Indianapolis, IN – Eli Lilly and Company, a venerable pharmaceutical giant, has officially seized the top position in global pharmaceutical revenue, marking a historic moment for the company. This ascent coincides with a pivotal regulatory victory: the U.S. Food and Drug Administration (FDA) on April 1 approved Foundayo (orforglipron), Lilly’s novel once-daily oral GLP-1 receptor agonist designed for obesity management. Foundayo distinguishes itself as the first non-peptide oral GLP-1 approved for weight loss and achieved one of the fastest new molecular entity (NME) approvals in over two decades, cleared in just 50 days under the FDA’s Commissioner’s National Priority Voucher program. Despite these monumental achievements – claiming the revenue crown and securing a rapid, groundbreaking drug approval – Eli Lilly’s stock has experienced a notable decline, shedding nearly 14% since January 1, leaving market observers to ponder the disconnect between corporate success and investor sentiment.
A Rapid Regulatory Triumph for Foundayo
The approval of Foundayo represents a significant leap forward in the treatment landscape for obesity. Its expedited clearance, nearly 10 months ahead of its scheduled review date, underscores the urgent public health need for effective and accessible weight management solutions. The FDA’s National Priority Voucher program, designed to incentivize the development of treatments for neglected tropical diseases and medical countermeasures, inadvertently accelerated Foundayo’s path to market due to a voucher Lilly acquired. This approval marks the fastest NME clearance since 2002, highlighting both the drug’s potential and the regulatory agency’s commitment to addressing critical health challenges.
Foundayo’s mechanism as a GLP-1 receptor agonist mimics the action of a natural hormone that helps regulate appetite and blood sugar. What sets it apart is its oral, small-molecule formulation. Until recently, the dominant GLP-1 blockbusters, including Lilly’s Mounjaro and Zepbound, and Novo Nordisk’s Ozempic and Wegovy, have primarily been injectables. While Novo Nordisk did receive approval for an oral peptide formulation of Wegovy in December 2025, that drug requires strict adherence to an empty stomach and restricted water intake regimen. Foundayo, conversely, offers unparalleled convenience, allowing patients to take it at any time of day without food or water restrictions, a factor that could significantly enhance patient adherence and broaden its appeal.
Lilly CEO Dave Ricks has emphasized that while Foundayo may not achieve the same magnitude of weight loss as the company’s injectable tirzepatide (Zepbound), it excels in accessibility. Clinical trials, such as the ATTAIN-1 study, demonstrated an average weight loss of 12.4% over 72 weeks with the highest dose of Foundayo. This contrasts with the 20%-plus weight loss often observed with injectable tirzepatide. Ricks views Foundayo as a complementary offering within Lilly’s expanding portfolio, designed to meet diverse patient needs and preferences. "We want people to be on the medicine that meets their health goals," Ricks stated in a recent interview, adding, "If it has Lilly on the box, that’s the goal we have." This strategy aims to capture a wider segment of the obesity market by offering both highly potent injectable options and more convenient, albeit slightly less efficacious, oral alternatives.
Lilly’s Unprecedented Revenue Surge to the Top
The regulatory triumph for Foundayo arrives as Eli Lilly celebrates an extraordinary financial milestone. For the fiscal year 2025, the company reported a staggering $65.18 billion in revenue, representing a monumental 44.7% year-over-year growth. This surge propelled Lilly past industry titans Merck & Co. ($65.01 billion), Pfizer ($62.58 billion), and AbbVie ($61.16 billion) to claim the No. 1 spot in global pharmaceutical revenue for the first time in its history. While the margin over Merck was a slim $170 million, the stark difference in growth rates – Lilly’s 44.7% compared to Merck’s 1.3% – underscores the dynamism and momentum driving Lilly’s ascent.
This unprecedented financial performance is largely attributable to the explosive demand for its GLP-1 agonists, particularly Mounjaro (tirzepatide) for type 2 diabetes and Zepbound (tirzepatide) for obesity. Mounjaro alone generated $9.0 billion in the first half of 2025, marking a 90% increase year-over-year. Zepbound, which was only launched in late 2023 and is now available in a multi-dose KwikPen, rapidly contributed an additional $5.7 billion in H1 2025. Cumulatively, Lilly’s GLP-1 franchise is on track to exceed an annualized revenue run rate of $30 billion, showcasing its profound impact on the company’s top line.
This revenue growth has not been an overnight phenomenon but rather the culmination of strategic investments in research and development, particularly in the metabolic disease space. Lilly has been a long-standing leader in diabetes care, and its pivot and expansion into the obesity market with highly effective GLP-1s have proven to be a masterstroke. The company’s robust pipeline and commercial execution have allowed it to capitalize on the rapidly expanding global need for effective weight management and diabetes treatments.
