Researchers at Oregon Health & Science University (OHSU) have unveiled a previously unrecognized internal cellular system that functions akin to dynamic "trade winds," actively and rapidly transporting vital proteins to the cell’s leading edge. This groundbreaking discovery fundamentally alters the scientific paradigm of cell motility, with profound implications for understanding cancer metastasis, wound healing, and a spectrum of cellular processes. The findings, meticulously detailed in the esteemed journal Nature Communications, challenge long-held assumptions about intracellular organization and protein delivery mechanisms.
For decades, the prevailing scientific model depicted intracellular protein movement as a largely passive and random phenomenon known as diffusion. This textbook explanation posited that proteins drifted throughout the cell, eventually reaching their intended destinations by chance. However, the OHSU study provides compelling evidence that cells are far more sophisticated, employing directed fluid flows to proactively propel proteins to the precise locations where they are needed for critical functions such as extension, migration, and tissue regeneration. This active transport system suggests a level of cellular intentionality and efficiency previously underestimated.
From Classroom Curiosity to Cellular Revolution
The genesis of this monumental discovery can be traced to an unexpected observation during a routine neurobiology course at the prestigious Marine Biological Laboratory in Woods Hole, Massachusetts. Catherine (Cathy) Galbraith, Ph.D., and James (Jim) Galbraith, Ph.D., co-corresponding authors of the study, were engaged in a standard laboratory experiment with students when they noticed an anomaly that would ultimately redefine cellular transport.
"It actually started out as an unexpected finding," Cathy Galbraith recounted, emphasizing the serendipitous nature of the breakthrough. "We were just conducting an experiment with students in class." The experiment involved employing a laser to temporarily render a strip of proteins at the rear of a living cell invisible, a common technique for tracking intracellular transport dynamics. During this observation, the researchers were astonished to witness the spontaneous appearance of an additional, darkened band at the cell’s anterior edge – the very region that actively extends and drives cellular locomotion.
"We kind of did it for fun and then realized this gave us a way of measuring something that wasn’t able to be measured before," she added, highlighting the immediate recognition of the experiment’s potential.
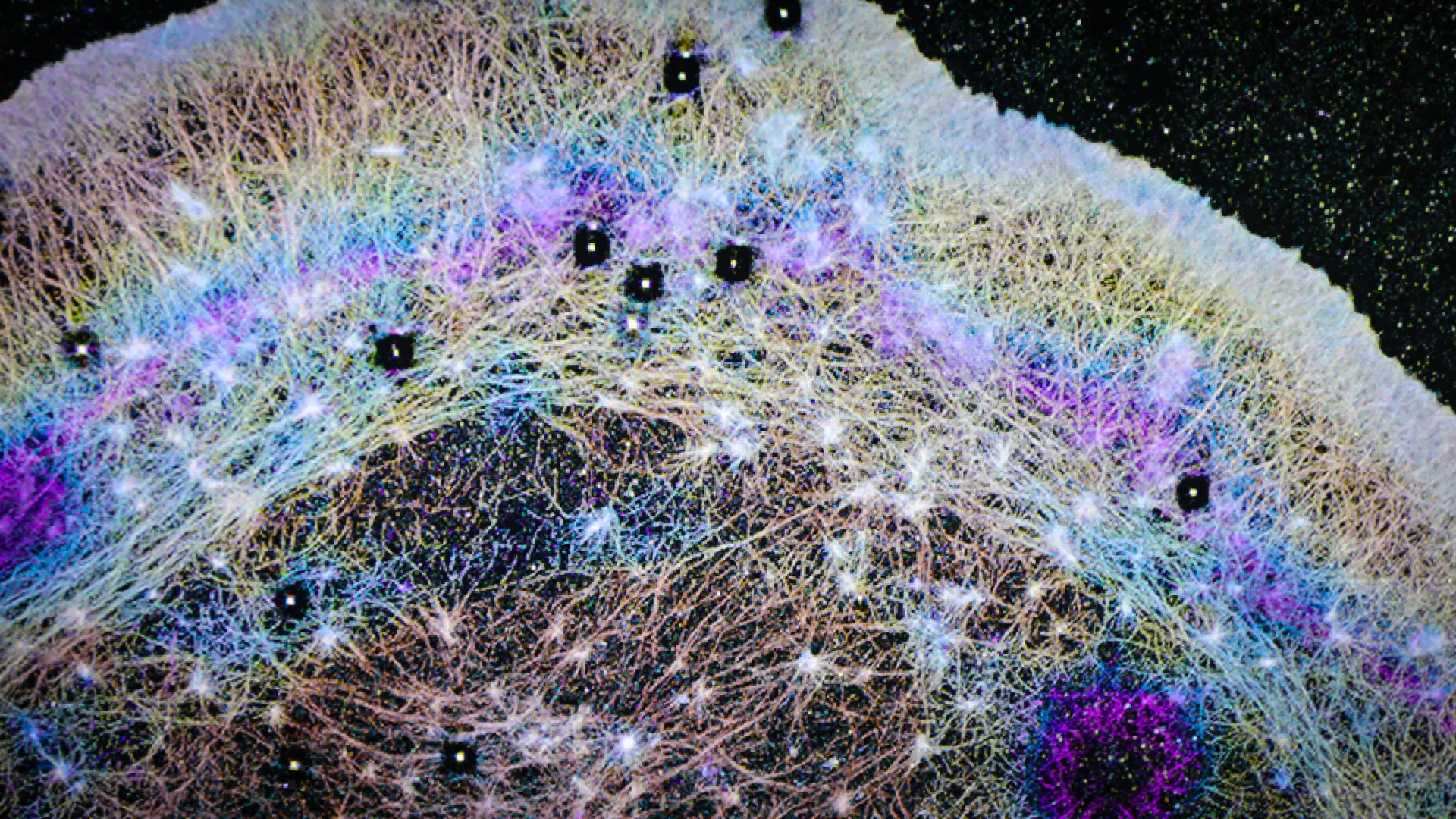
Subsequent in-depth investigations revealed that this anomalous dark band was not a random occurrence but rather a visible manifestation of a wave of soluble actin, a critical protein indispensable for cell movement, being rapidly propelled forward. This directly contradicted the prevailing scientific consensus that actin primarily reached these leading-edge regions through gradual, random diffusion. The new findings illuminated a distinctly different, active mechanism at play.
