Every second, a silent, intricate dance of electrical charges unfolds within the human body, a microscopic ballet fundamental to life itself. These imperceptible signals orchestrate communication between our cells, fuel the intricate machinery of energy production, and underpin the complex processes of metabolism. Without the precise and meticulously controlled migration of charges across cellular membranes and within the cellular milieu, none of this vital activity would be possible. In essence, charge transport serves as a foundational control system for all biological functions. At the heart of many of these crucial charge-carrying mechanisms are phosphoric acid (H₃PO₄) and its related molecular cousins. These ubiquitous compounds are not merely present in living systems; they are integral to their very fabric. They form the backbone of our genetic code as key components of DNA and RNA, contribute to the structural integrity of cell membranes, and are central to the function of adenosine triphosphate (ATP), the universal cellular currency for energy storage and transfer. Their profound importance extends beyond the biological realm, with phosphoric acid’s exceptional proton conductivity making it a vital player in advanced technologies such as modern batteries and sophisticated fuel cells.
The Enigmatic Journey of Protons: Unraveling Proton-Shuttling
The mechanism by which protons, the positively charged nuclei of hydrogen atoms, traverse phosphate-containing materials is a phenomenon of remarkable elegance and efficiency, often described as "proton-shuttling." Unlike the free, unimpeded movement of charges in many conductive materials, protons in these systems embark on a journey of molecular leaps, hopping sequentially from one phosphate molecule to another. This intricate relay race is facilitated by a network of hydrogen bonds, which act as pre-defined pathways, guiding the proton’s directional movement. This proton-shuttling process allows for an astonishingly rapid transport of positive charge, a speed that has long fascinated scientists.
While the overarching principle of proton-shuttling has been understood for decades, the precise molecular choreography—the detailed, step-by-step mechanics of this charge transfer—has remained an elusive puzzle. This lack of granular understanding has presented a significant hurdle in fully harnessing the potential of these materials for both biological and technological applications.
A Deep Dive into the Molecular Mechanism: The Fritz Haber Institute’s Groundbreaking Investigation
Addressing this knowledge gap, a collaborative effort spearheaded by researchers from the Department of Molecular Physics at the prestigious Fritz Haber Institute of the Max Planck Society, in conjunction with esteemed colleagues from Leipzig University and institutions in the United States, embarked on an ambitious quest. Their singular focus was to meticulously dissect the very earliest stages of proton transfer within a critical molecular architecture implicated in this process. The primary objective was to definitively identify and characterize the initial molecular events that trigger and sustain the proton-shuttling phenomenon.
Illuminating the Unseen: Extreme Cold as a Scientific Microscope
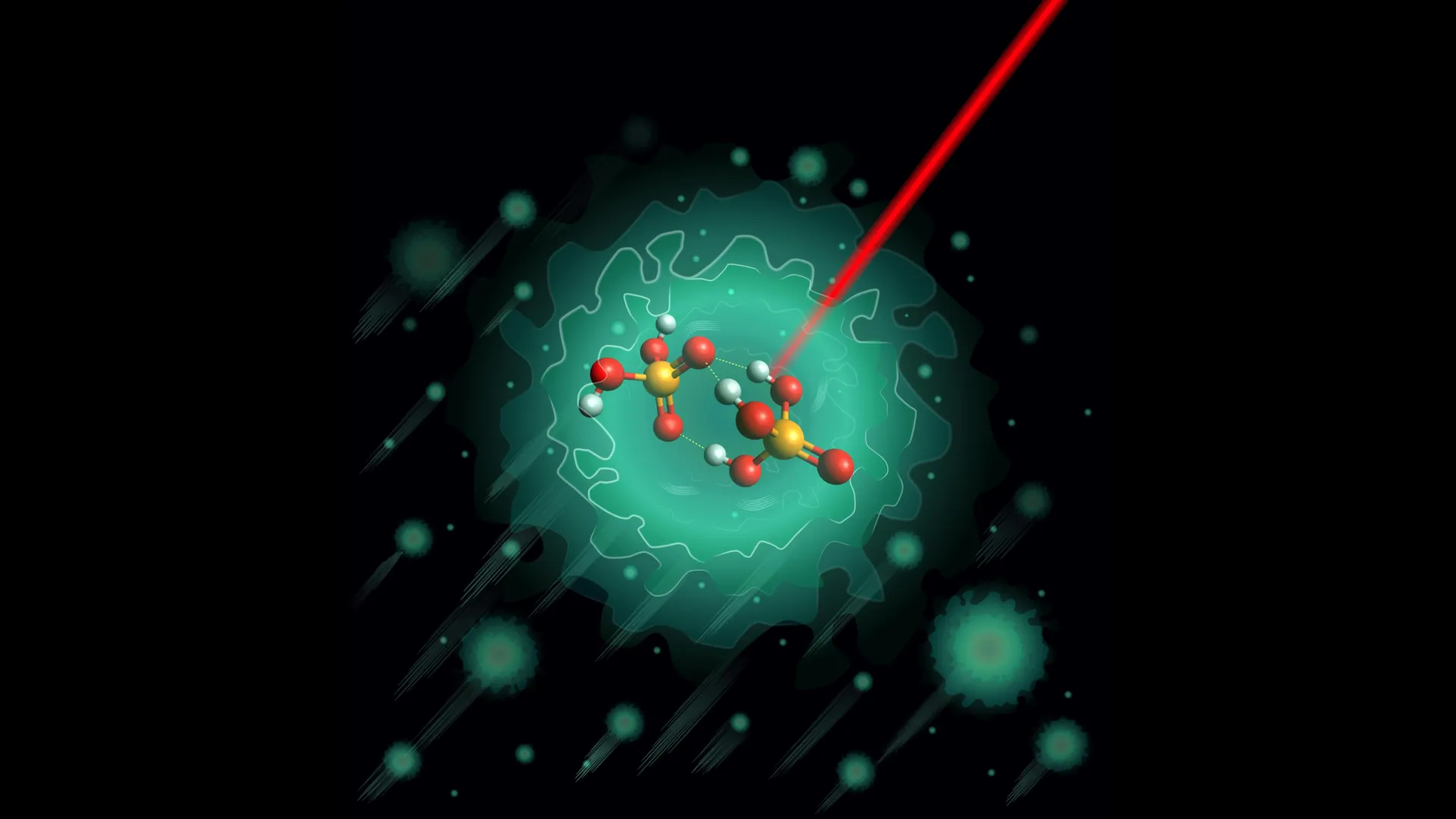
Previous theoretical investigations had posited that a specific, negatively charged molecular species might serve as the initiator of the proton-shuttling cascade. This hypothetical precursor molecule, identified as the deprotonated dimer of phosphoric acid, H₃PO₄·H₂PO₄⁻, became the central subject of the Fritz Haber Institute’s investigation.
