The landscape of Alzheimer’s disease treatment has undergone a seismic shift in recent years, moving from purely symptomatic management to the introduction of disease-modifying therapies. However, while the current generation of monoclonal antibodies has proven that clearing amyloid-beta proteins can slow cognitive decline, these treatments come with significant logistical and physiological burdens. In a groundbreaking study published on March 5 in the journal Science, researchers at Washington University School of Medicine in St. Louis have unveiled a novel cellular immunotherapy that could potentially revolutionize the field. By reprogramming astrocytes—the most abundant cells in the central nervous system—to act as specialized waste-removal agents, the team has demonstrated a method that requires only a single intervention to achieve dramatic reductions in brain-clogging plaques.
The Evolution of Alzheimer’s Therapeutics
To understand the significance of this new development, one must first look at the current gold standard of care. The latest class of Alzheimer’s medications, known as monoclonal antibodies, includes drugs like lecanemab and donanemab. These therapies represent the first time medical science has successfully slowed the progression of the disease itself rather than merely masking its symptoms. Clinical trials have shown that these treatments allow patients to maintain their independence and cognitive function for approximately 10 additional months compared to those on a placebo.
The mechanism of these drugs involves the intravenous administration of antibodies that travel to the brain to bind with and help clear amyloid-beta, a protein that aggregates into toxic plaques. Despite their success, these treatments are far from a "silver bullet." They require large, repeated doses through infusions once or twice a month. This creates a significant burden for patients and caregivers, necessitates specialized infusion centers, and carries risks of side effects such as Amyloid-Related Imaging Abnormalities (ARIA), which can involve brain swelling or microhemorrhages. The search for a more "one-and-done" approach that is both more effective and less invasive has led researchers to look toward the burgeoning field of cellular immunotherapy.
Repurposing Cancer Treatment Strategies for Neurodegeneration
