Every second, countless electrical charges move through the human body, acting as the invisible engine for nearly every biological process. These microscopic signals are essential for life, driving the complex communication between cells, enabling the production of energy, and supporting the myriad chemical reactions that constitute metabolism. None of this sophisticated biological machinery would function without the careful and controlled movement of charges across cell membranes and within the interior of cells. In many ways, charge transport acts as a fundamental control system for biology, a microscopic power grid that dictates the rhythm of life itself. At the heart of this system lies a specific class of molecules whose behavior has long fascinated and puzzled the scientific community: phosphoric acid and its derivatives.
Phosphoric acid (H3PO4) and related phosphate compounds are ubiquitous in living systems. They are the structural backbone of DNA and RNA, the very blueprints of life. They form the essential building blocks of cell membranes, providing the necessary barrier and gateway for cellular integrity. Perhaps most importantly, they are central to adenosine triphosphate (ATP), the universal "energy currency" of the cell. ATP stores and transfers energy by breaking and reforming phosphate bonds, a process that relies heavily on the movement of positive charges, specifically protons. Beyond the realm of biology, phosphoric acid has become a cornerstone of modern green technology. Its exceptional ability to conduct protons makes it an ideal material for electrolytes in fuel cells and advanced battery systems, which are critical for the transition to a sustainable energy economy.
The Mechanics of the Proton Highway
The movement of protons through phosphate-containing materials is a unique physical phenomenon that differs significantly from the way electricity moves through a copper wire. In a metal, electrons flow relatively freely as a "gas" of particles. In contrast, protons in phosphoric acid move through a process often described as "proton-shuttling." Instead of traveling as independent entities through a medium, protons hop from one molecule to another in a highly coordinated fashion.
This mechanism relies on the presence of hydrogen bonds, which act as pre-defined pathways or "tracks" that guide the proton’s journey. When a proton attaches to one side of a molecule, it triggers a cascade of shifts that allows a different proton to exit from the opposite side, effectively moving the charge across a distance without a single particle having to travel the entire length. This process, also known as the Grotthuss mechanism, allows charges to move with remarkable speed and efficiency. It is why phosphoric acid is often referred to as "Nature’s proton highway."
