In a significant leap forward for the fields of hematology and regenerative medicine, scientists at the Indiana University School of Medicine have successfully engineered a sophisticated imaging methodology that allows for the unprecedented visualization of mouse bone marrow. This technological milestone, achieved by overcoming the structural and biochemical hurdles that have long obscured the inner workings of bone tissue, provides a high-definition window into the complex microenvironments where blood and immune cells are born. By utilizing cutting-edge multiplex imaging, the research team has established a protocol that could fundamentally alter the trajectory of drug development for leukemia, autoimmune diseases, and a host of musculoskeletal disorders.
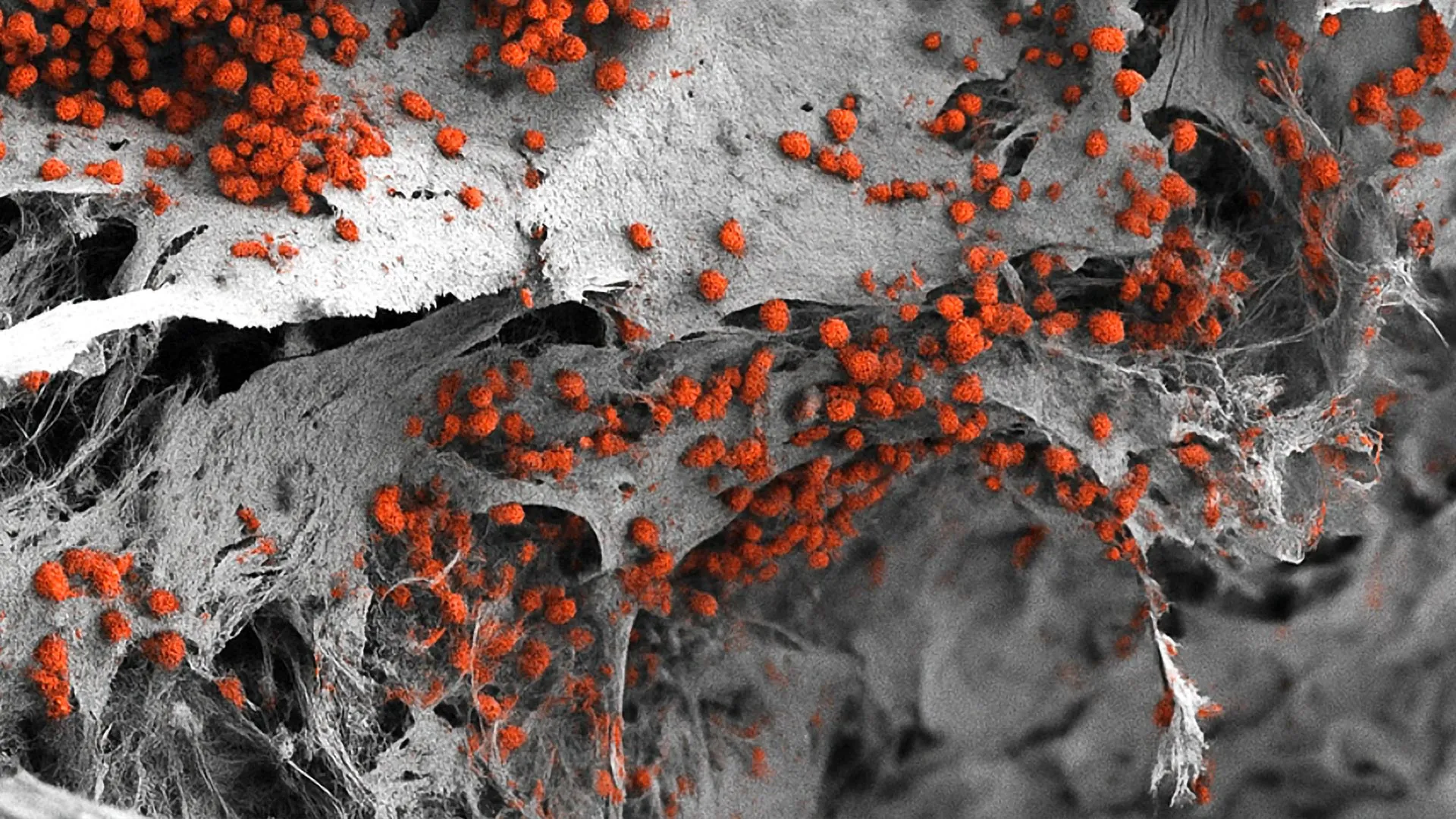
The breakthrough, recently detailed in the peer-reviewed journal Leukemia, centers on the application of the Phenocycler 2.0 platform. This tool has enabled researchers to map a record-breaking number of cellular markers simultaneously within intact tissue. For decades, the "spatial biology" of bone marrow remained one of the most difficult frontiers in medical imaging. Because bone marrow is a delicate, gelatinous substance encased within a rigid, mineralized shell, capturing its internal architecture without destroying the very structures being studied has been a persistent challenge for the global scientific community.
