clone" in this context refers to a group of cells that all originate from a single parent cell, sharing the same specific T-cell receptor.
This clonal relationship provides "smoking gun" evidence of a direct pipeline between the skull’s bone marrow and the brain tumor. The immune cells are not just sitting in the bone; they are actively proliferating there and then migrating into the tumor to engage the cancer. Furthermore, the researchers observed that these local CD8+ cells possessed an increased number of surface receptors associated with cell proliferation and activation, suggesting they are in a state of high readiness compared to their counterparts in the general circulation.
The Surgical Dilemma: Balancing Resection and Preservation
The discovery of these immune reservoirs presents a profound ethical and technical dilemma for neurosurgeons. For over a century, the standard approach to treating a suspected brain tumor has involved a craniotomy—the surgical removal of a section of the skull to gain access to the brain. This is necessary to biopsy the tissue, confirm the diagnosis, and debulk the tumor to reduce intracranial pressure.
However, the new data suggests that the very act of opening the skull may inadvertently destroy the primary staging ground for the body’s natural defense against the tumor. Dr. Ulrich Sure, Director of the Department of Neurosurgery at Essen and a member of the research team, acknowledged the gravity of this finding. "In view of the new findings, we find ourselves in a dilemma," Sure stated. "We have to gain access to the tumor in order to remove it… but we are thinking about how we can minimize damage to the local bone marrow in the future."
This realization may lead to a transformation in surgical techniques. Future neurosurgical protocols may need to incorporate "marrow-sparing" approaches, perhaps utilizing smaller "keyhole" incisions or navigated surgical paths that avoid the most dense immune niches in the skullcap. The goal would be to preserve the integrity of the regional bone marrow while still achieving the necessary surgical objectives within the brain.
Supporting Data: Correlation with Disease Progression
The clinical significance of these local immune cells is underscored by the correlation between their activity and patient outcomes. The study demonstrated that the course of the disease in glioblastoma patients is closely tied to the vigor of the local CD8+ T-cell response. Patients who exhibited higher levels of activity and clonal expansion within their skull bone marrow niches generally showed a more robust defense against the tumor, at least in the initial stages of the disease.
This correlation provides a potential new biomarker for clinicians. By assessing the state of the immune niches in the skull at the time of diagnosis, doctors might be able to better predict the aggressiveness of a patient’s tumor and tailor treatments accordingly. It also suggests that the "exhaustion" of these local immune reserves may be a key factor in why glioblastomas eventually overcome the body’s defenses and lead to terminal decline.
Re-evaluating Immunotherapy: Why Previous Trials Failed
The failure of checkpoint inhibitors in glioblastoma has been one of the great disappointments of modern oncology. Checkpoint inhibitors, such as those targeting PD-1 or CTLA-4, have revolutionized the treatment of melanoma and non-small cell lung cancer by "releasing the brakes" on the immune system, allowing T cells to attack tumors more effectively. However, in glioblastoma trials, these drugs have consistently failed to significantly extend survival.
The Essen study offers a compelling explanation for this failure. Previously, researchers focused on the systemic immune system or the environment within the tumor itself. They may have been looking in the wrong place. If the most "fit" and "potent" immune cells are located in regional niches in the skull, systemic administration of drugs might not be reaching these cells in sufficient concentrations, or the drugs might not be addressing the specific microenvironment of the bone marrow niches.
Björn Scheffler suggests that the local immune cells are clearly capable of fighting the tumor but are simply overwhelmed or lack the sustained "fuel" to finish the job. "This is where we can start," Scheffler says. The discovery breathes new life into the potential for immunotherapy in brain cancer, provided the delivery mechanism is refined.
Broader Impact and Future Therapeutic Prospects
The implications of this research extend far beyond the laboratory. If the regional bone marrow can be targeted directly, it opens the door for innovative delivery systems. This could include the use of specialized implants, regional infusions, or even modified surgical scaffolds that release immunotherapeutic agents directly into the bone marrow niches of the skullcap.
The challenge, as Scheffler notes, is timing and concentration. Delivering the right drugs to the regional bone marrow at the point when the immune cells are most active could provide the necessary boost to turn a temporary defense into a sustained therapeutic effect. This "niche-targeted" therapy represents a significant shift from the systemic "shotgun" approach of traditional chemotherapy and immunotherapy.
Furthermore, this research may have implications for other types of brain tumors or even neurodegenerative diseases where the immune system plays a role. If the skull functions as a specialized immunological hub for the brain, it could change our understanding of how the central nervous system interacts with the rest of the body in various disease states.
The work, funded by the Wilhelm Sander Foundation and the DKTK Joint Funding Program ‘HematoTrac’, marks a milestone in neuro-oncology. While it does not yet provide a cure for glioblastoma, it provides a roadmap for a new generation of treatments. By recognizing the skull not as a barrier, but as a vital ally in the fight against cancer, researchers and surgeons can begin to develop strategies that work in harmony with the body’s localized defenses, potentially offering a glimmer of hope for patients facing this devastating diagnosis. In the coming years, the focus will likely shift toward clinical trials that test these "marrow-sparing" and "niche-targeting" theories, moving the field closer to a reality where glioblastoma is no longer an automatic death sentence.












Leave a Reply