Eli Lilly and Company, a venerable titan in the pharmaceutical industry, has achieved an unprecedented milestone, claiming the top spot globally by revenue. This ascent coincides with a significant regulatory victory: the U.S. Food and Drug Administration (FDA) on April 1 approved Foundayo (orforglipron), Lilly’s once-daily oral GLP-1 receptor agonist designed for obesity management. This approval marks the first oral GLP-1 specifically cleared for weight management and represents the fastest new molecular entity approval since 2002. Foundayo’s clearance under the FDA’s Commissioner’s National Priority Voucher program in an astonishing 50 days post-filing, nearly ten months ahead of its anticipated review date, underscores its perceived importance and the efficiency of the expedited process. However, this dual triumph has been met with a surprising lack of enthusiasm from the financial markets, with Lilly’s stock experiencing a nearly 14% decline since the beginning of January. This paradoxical reaction highlights the complex interplay between scientific innovation, market expectations, intense competition, and broader economic factors that shape investor sentiment in the high-stakes pharmaceutical sector.
Lilly’s Remarkable Financial Ascent to Global Leadership
The pharmaceutical landscape has witnessed a dramatic shift, with Eli Lilly emerging as the new revenue leader. The company reported a staggering $65.18 billion in fiscal year 2025 revenue, representing an extraordinary 44.7% year-over-year growth. This explosive expansion rate not only surpassed all other top 20 pharmaceutical companies but also propelled Lilly past long-standing industry giants. For the first time, Lilly outranked Merck & Co., which posted $65.01 billion, Pfizer with $62.58 billion, and AbbVie at $61.16 billion. While the margin over Merck was a narrow $170 million, the stark contrast in growth rates – Lilly’s 44.7% compared to Merck’s modest 1.3% – underscores the divergent trajectories of these industry leaders. Pfizer, once a frontrunner, experienced a 1.7% decline, while AbbVie and Johnson & Johnson recorded growth rates of 8.6% and 5.8%, respectively.
This meteoric rise is unequivocally attributed to the unprecedented success of Lilly’s traditional injectable GLP-1 (glucagon-like peptide-1) receptor agonists. Mounjaro (tirzepatide), initially approved for type 2 diabetes and later for obesity under the brand name Zepbound, has been a monumental revenue driver. In the first half of 2025 alone, Mounjaro generated an astounding $9.0 billion, demonstrating a 90% year-over-year increase. Zepbound, the dedicated obesity indication for tirzepatide, contributed an additional $5.7 billion in H1 2025, a particularly impressive feat considering it was only launched in late 2023 and has rapidly gained market penetration, now available in a convenient multi-dose KwikPen. Cumulatively, Lilly’s GLP-1 portfolio is on track to exceed an annualized revenue of $30 billion, establishing a new benchmark for blockbuster drug performance and cementing the company’s dominance in the metabolic disease space.
The GLP-1 Revolution and the Global Obesity Crisis
To fully appreciate the significance of Lilly’s recent successes, it is crucial to understand the broader context of the GLP-1 receptor agonist class and the escalating global obesity epidemic. Obesity, recognized as a chronic disease by major health organizations, affects over a billion people worldwide and is a major risk factor for numerous comorbidities, including type 2 diabetes, cardiovascular disease, certain cancers, and musculoskeletal disorders. The economic burden of obesity, encompassing direct healthcare costs and indirect productivity losses, runs into trillions of dollars annually.
GLP-1 receptor agonists mimic the action of a natural hormone, GLP-1, which is released in the gut in response to food intake. These drugs stimulate insulin secretion, suppress glucagon secretion, slow gastric emptying, and promote satiety, leading to improved glycemic control and significant weight loss. While initially developed for type 2 diabetes, their profound impact on weight loss has transformed the treatment landscape for obesity. Prior to the advent of highly effective GLP-1s, pharmacological options for weight management were limited in efficacy and often plagued by side effects.