The Biological Frontier: Why Bone Marrow Imaging Matters
Bone marrow serves as the primary "factory" of the human body, responsible for hematopoiesis—the continuous production of red blood cells, white blood cells, and platelets. Within this tissue lies the hematopoietic stem cell (HSC) niche, a specialized environment where stem cells reside, self-renew, and differentiate. Understanding the spatial arrangement of these cells is not merely an academic exercise; it is essential for understanding how diseases like leukemia take root.
In many blood cancers, malignant cells hijack the bone marrow niche, altering the local environment to support tumor growth while suppressing healthy blood production. Until now, researchers lacked the tools to see these interactions in high resolution across a broad spectrum of cell types simultaneously. The ability to visualize 25 different cellular markers at once—compared to the three or four markers allowed by traditional methods—provides a "satellite view" of the cellular landscape, allowing scientists to see not just which cells are present, but exactly where they are located and which neighboring cells they are communicating with.
Sonali Karnik, PhD, an assistant research professor of orthopedic surgery at the IU School of Medicine and co-lead author of the study, emphasized the dual nature of the tissue that makes it so elusive. "Bone marrow is difficult to study because it is gelatinous and encased in hard bone," Karnik noted. "Since bone marrow plays an important role in blood and immune cell formation and houses valuable stem cells, our unique imaging approach offers a useful tool for a variety of research applications."
Overcoming the Limitations of Traditional Diagnostics
To appreciate the magnitude of the IU School of Medicine’s achievement, one must consider the limitations of the current gold standards in tissue analysis: flow cytometry and standard immunofluorescence.
Flow cytometry has long been the workhorse of hematology. It allows scientists to quantify different cell populations with extreme precision by suspending cells in a fluid and passing them through a laser. However, this process requires "dissociating" the tissue—essentially putting it into a blender to create a cellular soup. While this tells researchers what cells are in the marrow, it completely destroys the spatial context. It is the difference between having a list of every person in a city and having a map that shows who lives next to whom and where the boundaries of the neighborhoods are.
Standard fluorescence imaging, on the other hand, preserves the tissue structure but is severely limited by the "color barrier." Because of the way light wavelengths overlap, traditional microscopes can typically only distinguish between three or four different fluorescently labeled markers at a time. This is insufficient for studying the bone marrow, which contains dozens of distinct cell types, including various stages of stem cells, immune cells, blood vessel cells, and bone-forming cells.
The new methodology developed at Indiana University bypasses these constraints. By using the Phenocycler 2.0—a system that uses DNA-barcoded antibodies and iterative cycles of imaging—the team was able to visualize 25 different markers within a single, intact slice of mouse bone marrow. This "multiplexing" capability ensures that the delicate spatial relationships between stem cells and their protective "niches" remain preserved and visible.
Technical Execution and the Phenocycler 2.0 Integration
The success of the study relied on the IU Cooperative Center of Excellence in Hematology’s ability to adapt the Phenocycler technology—originally designed for softer organs like the spleen, kidneys, and brain—to the unique rigors of bone.
The preparation of bone tissue for multiplex imaging is a notoriously finicky process. The bone must be decalcified to allow for sectioning, yet the chemical process of decalcification often degrades the very proteins (antigens) that the antibodies need to bind to. The IU team spent months refining a protocol that balances structural integrity with molecular preservation.
By successfully applying this to mouse models, the researchers have created a blueprint for future human studies. Mouse models are the bedrock of preclinical medical research because their biological systems closely mimic human physiology, particularly in the realm of the immune system and skeletal development. The ability to map 25 markers in a mouse model means that drug manufacturers can now observe, in real-time, how a new chemotherapy agent or an immunotherapy drug affects specific cell populations within the bone marrow before moving to human trials.
