For decades, the prevailing scientific consensus painted a picture of the genome within a newly fertilized egg as a chaotic, unstructured entity. Researchers widely believed that this genetic material, the blueprint for all life, existed as a loose, disorganized bundle – a "blank slate" – awaiting the embryo’s burgeoning genetic activity to impose order. This traditional viewpoint posited that the genome remained largely a dishevelled collection of DNA until it "woke up," initiating its complex developmental program. However, groundbreaking new research, published in the prestigious journal Nature Genetics, has fundamentally challenged this long-held assumption, revealing an unexpected and intricate level of organization present from the very earliest stages of embryonic development.
Unveiling the Early Genome: A Technological Breakthrough
Professor Juanma Vaquerizas and his dedicated team have spearheaded this paradigm-shifting discovery. Their work hinges on the development of a revolutionary new technology, christened Pico-C. This sophisticated method empowers scientists to examine the three-dimensional (3D) structure of the genome with unprecedented detail and resolution. Employing this cutting-edge approach, the researchers observed that long before the crucial milestone of Zygotic Genome Activation (ZGA) – the point at which the embryo’s own genes begin to drive development – an elaborate and highly organized 3D scaffold of DNA is already meticulously taking shape.
"We used to think of the time before the genome awakens as a period of chaos," explained Noura Maziak, the lead author of the study, articulating the previous scientific dogma. "But by zooming in closer than ever before, we can see that it’s actually a highly disciplined construction site. The scaffolding of the genome is being erected in a precise, modular way, long before the ‘on’ switch is fully flipped." This statement underscores the profound shift in understanding, moving from a notion of primordial disorder to one of pre-ordained architectural precision.
The Significance of Early DNA Architecture
The implications of this early folding pattern extend far beyond mere structural curiosity. The spatial arrangement of DNA is a critical determinant of gene regulation. In essence, how DNA is folded and organized in three dimensions dictates which genes are accessible and, consequently, which genes can be switched on or off during development. This intricate control mechanism is not only fundamental for proper cell function but also plays a vital role in preventing a host of developmental abnormalities and diseases. Disruptions in this finely tuned architecture can have cascading negative effects, leading to cellular dysfunction and potentially severe health consequences.
Pico-C Technology: Revolutionizing Genome Mapping in Fruit Flies
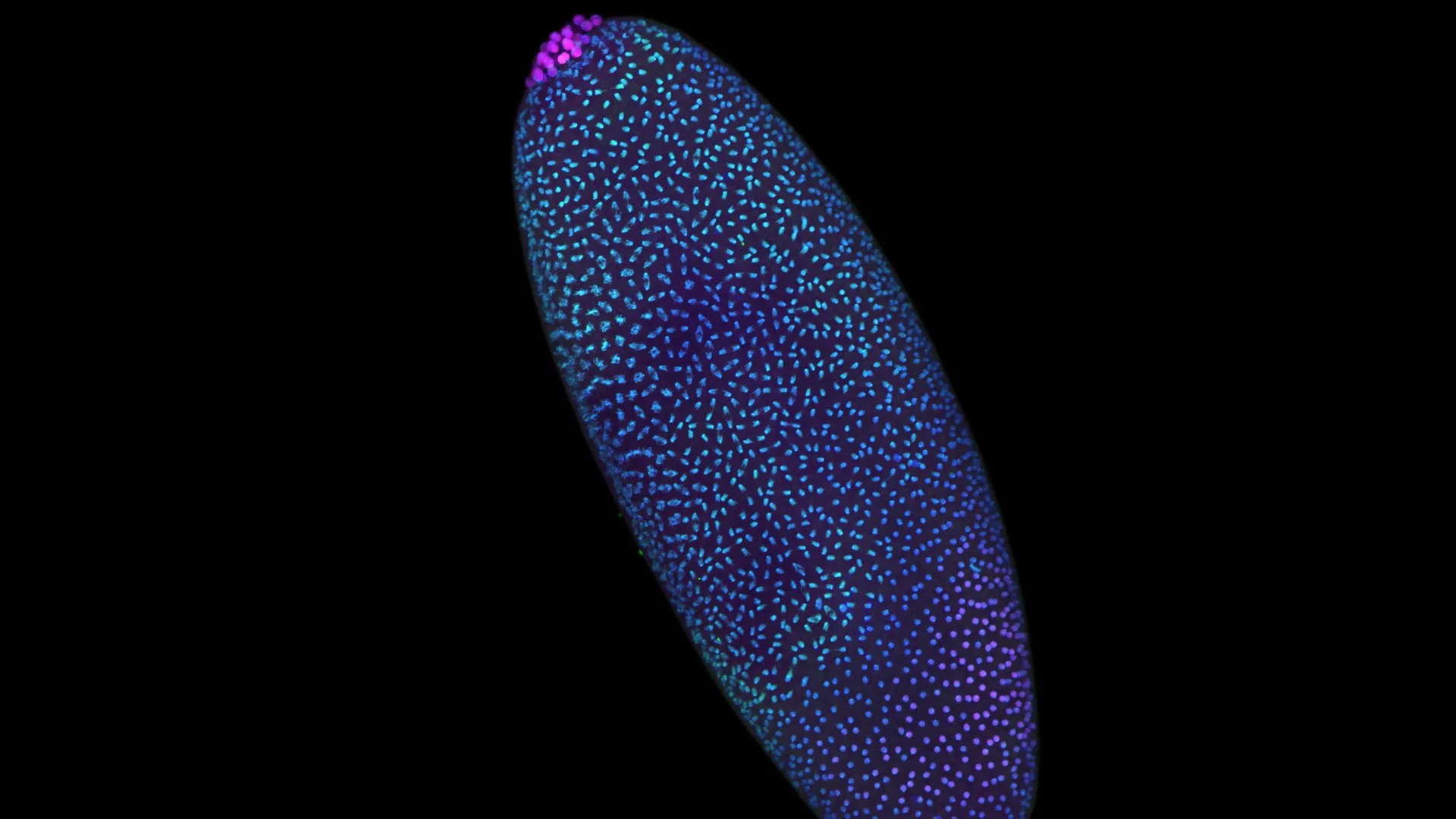