The market for GLP-1s has undergone an explosive expansion, growing from under $12 billion in 2022 to an estimated annualized run rate of approximately $65 billion by mid-2025. This rapid growth underscores the immense unmet need for effective obesity treatments and the transformative power of this drug class. However, until recently, the vast majority of these blockbuster GLP-1s, including Ozempic, Wegovy (Novo Nordisk’s offerings), Mounjaro, and Zepbound, were available only as injectables, requiring weekly or daily self-administration. While highly effective, the injectable format can be a barrier for some patients due to needle aversion, convenience, or perceived complexity.
Foundayo: A New Frontier in Oral Metabolic Therapy
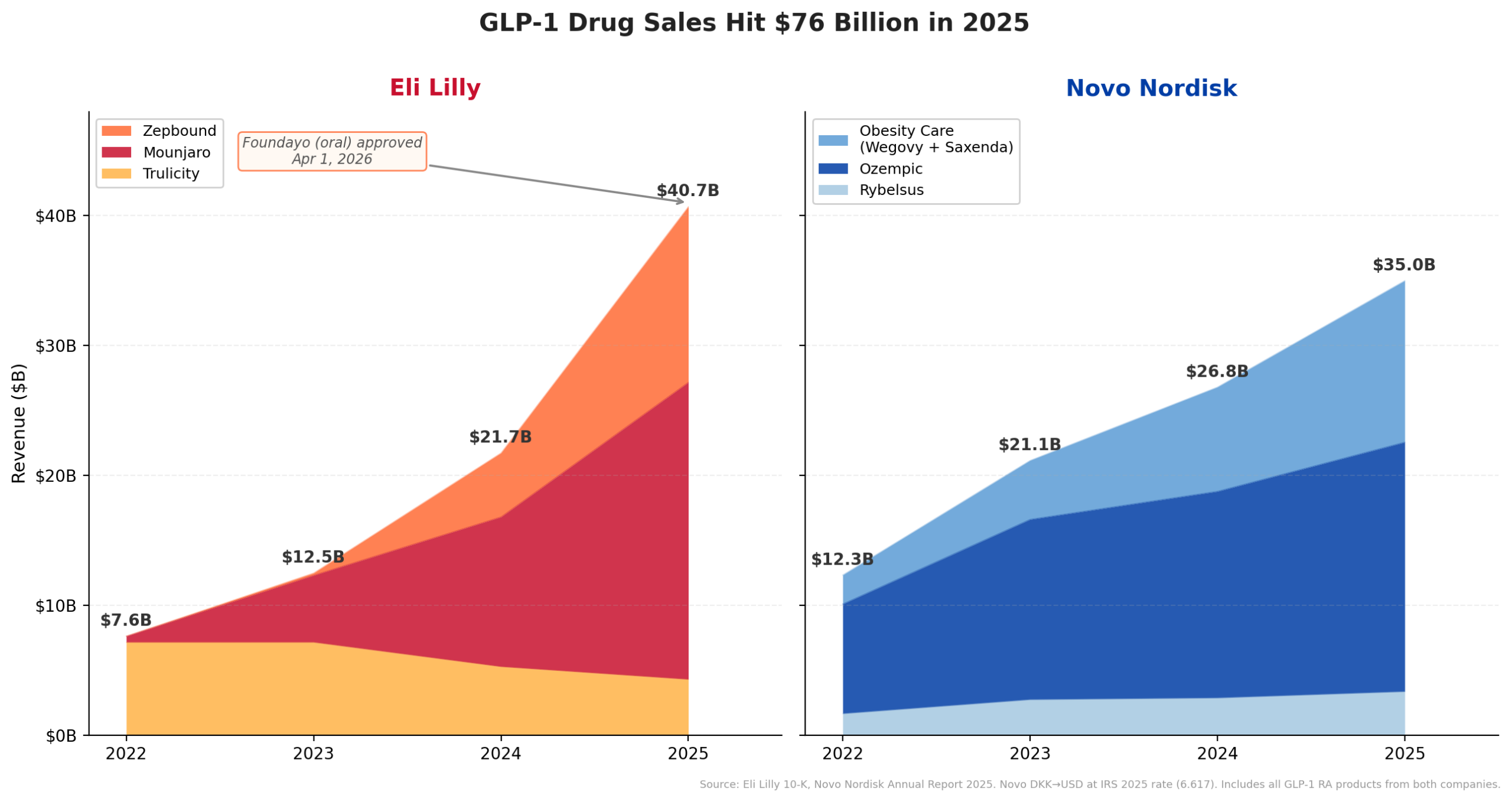
The FDA’s approval of Foundayo represents a pivotal moment in the GLP-1 saga, offering a critical alternative to injectables. While Novo Nordisk’s Rybelsus (oral semaglutide) and an oral formulation of Wegovy (approved December 2025) are also oral GLP-1s, Foundayo distinguishes itself through its pharmacological properties and administration profile. Rybelsus and oral Wegovy are peptide formulations that require strict administration protocols, such as being taken on an empty stomach with a limited amount of water, and waiting a specific period before consuming food or other medications. These requirements can impact patient adherence and convenience.