Strategic Implications for Oncology and Immunology
The implications for cancer research are particularly profound. In diseases such as acute myeloid leukemia (AML) or multiple myeloma, the spatial distribution of cancer cells often determines how a patient will respond to treatment. Some cancer cells may hide in "sanctuary sites" within the bone marrow where they are shielded from drugs by the surrounding tissue architecture.
"Because mouse models are widely used to study human diseases, this technique offers a promising new method for investigating a range of conditions like autoimmune diseases, leukemia and other disorders involving bone marrow," said Reuben Kapur, PhD, a co-senior author on the study. Kapur, who serves as the director of the IU School of Medicine’s Herman B Wells Center for Pediatric Research, highlighted that this tool is not just about seeing cells, but about understanding the "neighborhoods" of disease.
In the context of autoimmune diseases, such as lupus or rheumatoid arthritis, the bone marrow often serves as a reservoir for long-lived plasma cells that drive chronic inflammation. The new imaging technique will allow researchers to pinpoint where these cells reside and what signals are keeping them alive, potentially leading to therapies that can "evict" these harmful cells from their niches.
Chronology of Development and Future Expansion
The development of this technique marks a major milestone in a multi-year effort at Indiana University to modernize hematological research. The IU Innovation and Commercialization Office has already moved to protect the intellectual property generated by this research, filing a provisional patent for the specific imaging methodology.
The research team is not resting on the 25-marker milestone. According to the study authors, work is already underway to expand the marker panel. The goal is to create a "comprehensive atlas" of the bone marrow that includes:
- Neural Networks: Mapping how nerves within the bone influence blood cell production.
- Vasculature: Visualizing the intricate web of blood vessels that transport new cells into the general circulation.
- Muscular Interfaces: Examining how the surrounding muscle tissue interacts with the bone surface.
- Advanced Signaling: Identifying the specific chemical signals (cytokines and chemokines) that cells use to communicate in real-time.
By adding these layers, the IU School of Medicine aims to provide the most complete picture of the bone marrow microenvironment ever produced.
Collaborative Effort and Institutional Support
The project was a massive collaborative effort, involving a diverse team of experts from various departments within the IU School of Medicine. In addition to Dr. Karnik and Dr. Kapur, the study included contributions from Connor Gulbronson, Paige C. Jordan, Rahul Kanumuri, Baskar Ramdas, Ramesh Kumar, Melissa L. Hartman, Izza Khurram, Drew M. Brown, Karen E. Pollok, Pratibha Singh, and Melissa A. Kacena.
The research was made possible through significant federal investment, primarily from the National Institutes of Health (NIH). This funding underscores the federal government’s commitment to "spatial biology" as the next major frontier in the fight against complex diseases. The IU Cooperative Center of Excellence in Hematology, where much of the work was conducted, is one of only a handful of such centers in the United States, positioning Indiana as a global hub for blood-related research.
Analysis: The Future of Precision Medicine
The move toward high-plex imaging reflects a broader shift in medicine toward "precision diagnostics." As therapies become more targeted—such as CAR-T cell therapy or gene editing—the tools used to evaluate these therapies must become equally precise.
If a scientist develops a drug meant to protect hematopoietic stem cells during radiation, they need to see exactly where those stem cells are and whether the drug is reaching them. The IU imaging technique provides that verification. Furthermore, as the marker panel expands to include more than 40 or 50 markers, the "resolution" of medical understanding will move from the tissue level to the molecular level, all while maintaining the vital context of the living organism.
As the IU School of Medicine moves toward commercializing this methodology and expanding its marker library, the scientific community anticipates a surge in similar studies across other "difficult" tissues. The success in bone marrow suggests that no part of the anatomy is truly unreachable if the right combination of multiplex technology and biochemical preparation is applied.
In the long term, this technique may pave the way for "spatial biopsies" in human patients, where a small sample of bone marrow could be mapped in 3D to provide a personalized treatment plan for cancer or immune disorders. For now, the IU team has provided the global research community with a powerful new lens through which to view the hidden complexities of the bone.












Leave a Reply