The pivotal discovery was achieved through meticulous experimentation on the fruit fly, Drosophila melanogaster. This organism is a cornerstone of genetic research, widely utilized due to its rapid reproductive cycle and relatively simple yet conserved genetic mechanisms. In the initial hours following fertilization, a fruit fly embryo undergoes rapid nuclear divisions, generating thousands of cells in a remarkably short period. This accelerated developmental pace makes it an ideal model system for dissecting the complex processes of genome organization and regulation.
Using their highly sensitive Pico-C method, the research team was able to precisely map the 3D arrangement of the fruit fly genome during these critical early hours. Their findings revealed a sophisticated modular pattern of DNA looping and folding. This architecture is not random; it is designed to allow different regulatory signals to exert their influence on specific, targeted regions of the genome. This intricate spatial organization ensures that genetic information is not only stored but also strategically positioned and prepared for activation precisely at the moment it is required for development.
The Pico-C technology offers a significant advantage in terms of sample efficiency. It requires approximately ten times less genetic material than conventional techniques, making it possible to conduct investigations that were previously infeasible. This increased efficiency and sensitivity allow for a more granular examination of how DNA folding influences gene regulation and, crucially, how alterations in this architecture might contribute to the development of various diseases.
A Companion Study: The Consequences of Architectural Collapse in Human Cells
While the initial structural "blueprint" was identified in fruit flies, its fundamental relevance extends deeply into human biology. In a parallel and complementary study, published concurrently in Nature Cell Biology, researchers led by Professor Ulrike Kutay and collaborators at ETH Zürich in Switzerland applied the same high-resolution mapping strategy to human cells. This international collaboration aimed to explore the consequences of disrupting the genome’s 3D structure in a mammalian context.
The human cell study focused on the molecular ‘anchors’ responsible for stabilizing the genome’s intricate 3D architecture. When these critical stabilizing elements were experimentally removed, the findings were nothing short of striking. The researchers observed that the human cells interpreted the breakdown of this structural framework as an invasion, akin to a viral attack. This misinterpretation triggered the cell’s innate immune system, activating a cascade of inflammatory responses. Such a false alarm, stemming from a compromised genome architecture, can have profound implications for health, potentially leading to chronic inflammation and various disease states.
