Eli Lilly and Company today announced groundbreaking results from a Phase 3 clinical trial for its investigational triple agonist, retatrutide, revealing an average weight loss of 16.8% in participants with type 2 diabetes. This significant achievement, coupled with substantial improvements in glycemic control, positions retatrutide as a potentially transformative therapeutic option in the increasingly competitive landscape of cardiometabolic medicine. The company’s announcement underscores a critical advancement in addressing the complex interplay between type 2 diabetes and obesity, conditions that affect millions globally and pose immense public health challenges.
Unprecedented Efficacy in Glycemic Control and Weight Management
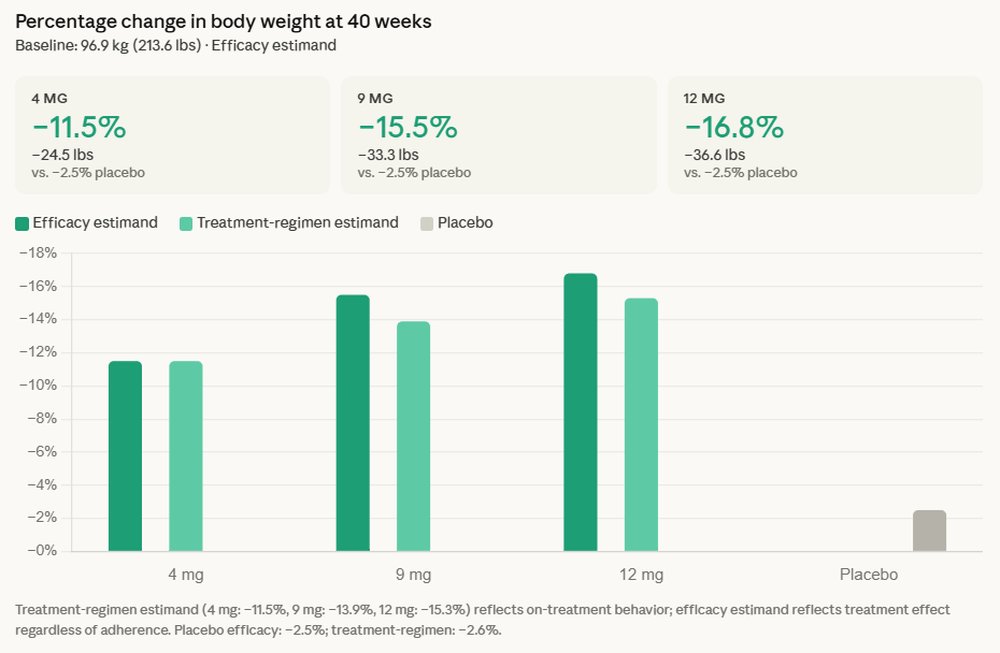
The Phase 3 trial, designed to evaluate the efficacy and safety of retatrutide in adults with type 2 diabetes, demonstrated that the novel compound met both its primary and key secondary endpoints. Participants treated with retatrutide achieved superior reductions in glycated hemoglobin (A1C), a key marker of long-term blood sugar control, and exhibited remarkable weight loss compared to those receiving a placebo. Specifically, the trial reported an average A1C reduction of up to 2.0% and an average weight loss of 36.6 pounds (equivalent to 16.8% of body weight) over a 40-week treatment period.
One of the most compelling findings highlighted by Eli Lilly is the absence of a weight-loss plateau observed during the 40-week study duration. This suggests that participants continued to lose weight throughout the trial, indicating a sustained and potentially ongoing therapeutic effect that could extend beyond the initial study period. This continuous weight reduction is particularly noteworthy in the context of chronic weight management, where plateaus often impede long-term success.
Beyond its primary effects on A1C and body weight, retatrutide also demonstrated clinically meaningful improvements across several crucial cardiovascular risk factors. These included reductions in non-HDL cholesterol, triglycerides, and systolic blood pressure – all significant indicators associated with an elevated risk of heart disease and stroke. The ability to positively impact these multifactorial risks simultaneously with glycemic control and weight loss could offer a holistic benefit to patients with type 2 diabetes, who are inherently at a higher risk for cardiovascular complications.