However, despite decades of research, the precise molecular details of this highway have remained elusive. While scientists understood that the shuttling occurred, they lacked a clear picture of the initial steps—the "on-ramps" of the highway where the first transfer of a proton takes place. To investigate this, a collaborative team of researchers from the Department of Molecular Physics at the Fritz Haber Institute of the Max Planck Society in Berlin, along with colleagues from the University of Leipzig and institutions in the United States, focused their attention on the most basic unit of this process: the phosphate dimer.
Investigating Molecular Structures at Absolute Zero
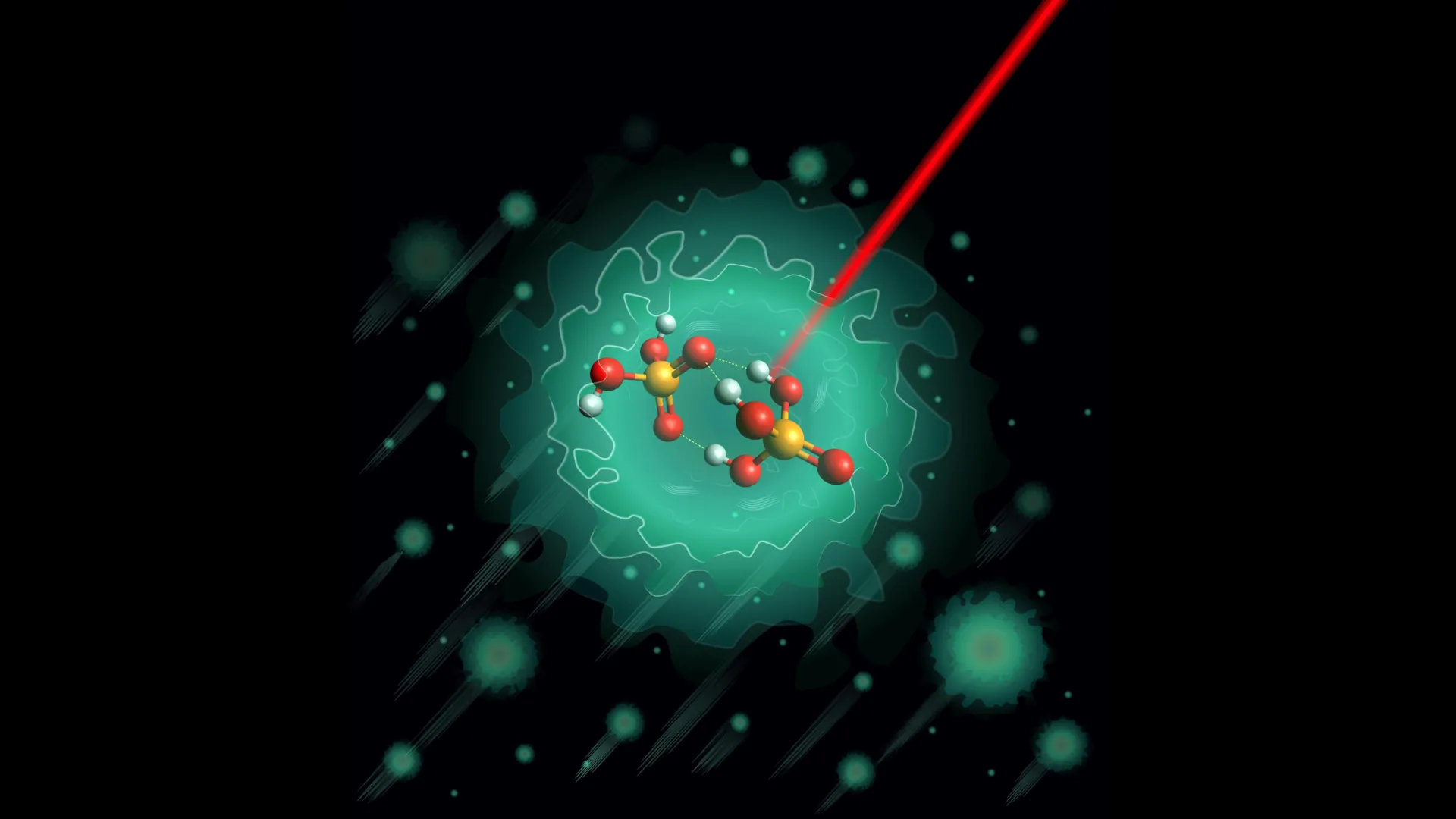
The research team hypothesized that a specific, negatively charged molecular complex was responsible for initiating the proton-shuttling process. This molecule, known as the deprotonated dimer (H3PO4·H2PO4⁻), consists of one neutral phosphoric acid molecule and one phosphate ion. Understanding the geometry and behavior of this dimer is crucial because it represents the smallest possible system where proton transfer between phosphate groups can be observed in isolation.
To examine this molecule without the interference of its environment, the scientists utilized a highly specialized technique known as Helium Nanodroplet Isolation (HENDI) combined with infrared spectroscopy. In the laboratory, the researchers created the H3PO4·H2PO4⁻ dimer and embedded it inside a nanodroplet of liquid helium. This environment is one of the coldest places in the universe, reaching a temperature of just 0.37 Kelvin—only a fraction of a degree above absolute zero.
At these extreme temperatures, molecular motion is almost entirely frozen. The "thermal noise" that usually blurs molecular images is eliminated, allowing researchers to capture a high-resolution "snapshot" of the molecule’s structure. By then hitting the molecule with infrared light, the team could observe how it vibrated. Because every molecular structure has a unique "vibrational fingerprint" based on its bonds, the resulting infrared spectra provided precise data on how the atoms were arranged.
A Conflict Between Theory and Reality
The experimental phase of the study was only half the battle. To interpret the complex infrared signals, the researchers employed quantum chemical calculations—sophisticated computer models based on the laws of quantum mechanics that predict how atoms should theoretically arrange themselves.
In the lead-up to the experiment, advanced theoretical models had predicted a degree of ambiguity. According to these simulations, the H3PO4·H2PO4⁻ dimer should have been able to exist in two different, equally stable configurations. In one version, the proton would be shared equally between the two phosphate groups; in another, it would be firmly attached to one side. This predicted "structural dualism" suggested a flexible and highly dynamic starting point for proton transport.