The experimental therapy developed at Washington University is inspired by Chimeric Antigen Receptor (CAR) T-cell therapy, a revolutionary treatment for certain types of blood cancers. In CAR-T therapy, a patient’s own immune T-cells are harvested and genetically engineered to produce a specific receptor—the CAR—on their surface. This receptor acts as a homing device, allowing the T-cells to identify, latch onto, and destroy cancer cells with surgical precision.
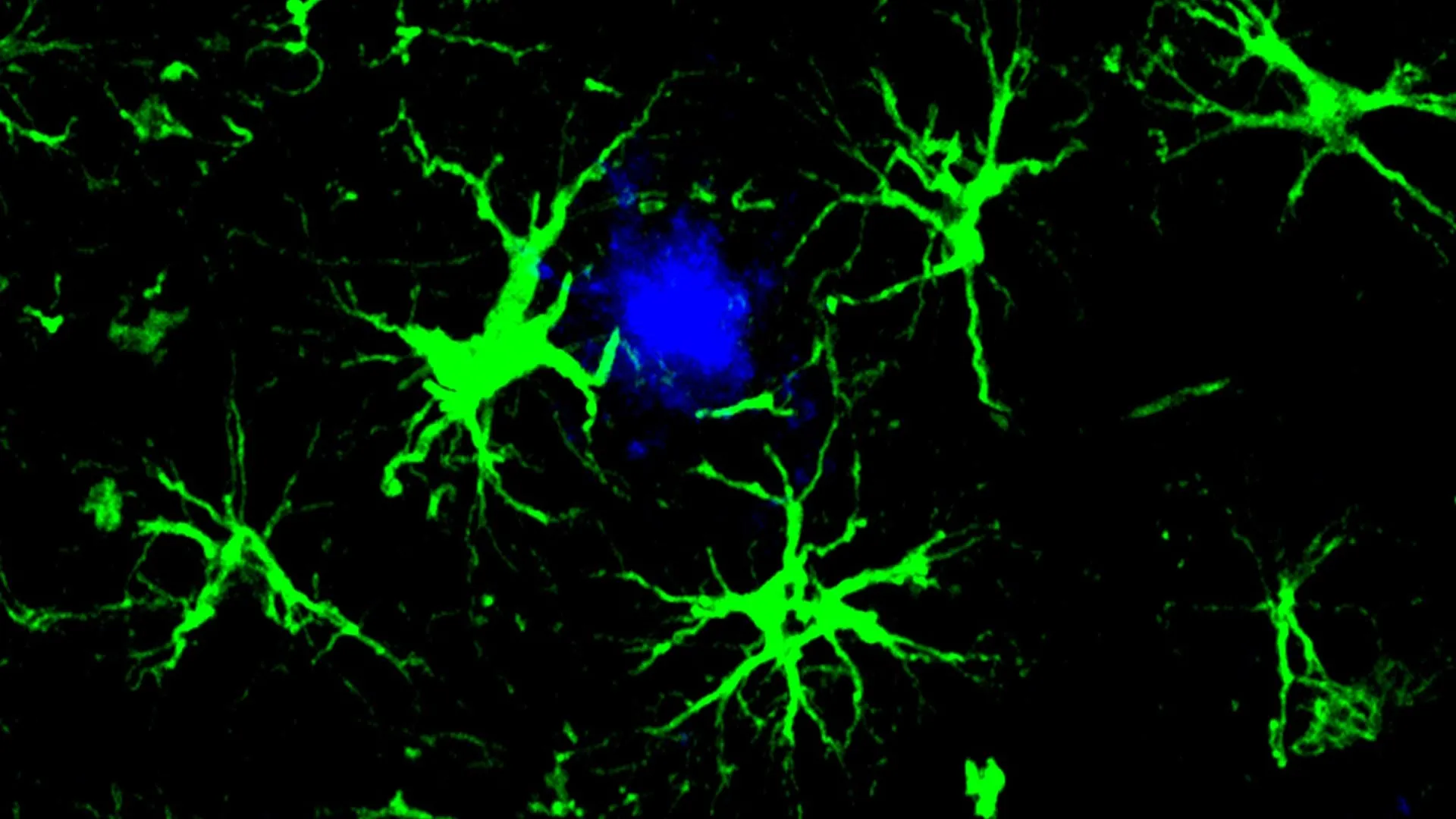
In applying this logic to Alzheimer’s, the research team, led by senior author Marco Colonna, MD, the Robert Rock Belliveau, MD, Professor of Pathology, and first author Yun Chen, PhD, decided against using T-cells. Instead, they turned their attention to astrocytes. Astrocytes are star-shaped glial cells that perform a variety of essential functions, including providing structural support, regulating the blood-brain barrier, and maintaining the chemical environment around neurons. Unlike T-cells, which are visitors to the brain, astrocytes are permanent residents, making them an ideal candidate for long-term, localized therapy.
The Science of CAR-Astrocytes
The researchers utilized a harmless viral vector to deliver a specific gene into the brains of mice. This gene instructs the astrocytes to produce a Chimeric Antigen Receptor designed specifically to recognize amyloid-beta. Once these "CAR-astrocytes" are activated, they transform from passive support cells into active "cleaning" cells.
Under normal circumstances, the brain relies on microglia—the primary immune cells of the central nervous system—to clear cellular debris and misfolded proteins. However, in the presence of Alzheimer’s disease, microglia often become overwhelmed. They can enter a state of chronic inflammation or become "exhausted," losing their ability to effectively phagocytose (engulf and digest) amyloid plaques. By reprogramming astrocytes, the researchers have essentially recruited a massive, untapped workforce to assist the failing microglia.
The engineered CAR-astrocytes are designed to capture and "swallow" amyloid-beta proteins. Because astrocytes are the most numerous cells in the brain, this strategy creates a ubiquitous defense network capable of monitoring the entire organ for the presence of harmful aggregates.
Experimental Results: Prevention and Reduction
The study conducted at Washington University tested the efficacy of CAR-astrocytes in two distinct scenarios using mouse models genetically predisposed to developing Alzheimer’s-like pathology. These mice typically begin to show amyloid buildup by six months of age, with their brains becoming saturated with plaques shortly thereafter.