The safety profile of retatrutide in this trial was consistent with that of other incretin-based therapies. The most commonly reported adverse effects were gastrointestinal in nature, including nausea, diarrhea, and vomiting. These side effects are generally manageable and often diminish over time as patients adjust to treatment, aligning with observations from existing GLP-1 receptor agonists and dual agonists.
The Science Behind the Triple Agonist: A Novel Mechanism of Action
Retatrutide is an investigational, first-in-class GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon triple hormone receptor agonist. This unique pharmacological profile distinguishes it from current therapies and represents a significant leap in drug development for metabolic disorders.
To understand the power of a triple agonist, it’s essential to appreciate the individual roles of these gut hormones:
- GLP-1: This hormone is well-known for its role in glucose homeostasis. It stimulates glucose-dependent insulin secretion from pancreatic beta cells, suppresses glucagon secretion (which lowers glucose production from the liver), slows gastric emptying, and promotes satiety, leading to reduced food intake and weight loss. Existing GLP-1 receptor agonists like semaglutide (Ozempic, Wegovy) have revolutionized the treatment of type 2 diabetes and obesity.
- GIP: Also an incretin hormone, GIP primarily enhances glucose-dependent insulin secretion. It has also been implicated in fat deposition and bone metabolism. Combining GIP agonism with GLP-1 agonism, as seen in Eli Lilly’s dual agonist tirzepatide (Mounjaro, Zepbound), has already demonstrated superior efficacy compared to GLP-1 monotherapy, particularly in weight loss.
- Glucagon: Historically known for its role in raising blood glucose levels by stimulating hepatic glucose production, glucagon’s role in weight loss and metabolism has garnered renewed interest. When activated by an agonist in the context of GLP-1 and GIP agonism, glucagon receptor activation can lead to increased energy expenditure and direct lipolysis (fat breakdown) in adipose tissue. This multifaceted action potentially offers an additional pathway for weight reduction and metabolic improvement that is not present in single or dual agonists.
By targeting all three receptors simultaneously, retatrutide aims to harness the synergistic effects of these hormones, leading to a more profound impact on glucose metabolism, appetite regulation, and energy expenditure. This comprehensive approach is believed to be the driving force behind the impressive weight loss and A1C reductions observed in the trial, potentially surpassing the efficacy of current single and dual agonists. The "first-in-class" designation underscores its innovative mechanism and potential to establish a new standard of care.
The Growing Epidemic: Type 2 Diabetes and Obesity
The positive results for retatrutide emerge against a backdrop of a global health crisis characterized by the escalating prevalence of type 2 diabetes and obesity. According to the International Diabetes Federation (IDF), approximately 537 million adults worldwide were living with diabetes in 2021, a number projected to rise to 783 million by 2045. A vast majority of these cases are type 2 diabetes, strongly linked to overweight and obesity.
Obesity itself is a chronic, relapsing disease affecting over 650 million adults globally. The World Health Organization (WHO) identifies it as a major risk factor for numerous non-communicable diseases, including cardiovascular diseases, various cancers, musculoskeletal disorders, and, critically, type 2 diabetes. The Centers for Disease Control and Prevention (CDC) reports that in the United States, the prevalence of obesity was 41.9% in 2017-2020, while the prevalence of severe obesity was 9.2%.
For individuals with type 2 diabetes, achieving sustained weight loss has historically been a formidable challenge. Conventional treatments for diabetes primarily focus on glycemic control, with weight management often being a secondary or difficult-to-achieve goal. However, mounting evidence unequivocally demonstrates that significant weight loss can lead to remission of type 2 diabetes in some individuals and substantially improve disease management and reduce complications in others.
Kenneth Custer, executive vice president and president of Lilly Cardiometabolic Health, articulated this challenge: "For many people with type 2 diabetes, it is a struggle to achieve both A1C control and weight loss, since obesity has historically been harder to treat for those with type 2 diabetes. With triple agonist retatrutide, we set out to make a molecule that could help patients achieve substantial A1C reduction and weight loss. These results support the potential of this molecule for people living with type 2 diabetes." His statement highlights the critical unmet need that retatrutide aims to address by offering a potent solution for both conditions simultaneously.