In contrast, Foundayo is a small molecule GLP-1 receptor agonist. This chemical structure provides distinct advantages: it can be taken once daily at any time, without the stringent food or water restrictions associated with peptide-based oral formulations. This enhanced flexibility is expected to significantly improve patient convenience and potentially adherence, broadening the appeal of GLP-1 therapy to a wider demographic. Lilly CEO Dave Ricks highlighted this strategic positioning, noting that while Foundayo might not be "more effective" than the company’s highly potent injectable Zepbound in terms of maximum weight loss, "it’s more accessible, it’s easier to fit into your daily routine."
Clinical trial data from the ATTAIN-1 program showed that patients receiving the highest dose of Foundayo achieved an average of 12.4% body weight loss over 72 weeks. While this is a substantial and clinically meaningful reduction, it is quantitatively less than the 20%-plus weight loss typically observed with injectable tirzepatide (Mounjaro/Zepbound). However, Ricks framed Foundayo as a complementary, rather than competitive, offering within Lilly’s portfolio. The strategy is to provide a spectrum of options, ensuring "people to be on the medicine that meets their health goals. If it has Lilly on the box, that’s the goal we have." This approach recognizes that different patients have varying needs, preferences, and clinical profiles, and a diverse therapeutic arsenal can maximize market penetration.
The regulatory journey for Foundayo was exceptionally swift. The approval under the FDA’s Commissioner’s National Priority Voucher program, which aims to incentivize the development of drugs addressing significant public health needs, allowed for an expedited review process. The approval in just 50 days is an extraordinary feat, starkly contrasting with the typical 8-12 month standard review period for new molecular entities. This rapid clearance underscores the FDA’s recognition of the urgent need for accessible obesity treatments. Lilly had originally licensed orforglipron from Japanese drugmaker Chugai in 2018 for an upfront payment of $50 million, a testament to the long-term strategic vision that has now come to fruition.
The Wall Street Paradox: Why the Muted Celebration?
Despite achieving the top revenue spot and securing a landmark oral GLP-1 approval, Eli Lilly’s stock has experienced a significant decline of nearly 14% since January 1. This "Wall Street paradox" can be attributed to several converging factors:
- High Expectations and "Buy the Rumor, Sell the News": Much of Lilly’s growth potential and pipeline strength, particularly concerning its GLP-1 portfolio, has been heavily anticipated and, arguably, already "priced in" by investors. The market often front-runs positive news, causing a dip when the actual announcement occurs, as investors who bought on speculation take profits.
- Intense Competitive Landscape: While Lilly and Novo Nordisk currently dominate the GLP-1 market, the field is becoming increasingly crowded. Numerous other pharmaceutical companies are developing their own GLP-1 or multi-agonist drugs, some with potentially differentiated profiles or improved convenience. The long-term market share for any single product, even a highly effective one, is subject to intense future competition.
- Efficacy vs. Convenience Trade-off: While Foundayo offers superior convenience, its 12.4% weight loss is less than the 20%+ seen with injectable tirzepatide. Some analysts may be concerned that this efficacy difference could limit its market potential, especially if patients prioritize maximum weight loss over oral convenience, or if insurance coverage prioritizes more potent options.