To bring this elusive molecule under intense scientific scrutiny, the researchers devised a sophisticated experimental setup. They meticulously synthesized the H₃PO₄·H₂PO₄⁻ dimer in the laboratory. The critical next step involved cooling this molecule to an unprecedentedly low temperature. By encapsulating the dimer within a superfluid helium nanodroplet, the scientists were able to achieve temperatures as low as 0.37 degrees above absolute zero (0.37 K). At such extreme cryogenic conditions, thermal vibrations and other random molecular disturbances, which typically obscure subtle molecular behaviors, are virtually eliminated. This near-absolute stillness provided an unparalleled environment for high-precision structural analysis.
The analytical tool of choice for this ultra-cold investigation was infrared (IR) spectroscopy. This technique probes the vibrational modes of molecules, providing a unique fingerprint that can reveal detailed information about their structure, bonding, and dynamics. The experimental data gathered through IR spectroscopy was then synergistically combined with advanced quantum chemical calculations. These computational methods are indispensable for predicting molecular arrangements, electronic structures, and the energetics of chemical processes. By integrating the empirical evidence from spectroscopy with the predictive power of quantum chemistry, the research team achieved a level of molecular insight that neither approach could have furnished independently. This dual-pronged strategy offered a far more comprehensive and accurate portrayal of the molecule’s behavior than previously attainable.
A Singular Structure Emerges from Theoretical Ambiguity
The results emanating from this rigorous investigation yielded a surprising and significant revelation. Prior theoretical models had suggested that the H₃PO₄·H₂PO₄⁻ dimer could exist in at least two distinct, energetically comparable structural configurations. These models predicted a degree of structural flexibility, implying that the molecule could readily transition between these forms. However, the experimental data, meticulously collected at cryogenic temperatures, painted a dramatically different picture. Instead of observing a mixture of structures or evidence of rapid interconversion, the researchers found compelling evidence for the existence of only one stable, well-defined structural configuration.
This single, dominant structure, as elucidated by the combined experimental and computational findings, is characterized by a remarkable degree of rigidity. It features a specific arrangement of three robust hydrogen bonds, intricately linked through a central, shared oxygen atom. Furthermore, this particular structural arrangement presents significant energetic barriers to proton movement within the molecule. These barriers, while not entirely preventing proton transfer, dictate a more controlled and perhaps slower initial step compared to what might have been anticipated from more fluid theoretical models.
The implications of this finding are substantial. The observed bonding pattern—the specific way hydrogen bonds are formed and linked—bears a striking resemblance to configurations previously identified in other clusters of phosphoric acid molecules. This similarity strongly suggests that this rigid, well-defined structural arrangement may not be an anomaly but rather a prevalent and recurring structural motif within larger phosphoric acid assemblies.