"We realized the cartoon models in textbooks were missing a huge piece," Jim Galbraith stated, underscoring the profound shift in understanding. "There had to be some kind of flow in the cell pushing things forward. Cells really do ‘go with the flow.’" This evocative phrasing captures the essence of the newly identified system, suggesting a cellular environment far more dynamic and orchestrated than previously imagined.
The Dynamics of Directed Cellular Currents
The Galbraiths, who joined OHSU in 2013 following their tenure at the National Institutes of Health, brought with them a wealth of expertise in advanced imaging techniques. Their prior collaboration with Nobel Laureate Eric Betzig, Ph.D., at the Howard Hughes Medical Institute’s Janelia Research Campus had equipped them with cutting-edge tools and methodologies crucial for visualizing cellular processes at unprecedented resolution.
Leveraging these specialized imaging technologies, the OHSU team was able to conclusively demonstrate that cells actively generate directional fluid flows. The researchers drew a compelling analogy between these internal cellular currents and atmospheric rivers, vast aerial currents that transport enormous quantities of water vapor across continents. These cellular "rivers" effectively transport actin and a multitude of other essential proteins toward the cell’s front, achieving speeds significantly exceeding what diffusion alone could accomplish.
"We found that the cell can actually squeeze at the back and target where it sends that material," Jim Galbraith explained, further elaborating on the mechanism. "If you squeeze half a sponge, the water only goes on that half. That’s basically what the cell is doing." This analogy vividly illustrates the controlled and targeted nature of the fluid flow, emphasizing the cell’s ability to direct its internal resources with remarkable precision.
Crucially, these newly identified flows are characterized by their nonspecificity, meaning they possess the capacity to carry a diverse array of proteins simultaneously. This inherent versatility establishes a highly efficient transport network that underpins a spectrum of essential cellular activities, including the formation of cellular protrusions, adhesion to surfaces, and rapid shape alterations. These dynamic processes are fundamental for cell migration, the intricate workings of the immune system, and the complex process of tissue repair.
Furthermore, the research elucidated the existence of a specialized compartment at the anterior of the cell where these directed flows are concentrated. This region is demarcated from the rest of the cell by a sophisticated actin-myosin condensate barrier, a dynamic structure that acts as a physical boundary, effectively channeling proteins towards the advancing edge of the cell. This compartmentalization ensures that the directed transport system operates with maximum efficiency and precision.
Visualizing the Invisible: Innovations in Cellular Imaging
To achieve their remarkable observations, the OHSU team developed a sophisticated modification of a standard fluorescence microscopy technique. Instead of the conventional method of using a laser to quench fluorescence, they ingeniously activated fluorescent molecules at a single point and meticulously tracked their subsequent movement. This innovative approach allowed them to visualize and quantify the internal fluid dynamics within the cell.