- Manufacturing and Supply Constraints: The demand for GLP-1s has far outstripped supply, leading to widespread shortages of Mounjaro and Zepbound. While Lilly is aggressively investing in manufacturing expansion, concerns persist about the company’s ability to scale production sufficiently to meet the colossal global demand for both existing injectables and the newly approved oral Foundayo. Unmet demand could cap revenue growth.
- Valuation Concerns: Lilly’s stock has surged dramatically in recent years, making it one of the most highly valued pharmaceutical companies. Some analysts may view the current valuation as stretched, suggesting that the stock’s price already reflects an optimistic outlook for future growth, leaving less room for further upside in the short term.
- Broader Market Dynamics: General market sentiment, interest rate expectations, and sector rotations can also influence stock performance, even for companies with strong fundamentals. Investors might be reallocating capital to other sectors or seeking less volatile assets.
- Thin Margin Over Competitors: While Lilly did achieve the #1 revenue spot, the $170 million lead over Merck is relatively slim. This might lead some investors to question the sustainability of this lead if Merck’s pipeline or other products gain traction.
Broader Implications and Future Outlook
The approval of Foundayo and Lilly’s ascension to the top revenue spot carry significant implications for the pharmaceutical industry, healthcare systems, and patients worldwide.
- Expanded Patient Access and Adherence: An easily administrable oral GLP-1 like Foundayo has the potential to dramatically expand the addressable patient population for obesity treatment. Many individuals who are hesitant about injections or find the strict requirements of other oral GLP-1s challenging may now be more willing to initiate and adhere to therapy. This could lead to a greater proportion of the obese population receiving effective medical intervention, potentially mitigating the long-term health consequences and economic burden of the disease. Lilly’s decision to submit orforglipron for approval in over 40 countries underscores its global ambition for this product.
- Competitive Landscape Intensification: The entry of a highly convenient oral GLP-1 from Lilly intensifies the rivalry with Novo Nordisk. While Novo Nordisk has its own oral GLP-1s, Foundayo’s small molecule nature and flexible dosing could provide a distinct competitive edge. This competition is likely to spur further innovation, potentially leading to even more effective and convenient treatment options in the future. The "race to oral" and the development of next-generation multi-agonists will define the coming decade in metabolic medicine.
- Healthcare System Impact: Widespread adoption of effective obesity treatments could have a transformative impact on healthcare systems. By addressing obesity, there is potential to reduce the incidence and severity of associated chronic diseases, leading to lower healthcare expenditures in the long run. However, the initial costs of these innovative therapies and challenges related to insurance coverage will remain significant considerations.
- Lilly’s Strategic Diversification: With a comprehensive GLP-1 portfolio spanning highly efficacious injectables (Mounjaro, Zepbound) and a convenient oral option (Foundayo), Lilly is strategically positioned to capture various segments of the metabolic disease market. This diversification within a single therapeutic class reduces reliance on any single product format and allows the company to cater to a broader range of patient needs and preferences, solidifying its long-term leadership.
- Research and Development Focus: The success of GLP-1s will undoubtedly fuel further research and development in metabolic science. Companies will continue to explore novel mechanisms of action, combination therapies, and even more advanced delivery systems to push the boundaries of treatment for obesity and related conditions.
In conclusion, Eli Lilly’s achievement of becoming the world’s top-revenue pharmaceutical company, largely driven by its groundbreaking GLP-1 franchise, coupled with the landmark approval of its oral GLP-1, Foundayo, marks a pivotal moment in its history. While Wall Street’s immediate reaction reflects a complex blend of high expectations, competitive pressures, and market dynamics, the underlying narrative is one of profound innovation and strategic execution. Lilly’s ability to offer a diverse arsenal of highly effective and increasingly convenient metabolic therapies positions it not only as a financial leader but also as a critical player in addressing one of the most pressing global health challenges of our time. The journey ahead will involve navigating intense competition, ensuring robust supply, and demonstrating sustained innovation, but Lilly has undeniably laid a strong foundation for continued influence and growth.














Leave a Reply