Eli Lilly and Company today announced groundbreaking results from a Phase 3 clinical trial for its investigational triple agonist, retatrutide, revealing an average weight loss of 16.8% in participants with type 2 diabetes. This significant outcome positions retatrutide as a potential next-generation therapy in the rapidly evolving landscape of metabolic disease management, offering robust improvements in both glycemic control and body weight reduction. The company formally released these findings, underscoring the drug’s potential to address the complex interplay between type 2 diabetes and obesity.
Unprecedented Efficacy in Metabolic Management
The Phase 3 trial, designed to evaluate the efficacy and safety of retatrutide in adults with type 2 diabetes, demonstrated that the drug met both its primary and key secondary endpoints. Beyond the impressive average weight loss of 16.8%, participants also experienced an average reduction in A1C levels of up to 2.0% over 40 weeks when compared to placebo. This dual-action superiority in glycemic control and weight management is particularly noteworthy, as traditional diabetes treatments often struggle to achieve substantial weight loss concurrently with A1C targets. On average, individuals in the trial shed approximately 36.6 pounds, showcasing a clinically meaningful impact on body mass.
One of the most compelling aspects of the trial data was the observation that no weight-loss plateau was reached during the 40-week study period. Participants continued to exhibit a consistent weight loss trajectory, suggesting that the full extent of retatrutide’s weight-reducing potential might extend beyond the study duration. This "no plateau" finding differentiates retatrutide from many existing weight management therapies, where efficacy often tapers off after an initial period. This sustained effect could translate into more profound and durable health benefits for patients over the long term.
A Novel Mechanism: The Triple Agonist Advantage
Retatrutide is an investigational first-in-class GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon triple hormone receptor agonist. This unique pharmacological profile distinguishes it from current market leaders, including Lilly’s own dual GIP/GLP-1 agonist, tirzepatide (marketed as Mounjaro for type 2 diabetes and Zepbound for obesity), and GLP-1 receptor agonists like semaglutide (Ozempic for type 2 diabetes and Wegovy for obesity) from Novo Nordisk.
To understand the significance of a "triple agonist," it’s crucial to grasp the roles of these three gut hormones:
- GLP-1: This hormone enhances insulin secretion in a glucose-dependent manner, suppresses glucagon release after meals, slows gastric emptying, and promotes satiety, all contributing to lower blood glucose and reduced food intake.
- GIP: While also enhancing glucose-dependent insulin secretion, GIP has additional metabolic effects, including direct action on adipose tissue and bone, potentially contributing to energy storage and expenditure.
- Glucagon: Traditionally known for its role in raising blood glucose, glucagon agonism, when combined with GLP-1 and GIP, can paradoxically lead to weight loss. This is believed to occur through increased energy expenditure, potentially by activating glucagon receptors in the liver and adipose tissue, leading to fat burning and improved metabolic rate.
By simultaneously activating receptors for GIP, GLP-1, and glucagon, retatrutide targets multiple pathways involved in glucose homeostasis, energy balance, and appetite regulation. This multi-pronged approach is hypothesized to unlock synergistic effects, leading to the superior weight loss and glycemic control observed in the trial. The combined action aims to optimize the body’s natural metabolic responses, offering a more comprehensive treatment strategy than single or dual agonist therapies.
Addressing a Complex Clinical Challenge
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, articulated the significant need that retatrutide aims to address. "For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes," Custer stated in the company’s announcement. He added, "With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes."
Custer’s comments highlight a long-standing challenge in managing type 2 diabetes. Obesity is a major risk factor for the development and progression of type 2 diabetes, and the two conditions often co-exist, forming a complex web of cardiometabolic dysfunction. While some diabetes medications can lead to modest weight gain or are weight-neutral, the advent of incretin-based therapies has revolutionized treatment by offering both glycemic control and significant weight reduction. Retatrutide’s ability to achieve both substantial A1C reduction and unprecedented weight loss in this patient population signifies a major step forward, potentially simplifying and improving treatment outcomes for millions.
Broader Health Benefits and Safety Profile
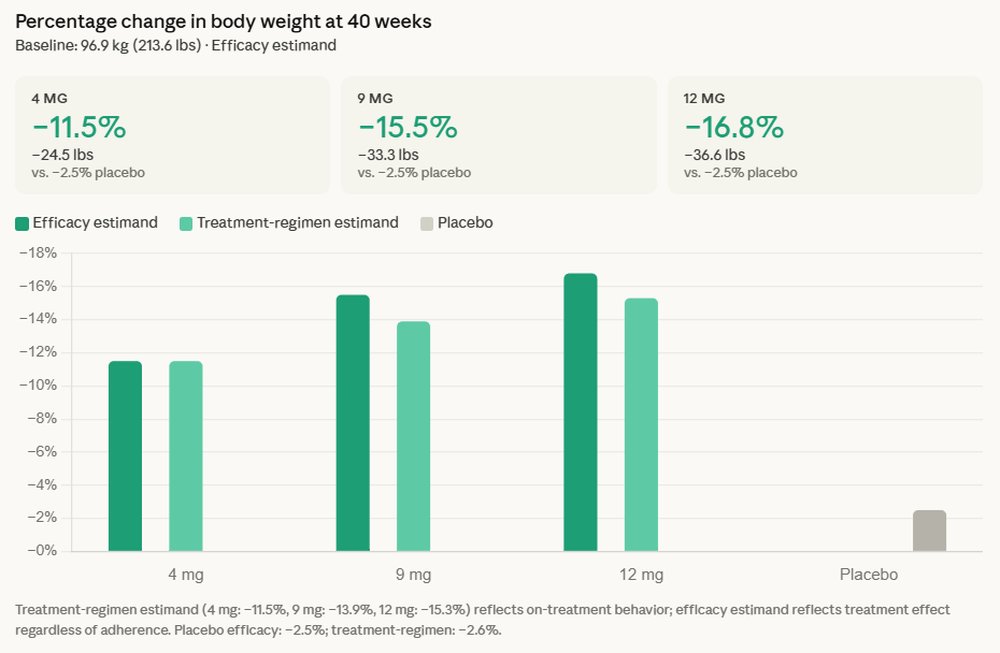
Beyond its effects on weight and A1C, retatrutide also demonstrated clinically meaningful improvements across several key cardiovascular risk factors. These included reductions in non-HDL cholesterol, triglycerides, and systolic blood pressure. These improvements are critical, as individuals with type 2 diabetes and obesity are at a significantly higher risk for cardiovascular diseases, which remain the leading cause of morbidity and mortality in this population. By positively influencing these markers, retatrutide holds the promise of not just managing diabetes and obesity, but also mitigating associated cardiovascular complications, thereby improving overall patient health and longevity.