In the first group, the researchers administered the therapy to young mice before any plaques had formed. The results were definitive: the engineered astrocytes completely prevented the formation of amyloid deposits. By the time these mice reached six months of age—a point where untreated mice would show significant brain pathology—the treated mice remained entirely clear of detectable plaques.
The second group consisted of older mice that already possessed significant plaque burdens. This scenario more closely mirrors the condition of human patients who are diagnosed with Alzheimer’s after symptoms have already appeared. In these mice, a single injection of the CAR-astrocyte therapy reduced the existing amyloid levels by approximately 50 percent over a three-month period. This reduction is statistically significant and suggests that the therapy is capable of not only preventing the disease but also reversing existing damage.
A Potential Paradigm Shift in Clinical Care
The implications of a "single injection" therapy are profound. David M. Holtzman, MD, the Barbara Burton and Reuben M. Morriss III Distinguished Professor of Neurology and a co-author of the study, noted that while the therapy follows the same logic as antibody treatments—being most effective in early stages—its delivery method is a potential game-changer.
"Where it differs, and where it could make a difference in clinical care, is in the single injection that successfully reduced the amount of harmful brain proteins in mice," Holtzman stated. A single-dose regimen would eliminate the need for monthly hospital visits, reduce the long-term cost of treatment, and potentially lower the risk of systemic side effects associated with high-dose antibody infusions. Furthermore, because the astrocytes are native to the brain and the CAR expression is localized, the therapy may offer a more controlled and sustainable immune response than current methods.
Safety, Optimization, and Future Directions
While the results in mice are promising, the researchers emphasize that significant work remains before this can be tested in human clinical trials. One of the primary concerns in any CAR-based therapy is "off-target" effects—ensuring that the engineered cells do not attack healthy tissue or disrupt normal brain functions. Astrocytes play a critical role in neurotransmitter recycling and blood flow regulation; any modification must ensure these vital tasks are not compromised.
The team at Washington University is currently working to optimize the CAR design to improve the precision of the targeting mechanism. They are also investigating the longevity of the CAR expression. Because the therapy relies on a viral vector to modify the cells, it is crucial to determine how long the astrocytes remain in their "cleaning" state and whether the treatment would eventually need to be repeated or if it persists for the life of the patient.
Expanding the Horizon: From Alzheimer’s to Brain Tumors
The versatility of the CAR-astrocyte platform extends beyond neurodegenerative diseases. The researchers believe that by simply changing the "homing device" or the target of the receptor, they could redirect astrocytes to treat a variety of central nervous system disorders.
One of the most exciting potential applications is in the treatment of glioblastoma and other aggressive brain tumors. Brain tumors are notoriously difficult to treat because the blood-brain barrier prevents many chemotherapy drugs from reaching the site of the cancer. By engineering astrocytes to recognize tumor-specific markers, scientists could turn the brain’s own cellular architecture against the malignancy. Instead of clearing amyloid, these redirected astrocytes would be programmed to identify and destroy tumor cells directly.
Conclusion and Economic Impact
The study’s findings have already led to a patent filing through the Washington University Office of Technology Management, signaling the commercial and clinical potential of the discovery. As the global population ages, the economic and social burden of Alzheimer’s disease is expected to skyrocket. Current estimates suggest that without a significant breakthrough, the cost of caring for Alzheimer’s patients could exceed $1 trillion annually in the United States alone by 2050.
The development of a single-injection immunotherapy like the CAR-astrocyte approach offers a dual promise: a more effective biological intervention for the patient and a more sustainable economic model for healthcare systems. While the journey from laboratory mice to human pharmacy is long and fraught with regulatory and scientific hurdles, the work of Colonna, Holtzman, Chen, and their colleagues provides a clear and hopeful roadmap for the future of neurology. By turning the brain’s most common cells into its most powerful defenders, the medical community may finally be closing in on a way to not just slow, but potentially halt, the progression of neurodegenerative disease.














Leave a Reply