However, when the experimental data from the helium nanodroplets was analyzed, the results were startlingly clear. The data did not show a mixture of two structures, nor did it show the flexibility predicted by the models. Instead, it revealed a single, highly stable, and relatively rigid configuration.
This discovered structure features three distinct hydrogen bonds connected through a shared oxygen atom. Unlike the predicted flexible models, this real-world arrangement presents high energy barriers that limit the internal movement of protons within the dimer itself. This rigidity was an unexpected finding, suggesting that the "proton highway" is built upon a much more structured and disciplined foundation than previously assumed. Similar bonding patterns have since been identified in larger phosphoric acid clusters, indicating that this specific three-bond arrangement is a fundamental structural motif in phosphate chemistry.
Chronology of the Discovery and Research Context
The path to this discovery has been built over more than a century of chemical inquiry. The timeline of understanding proton transport highlights the significance of the Fritz Haber Institute’s recent breakthrough:
- 1806: Theodor Grotthuss proposes the first theory of charge transport in liquids, suggesting that protons "hop" between molecules rather than diffusing through them.
- 1950s-1970s: The emergence of modern biochemistry identifies the role of phosphate groups in ATP and DNA, sparking intense interest in how these groups manage charge.
- 1990s: The development of Phosphoric Acid Fuel Cells (PAFCs) highlights the industrial importance of proton conductivity, leading to more rigorous molecular modeling.
- 2010-2020: Quantum chemical modeling becomes the standard for predicting molecular behavior, though discrepancies between models and experiments persist.
- 2023-2024: The Fritz Haber Institute and its international partners utilize helium nanodroplet technology to provide the first definitive experimental map of the H3PO4·H2PO4⁻ dimer, resolving long-standing theoretical conflicts.
Expert Analysis and Implications
The discovery that a single, rigid structure dominates the initial phase of proton transport has profound implications for several fields of science.
Refining Quantum Chemistry:
The primary takeaway for the scientific community is the necessity of experimental verification. Even the most advanced quantum models, which use Density Functional Theory (DFT), failed to accurately predict the singular nature of the phosphate dimer. By providing an exact experimental benchmark, this research allows theorists to "tune" their models, leading to more accurate simulations of complex biological and chemical systems in the future.
Advancing Sustainable Energy:
In the technological sector, this research provides a blueprint for the design of next-generation electrolytes. Fuel cells, which convert hydrogen into electricity with water as the only byproduct, rely on efficient proton exchange membranes. Understanding why certain phosphate structures are more stable than others allows materials scientists to engineer synthetic membranes that mimic "Nature’s proton highway," potentially leading to fuel cells that are more durable and operate at higher efficiencies.
Biological Insights:
In biology, the findings offer a deeper look at the mechanism of ATP hydrolysis and synthesis. If the phosphate groups in our cells are predisposed to form these rigid, triple-bonded structures, it changes how we calculate the energy required for metabolic reactions. It suggests that the "hopping" of protons in our bodies is not a random occurrence but a highly regulated event dictated by the specific geometry of phosphate dimers.
Official Perspectives and Future Directions
While the research team has not released personalized quotes, the consensus reflected in the study’s conclusion emphasizes a shift in the department’s focus. The Fritz Haber Institute has indicated that this work is part of a broader effort to map the "solvation shells" of essential biological molecules. By understanding how a single dimer behaves, they can now begin to add more molecules—water, additional phosphates, or metal ions—to see how the "highway" expands into a complex network.
The collaboration between German and American institutions also underscores the international importance of this fundamental research. As the world seeks to understand the molecular basis of life and develop cleaner energy sources, the humble phosphate molecule remains at the center of the conversation.
This breakthrough serves as a reminder that even the most common substances—found in every cell of our bodies and every bottle of soda—still hold deep secrets. By cooling these molecules to the brink of absolute zero, scientists have finally been able to see the first step in the journey of a proton, providing a clearer view of the fundamental forces that power our world. The "Nature’s proton highway" is no longer a theoretical concept; it is a mapped reality, paved with specific molecular bonds that define the very limits of biological energy and technological potential.














Leave a Reply