One of the pivotal experiments developed by the team was aptly named FLOP, an acronym for Fluorescence Leaving the Original Point. Cathy Galbraith humorously remarked on the name’s initial connotations, stating, "It wasn’t a flop at all. It was the opposite. It is anything but a flop, because it worked." The success of FLOP and related experiments paved the way for a deeper understanding of cellular mechanics and has significant implications for various biological fields.
Implications for Cancer Cell Aggression and Metastasis
The discovery of these rapid protein transport systems holds particularly significant promise for unraveling the complex mechanisms underlying cancer cell migration and metastasis. The aggressive and invasive nature of certain cancer cells may be directly attributable to their enhanced utilization of these directed cellular currents.
"We know these highly invasive cells have this really cool mechanism to push proteins really fast, really rapidly where they need them at the front of the cell," Jim Galbraith observed, drawing a parallel between cellular components and mechanical systems. "All cells have basically the same components inside, much like a Porsche and a Volkswagen have many of the same parts, but when those parts are assembled into the final machine, they behave and function very differently." This analogy highlights that while normal and cancerous cells share fundamental building blocks, the way these components are organized and utilized can lead to vastly different functional outcomes.
By discerning the specific ways in which cancer cells exploit these internal transport systems more effectively than their healthy counterparts, scientists may be able to devise novel therapeutic strategies aimed at impeding or halting cancer cell dissemination. "If you can understand the differences, you can target future therapies based on how cancer cells and normal cells work differently," he emphasized, pointing towards a future of more personalized and targeted cancer treatments.
A Symphony of Disciplines: The Power of Interdisciplinary Collaboration
The success of this ambitious research endeavor was a testament to the power of interdisciplinary collaboration, bringing together leading experts from diverse fields including engineering, physics, microscopy, and cell biology. Crucial contributions emanated from collaborators at the Janelia Research Campus in Virginia, who provided specialized expertise in fluorescence correlation spectroscopy and three-dimensional super-resolution imaging.
"The instrumentation we needed doesn’t exist in most places," Cathy Galbraith noted, underscoring the unique resources required for this cutting-edge research. "Janelia had a one-of-a-kind setup that let us test and confirm what we were seeing." The availability of advanced imaging platforms at Janelia was instrumental in validating the team’s observations and providing the resolution necessary to visualize the intricate details of cellular transport.
The study leaned heavily on sophisticated imaging tools developed at Janelia, notably iPALM (interferometric photoactivated localization microscopy), a technique renowned for its ability to resolve structures at the nanometer scale, far beyond the diffraction limit of conventional light microscopy. "iPALM allowed us to physically see the compartments," Jim Galbraith stated, emphasizing the transformative impact of this technology. "There’s no other light-based technique that could do that." The ability to visualize these previously inaccessible cellular compartments was a critical factor in understanding the directed transport system.
The Emergence of a "Pseudo-Organelle"
The researchers conceptualize this newly identified system as a "pseudo-organelle" – a functional compartment within the cell that, unlike traditional organelles, is not enclosed by a membrane but nonetheless plays a pivotal role in orchestrating cellular behavior. This designation highlights its functional significance and unique structural characteristics.
"Just as small shifts in the jet stream can change the weather, small changes in these cellular winds could change how diseases begin or progress," Cathy Galbraith remarked, drawing a parallel to large-scale meteorological phenomena to illustrate the far-reaching implications of these cellular dynamics. The team posits that this discovery has the potential to catalyze advancements across multiple scientific domains, including oncology, drug delivery strategies, regenerative medicine, and synthetic biology.
"All you had to do was look," Cathy Galbraith concluded, reflecting on the profound simplicity of the initial observation. "The flows were there all along. Now we know how cells use them." This statement encapsulates the essence of scientific discovery – the unveiling of hidden truths through diligent observation and persistent inquiry.
The groundbreaking study was made possible through substantial funding from various prestigious institutions, including the National Institute of General Medical Sciences of the National Institutes of Health (Award number R01GM117188), the U.S. National Science Foundation (Award numbers 2345411 and 171636), the W. M. Keck Foundation, and the Howard Hughes Medical Institute, including its Janelia Visiting Scientist Program. The development and application of the iPALM technology were partially supported by an award from the Advanced Imaging Center at Janelia, while the super-resolution imaging (SIM) component received support from a Core Research Facilities grant from the OHSU School of Medicine.
In addition to the Galbraiths, the research team included Brian English, Ph.D., from the Janelia Research Campus, and Ulrike Boehm, Ph.D., who was formerly affiliated with Janelia and is now with Carl Zeiss AG in Germany, further underscoring the international and collaborative nature of this significant scientific undertaking.















Leave a Reply