A Unified Narrative: From Genesis to Disease
Professor Vaquerizas summarized the profound synergy between the two studies, stating, "These two studies tell a complete story. The first shows us how the genome’s 3D structure is carefully built at the start of life. The second shows us the disastrous consequences for human health if that structure is allowed to collapse." This powerful statement encapsulates the unified narrative that emerges from this research: the intricate, pre-programmed architecture of the genome is not merely a passive structure but an active participant in early development, and its integrity is paramount for long-term health.
The research was made possible through crucial funding from the Medical Research Council and the Academy of Medical Sciences (AMS), including an AMS Professorship award, highlighting the significant investment in advancing fundamental biological understanding.
Broader Implications and Future Directions
This research represents a significant leap forward in our understanding of developmental biology and epigenetics. The discovery of pre-programmed genome architecture challenges the simplistic "blank slate" model and suggests a far more dynamic and orchestrated process unfolding from the moment of conception.
Key implications include:
- Rethinking Early Development: The findings necessitate a re-evaluation of the earliest stages of embryonic development, emphasizing the critical role of physical organization in genetic regulation. This could lead to new hypotheses about how developmental pathways are initiated and controlled.
- Disease Etiology: The link established between genome architectural collapse and the activation of the innate immune system in human cells opens new avenues for understanding the origins of inflammatory diseases, autoimmune disorders, and potentially even certain types of cancer. If structural integrity is compromised, the body’s own defense mechanisms can be misdirected, causing harm.
- Therapeutic Potential: A deeper understanding of genome architecture could pave the way for novel therapeutic interventions. Strategies aimed at restoring or maintaining the correct 3D folding of DNA might offer new ways to combat diseases linked to genetic misregulation or epigenetic instability.
- Advancements in Reproductive Health: For conditions involving infertility or recurrent miscarriage, understanding the precise organizational requirements of the early genome could offer critical insights into causes and potential treatments.
- Technological Advancement: The Pico-C technology itself represents a significant advancement, enabling a host of new research possibilities across various fields of biology. Its efficiency and precision will likely drive further discoveries in genomics and epigenomics.
Timeline of Discovery (Inferred):
While specific dates for the research phases are not provided, the typical progression of such scientific endeavors can be inferred:
- Years Prior: Initial development and refinement of the Pico-C technology. This would involve extensive pilot studies and optimization.
- Recent Years: Execution of the fruit fly experiments, mapping the 3D genome structure at early developmental stages. This phase would involve data acquisition, analysis, and initial interpretation.
- Concurrent Period: The companion study on human cells was conducted, utilizing the developed methodology to investigate the impact of structural disruption.
- Publication: The findings from both studies were peer-reviewed and published in leading scientific journals (Nature Genetics and Nature Cell Biology), marking the formal dissemination of the research to the scientific community.
- Ongoing: Further research building upon these findings, exploring the precise mechanisms of architectural assembly and collapse, and investigating their links to specific diseases.
Supporting Data and Future Research:
The current research provides a foundational understanding. Future studies will likely focus on quantifying the extent of DNA folding, identifying the specific proteins and molecular machinery involved in establishing and maintaining the genome’s 3D scaffold, and cataloging the range of genes affected by different architectural configurations. Quantitative data on the degree of DNA looping, the size of chromatin domains, and the frequency of specific gene-promoter interactions within these loops would further solidify the findings. Additionally, comparative studies across different species and developmental stages will be crucial to understand the evolutionary conservation and plasticity of genome architecture.
Official Reactions and Broader Scientific Community Response:
While direct quotes from external parties are not available in the provided text, it is highly probable that the scientific community has reacted with significant interest and excitement. Research that fundamentally challenges long-held paradigms, especially in a field as central as developmental biology, typically generates considerable discussion and spurs further investigation. Funding bodies such as the Medical Research Council and the Academy of Medical Sciences would likely view this as a successful investment, demonstrating the value of supporting innovative research. The publication in high-impact journals like Nature Genetics and Nature Cell Biology indicates a strong endorsement from expert reviewers within the field.
In conclusion, the work by Professor Vaquerizas and his colleagues, bolstered by the parallel study on human cells, represents a monumental shift in our understanding of the earliest moments of life. It reveals that the genome is not a passive recipient of order but an actively architected entity from its inception, a revelation that promises to reshape developmental biology and offer new insights into the origins of disease and the potential for novel therapeutic strategies.















Leave a Reply