The Exploding GLP-1 Market and Competitive Dynamics
The GLP-1 market is currently one of the most dynamic and lucrative segments in pharmaceuticals. From an estimated market size of under $12 billion in 2022, it has ballooned to an approximate $65 billion annualized run rate by mid-2025. Analysts project this market could surpass $100 billion by the end of the decade, driven by increasing awareness of obesity’s health risks, evolving treatment paradigms, and expanding patient access.
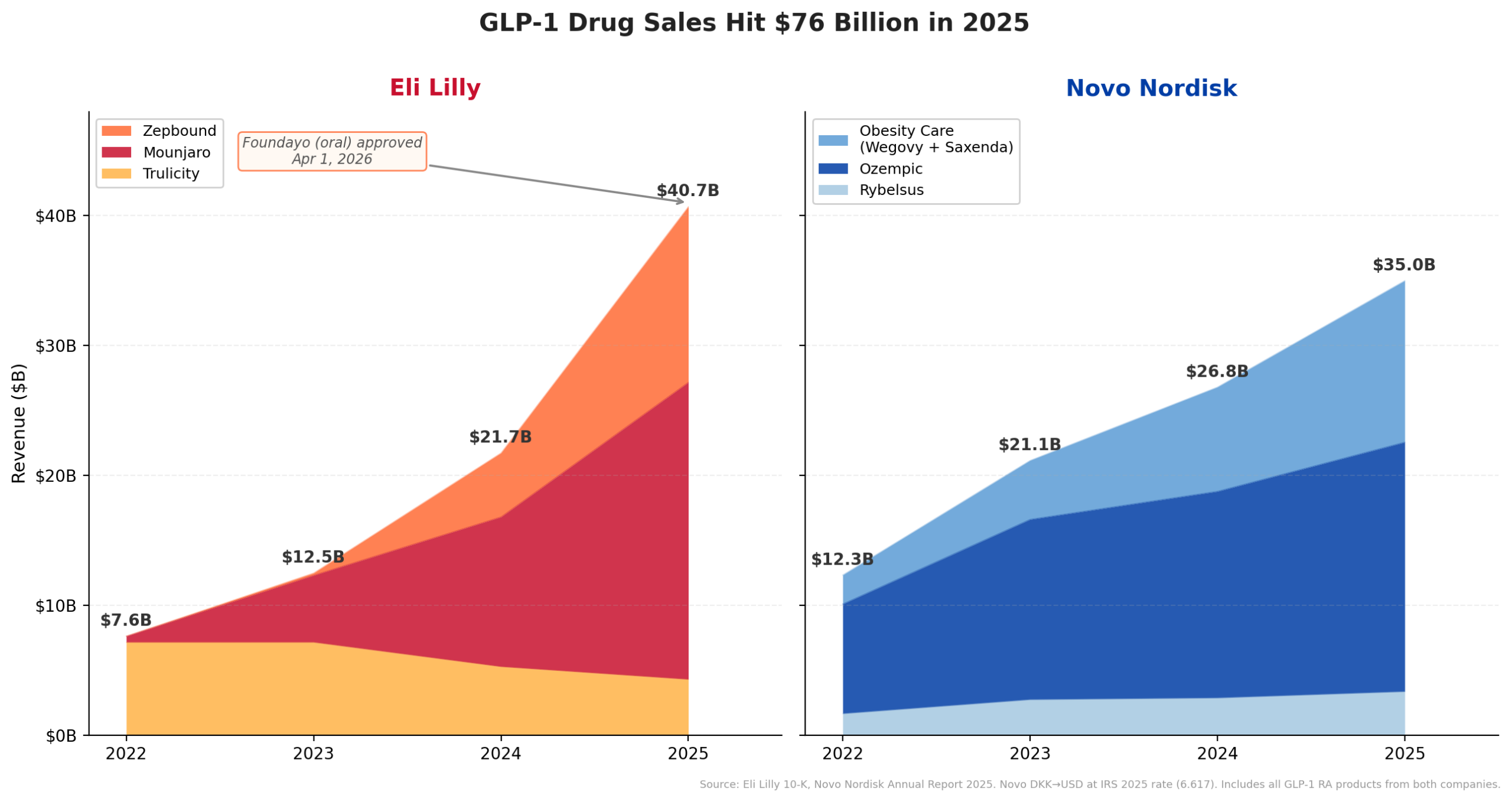
Lilly and Novo Nordisk are the primary architects of this market expansion, locked in an intense competitive battle. While Novo Nordisk pioneered the GLP-1 space with drugs like Ozempic and Wegovy, Lilly’s tirzepatide (Mounjaro/Zepbound) has offered a compelling alternative, demonstrating superior efficacy in some studies due to its dual GIP/GLP-1 agonism. The introduction of oral formulations adds another critical dimension to this rivalry. Foundayo’s unique small-molecule structure and flexible dosing regimen position it as a strong contender against Novo Nordisk’s oral peptide Wegovy, potentially appealing to a broader patient population due to its convenience.