The safety profile of retatrutide in this Phase 3 trial was consistent with that of other incretin-based therapies. The most commonly reported adverse effects included gastrointestinal symptoms such as nausea, diarrhea, and vomiting. These side effects are generally mild to moderate in severity, often transient, and tend to be dose-dependent, meaning they can sometimes be managed by adjusting the medication dosage. The consistency of these side effects with similar drugs suggests a predictable tolerability profile, which is important for patient adherence and long-term use.
The Road Ahead: Extensive Clinical Development
The successful completion of this Phase 3 trial for type 2 diabetes is just one piece of a much larger clinical development program for retatrutide. Eli Lilly is actively studying the drug in a comprehensive series of additional Phase 3 clinical trials, underscoring its ambition for retatrutide to become a foundational therapy across a broad spectrum of cardiometabolic and weight-related conditions.
These ongoing trials are evaluating retatrutide’s potential efficacy and safety in:
- Obesity and overweight with at least one weight-related medical problem: This is a crucial indication, given the staggering global prevalence of obesity and the success of similar drugs in this area.
- Knee osteoarthritis: Weight loss is a primary intervention for managing osteoarthritis, and a drug offering substantial weight reduction could significantly impact the progression and symptoms of this debilitating condition.
- Moderate-to-severe obstructive sleep apnea: Obesity is a major risk factor for sleep apnea, and effective weight loss could provide a non-surgical solution for many patients.
- Chronic low back pain: Similar to osteoarthritis, weight reduction can alleviate mechanical stress on the spine, potentially improving chronic low back pain.
- Cardiovascular and renal outcomes: These trials are essential to establish whether the observed improvements in cardiovascular risk factors translate into reduced rates of major adverse cardiovascular events and kidney disease progression, which are critical endpoints for regulatory approval and clinical adoption.
- Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly known as non-alcoholic fatty liver disease (NAFLD): MASLD is strongly linked to obesity and type 2 diabetes, and there is a significant unmet need for effective pharmacological treatments. Weight loss is a key strategy, making retatrutide a promising candidate.
The sheer breadth of these ongoing trials highlights Lilly’s strategic vision for retatrutide as a versatile, blockbuster drug capable of addressing multiple interconnected health crises. Should these trials yield positive results, retatrutide could offer a single therapeutic agent for managing a constellation of conditions, potentially simplifying treatment regimens and improving patient outcomes across various medical specialties.
Competitive Landscape and Market Implications
The pharmaceutical market for diabetes and obesity treatments is experiencing unprecedented growth and innovation, largely driven by the success of incretin-based therapies. Eli Lilly already holds a strong position with tirzepatide (Mounjaro/Zepbound), a dual GIP/GLP-1 agonist that has demonstrated impressive efficacy. Novo Nordisk, with its GLP-1 agonist semaglutide (Ozempic/Wegovy), is another major player.
Retatrutide’s emergence as a triple agonist, showing even greater weight loss potential than its predecessors, could further intensify this competition. The "no weight-loss plateau" finding is a particularly strong differentiator, suggesting a potentially superior long-term efficacy profile. If approved, retatrutide would represent a significant leap forward, potentially setting a new benchmark for weight loss and metabolic control.
The implications for patients are profound. A new class of highly effective medications could offer hope to millions who struggle with the dual burden of type 2 diabetes and obesity, and the numerous associated comorbidities. For healthcare systems, the potential to address multiple chronic conditions with a single therapy could lead to more integrated care pathways and improved public health outcomes, albeit with considerations for access and affordability.
Conclusion: A New Era in Metabolic Medicine
The results from Eli Lilly’s Phase 3 trial for retatrutide mark a pivotal moment in the fight against type 2 diabetes and obesity. The drug’s ability to achieve an average of 16.8% weight loss and up to a 2.0% A1C reduction, alongside improvements in cardiovascular risk factors, positions it as a highly promising therapeutic agent. Its unique triple agonist mechanism and the observed sustained weight loss trajectory differentiate it within an increasingly competitive market. As Lilly continues its extensive clinical development program across multiple indications, retatrutide stands poised to redefine the treatment landscape for a range of cardiometabolic diseases, offering new hope for comprehensive and effective patient care. The scientific community and patient populations eagerly await further data and regulatory decisions that could usher in a new era of metabolic medicine.














Leave a Reply