This discovery also underscores a crucial point in scientific inquiry: even the most sophisticated theoretical models, while invaluable for generating hypotheses and guiding research, are not infallible. They can, as demonstrated in this case, sometimes overlook or misinterpret key structural details that are only revealed through precise experimental verification. The need for experimental validation remains paramount in solidifying our understanding of complex molecular phenomena.
The Far-Reaching Implications of Pinpointing Nature’s Proton Highway
The significance of this research extends across multiple scientific and technological frontiers, offering profound insights into the molecular underpinnings of phosphoric acid’s extraordinary proton conductivity, a phenomenon aptly termed "Nature’s proton highway." By definitively identifying a single, stable, and well-characterized structure for the key anionic dimer, H₃PO₄·H₂PO₄⁻, scientists now possess a much clearer and more accurate picture of how protons initiate their journey through these critical systems.
This breakthrough has direct and immediate consequences for the field of computational chemistry. The detailed experimental data provides an invaluable benchmark against which to refine and improve existing quantum chemical models specifically designed for phosphate-based molecules. More accurate models will, in turn, accelerate the discovery and design of novel materials with enhanced proton conductivity. Such materials are of paramount importance for the advancement of clean energy technologies, particularly in the development of more efficient and durable fuel cells. The ability to precisely control and optimize proton transport is a cornerstone of fuel cell technology, enabling the conversion of chemical energy into electrical energy with minimal environmental impact.
Beyond technological applications, this research deepens our fundamental understanding of biological processes. The efficient transfer of protons is not merely a feature of inorganic chemistry or material science; it is a fundamental requirement for countless biochemical reactions that sustain life. By unraveling the precise mechanisms of proton transfer in a well-defined model system like phosphoric acid, scientists gain crucial insights into how similar processes might occur within the complex molecular environments of biological cells. This knowledge could pave the way for a more nuanced understanding of cellular energy metabolism, signal transduction, and the development of new therapeutic strategies targeting diseases where cellular charge transport is dysregulated.
The journey to this discovery, while rooted in recent experimental work, is part of a long-standing scientific endeavor to comprehend the fundamental forces that govern matter and energy. The understanding of charge transport in biological systems dates back to the early 20th century, with pioneers like Archibald Hill studying muscle contraction and the role of energy transfer. The identification of ATP as the primary energy currency in the 1920s and 1930s further solidified the importance of molecular energy dynamics. The development of sophisticated spectroscopic techniques and computational methods in the latter half of the 20th century and into the 21st century has enabled researchers to probe these phenomena at an unprecedented level of detail. The work at the Fritz Haber Institute represents a significant leap forward within this continuum, providing a crucial piece of the puzzle that has eluded scientists for years.
This meticulous investigation not only clarifies a fundamental aspect of molecular behavior but also serves as a testament to the power of interdisciplinary research and the unwavering pursuit of scientific understanding. The ability to manipulate and observe molecules at extreme temperatures, combined with the predictive power of theoretical calculations, opens new avenues for exploration and innovation, promising to shape the future of both biology and technology.
Future Directions and Broader Impact
The implications of this research are multifaceted and extend to several key areas:
- Materials Science and Engineering: The precise understanding of proton-shuttling mechanisms in phosphoric acid systems will directly inform the design of next-generation materials for fuel cells, advanced battery electrolytes, and proton-exchange membranes. The ability to engineer materials with optimized proton conductivity could lead to more efficient and cost-effective energy storage and conversion devices.
- Biochemistry and Molecular Biology: This work provides a crucial molecular blueprint for understanding proton transfer in biological contexts. Many enzymes and cellular processes rely on the precise movement of protons. Further research could illuminate how these fundamental principles are applied in biological systems, potentially leading to new insights into diseases and novel therapeutic interventions.
- Computational Chemistry: The experimental validation of a single, stable structure for H₃PO₄·H₂PO₄⁻ offers a critical data point for refining and improving computational models. This will enable more accurate predictions for a wider range of phosphate-containing systems, accelerating discovery in various fields.
- Fundamental Physics and Chemistry: The study delves into the intricate interplay of hydrogen bonding, charge distribution, and molecular structure at the quantum level. This contributes to our foundational knowledge of chemical bonding and intermolecular forces.
In conclusion, the research conducted by scientists at the Fritz Haber Institute and their collaborators represents a significant advancement in our understanding of charge transport. By meticulously investigating the H₃PO₄·H₂PO₄⁻ dimer at ultra-low temperatures, they have unveiled a fundamental molecular structure and clarified the initial steps of proton-shuttling. This discovery not only enriches our comprehension of biological and chemical processes but also holds immense promise for driving innovation in critical technological fields. The "Nature’s proton highway" is now better illuminated, paving the way for future discoveries and applications that could profoundly impact our world.















Leave a Reply