The strategic importance of an oral option cannot be overstated. Oral medications traditionally boast higher patient acceptance and adherence rates compared to injectables, removing psychological barriers associated with needles. This enhanced accessibility could significantly expand the addressable population for GLP-1 therapies, drawing in individuals who might be hesitant to start or continue injectable treatments. Lilly’s proactive global strategy is evident in its submission of orforglipron for approval in over 40 countries, signaling its intent to establish Foundayo as a worldwide standard for oral obesity management.
Wall Street’s Skepticism: A Deeper Look
Despite Lilly’s unprecedented success and the promising outlook for Foundayo, the company’s stock performance since January 1 has presented a puzzle for many. A nearly 14% decline in its share price suggests that investors are not celebrating these victories with the expected enthusiasm. Several factors could be contributing to Wall Street’s measured response:
- "Buy the Rumor, Sell the News" Phenomenon: Much of Lilly’s impressive growth and pipeline potential, particularly regarding its GLP-1 drugs, has been priced into its stock over the past year. Investors may have already factored in these successes, leading to profit-taking or a re-evaluation once the news is officially released.
- High Valuation Concerns: Lilly’s stock has traded at a premium, reflecting its robust growth trajectory. Some analysts may view the current valuation as stretched, making it vulnerable to corrections even on positive news. Any slight perceived hurdle or competitive threat can trigger a re-assessment.
- Future Competition and Pipeline Pressure: While Lilly leads, the GLP-1 market is attracting intense R&D from other pharmaceutical companies. Investors may be looking ahead to potential new entrants or next-generation therapies that could challenge Lilly’s dominance in the long term.
- Manufacturing and Supply Chain Challenges: The immense demand for GLP-1s has strained manufacturing capabilities across the industry. Investors might be concerned about Lilly’s ability to scale production sufficiently to meet the anticipated global demand for Foundayo and its injectable counterparts, which could impact sales projections.
- Pricing and Reimbursement Pressures: As GLP-1s become more prevalent, healthcare payers and governments are likely to exert increasing pressure on pricing and reimbursement. This could cap future revenue growth, especially for oral formulations that might be positioned as more affordable entry points.
- Broader Market Dynamics: General market volatility or shifts in investor preference away from high-growth biotech stocks could also play a role, regardless of individual company performance.
Lilly’s licensing agreement for orforglipron from Japanese drugmaker Chugai in 2018 for a modest $50 million upfront now appears to be a shrewd investment, but the market might be weighing the substantial R&D and commercialization costs that followed. Investors are likely scrutinizing the ramp-up strategy for Foundayo, its market penetration against existing options, and its ultimate contribution to the bottom line, alongside ongoing performance of Mounjaro and Zepbound.
Broader Impact and Strategic Implications
The approval of Foundayo and Lilly’s rise to the top of the pharmaceutical industry have profound implications for patients, healthcare systems, and the competitive landscape.
For patients, Foundayo offers a new, convenient, and potentially more acceptable pathway to effective weight management. This could significantly increase the number of individuals seeking and adhering to treatment, addressing a critical public health crisis. The choice between injectable and oral, and between different efficacy profiles, empowers patients and physicians to tailor treatments more precisely to individual needs and lifestyles.
For healthcare systems, the increased accessibility of oral GLP-1s could lead to broader adoption, necessitating careful consideration of cost-effectiveness and reimbursement policies. While the convenience factor is a major plus, the long-term cost implications of widespread use of these highly effective drugs will remain a key discussion point.
Strategically, Foundayo strengthens Lilly’s already formidable position in the metabolic disease space. It provides a crucial oral complement to its injectable blockbusters, creating a comprehensive portfolio that can address various segments of the obesity and diabetes markets. This multi-pronged approach enhances Lilly’s competitive moat against rivals like Novo Nordisk and other companies developing GLP-1 and next-generation obesity drugs. By offering different administration methods and efficacy profiles, Lilly aims to capture market share across the spectrum of patient preferences and clinical needs, further cementing its leadership.
Looking ahead, Lilly faces the dual challenge of sustaining its incredible growth while navigating increased competition and potential pricing pressures. Its ability to scale manufacturing, secure broad market access and reimbursement for Foundayo globally, and continue innovating beyond its current GLP-1 portfolio will be critical to maintaining its top-tier status. The initial skepticism from Wall Street, despite the company’s clear successes, serves as a reminder that the pharmaceutical market is forward-looking, constantly evaluating future risks and opportunities beyond immediate victories. Eli Lilly’s journey to the pinnacle of the industry is a testament to its innovation, but the path ahead remains dynamic and demanding.














Leave a Reply