Eli Lilly’s Strategic Foothold in Cardiometabolic Health
Eli Lilly has firmly established itself as a frontrunner in the cardiometabolic disease space. The company’s recent successes with tirzepatide, marketed as Mounjaro for type 2 diabetes and Zepbound for chronic weight management, have already set new benchmarks for efficacy. Mounjaro, a GIP/GLP-1 dual agonist, has consistently shown superior A1C reduction and weight loss compared to GLP-1 monotherapies. Zepbound has achieved average weight reductions exceeding 20% in its pivotal obesity trials.
The emergence of retatrutide, a triple agonist, signifies Lilly’s aggressive pursuit of even more effective treatments. This strategic focus positions the company at the forefront of a rapidly expanding market driven by the global obesity and diabetes epidemics. The pharmaceutical industry is witnessing intense competition in this area, with major players like Novo Nordisk (maker of semaglutide, branded as Ozempic and Wegovy) also investing heavily in next-generation therapies. Lilly’s commitment to developing increasingly potent incretin-based therapies demonstrates its ambition to capture a dominant share of this lucrative market and, more importantly, to offer increasingly impactful solutions to patients.
Broader Implications and Future Outlook
The promising results from the type 2 diabetes trial are just one piece of the puzzle for retatrutide. Eli Lilly is conducting an extensive Phase 3 clinical program, evaluating the drug’s potential efficacy and safety across a wide range of cardiometabolic and related conditions. This comprehensive development plan underscores the company’s confidence in the molecule’s broad therapeutic utility.
Ongoing and planned Phase 3 trials for retatrutide include investigations into:
- Obesity and Overweight with at least one weight-related medical problem: This is a crucial area, as the profound weight loss observed in the type 2 diabetes trial suggests retatrutide could be a powerful tool for weight management in a broader population.
- Knee Osteoarthritis: Obesity is a major risk factor for osteoarthritis, and significant weight loss could alleviate joint pain and improve mobility.
- Moderate-to-Severe Obstructive Sleep Apnea: Weight reduction is a primary treatment strategy for sleep apnea, a condition closely linked to obesity.
- Chronic Low Back Pain: Similar to knee osteoarthritis, excess weight exacerbates back pain, and weight loss could provide relief.
- Cardiovascular and Renal Outcomes: Given the observed improvements in cardiovascular risk factors, dedicated outcome trials are essential to determine if retatrutide can reduce major adverse cardiovascular events and slow the progression of kidney disease.
- Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): Formerly known as non-alcoholic fatty liver disease (NAFLD), MASLD is strongly associated with obesity and type 2 diabetes, and weight loss is the cornerstone of its treatment.
The comprehensive nature of these trials suggests that Lilly envisions retatrutide not merely as a diabetes drug or an obesity drug, but as a broad metabolic agent capable of addressing multiple interconnected health issues. If these trials yield similar positive results, retatrutide could redefine treatment paradigms across several medical specialties, offering a single agent to manage a constellation of conditions.
From a regulatory perspective, successful completion of these extensive Phase 3 programs will pave the way for submissions to health authorities like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). While specific timelines are not yet public, such a robust data package would typically lead to applications in the coming years, potentially making retatrutide available to patients by the latter half of the decade.
The potential market impact of retatrutide is immense. With an aging global population and rising rates of obesity, the demand for highly effective weight loss and diabetes treatments is soaring. Analysts project the market for GLP-1 and related agonists to reach hundreds of billions of dollars annually within the next decade. Eli Lilly, with its strong pipeline including tirzepatide and now retatrutide, is strategically positioned to be a dominant force in this growth.
Challenges and Considerations Ahead
Despite the excitement surrounding retatrutide, several factors will be critical in determining its long-term success and accessibility. The cost of these advanced therapies is a significant consideration. Currently, GLP-1 and dual agonists carry high price tags, raising concerns about affordability, insurance coverage, and equitable access for all patients who could benefit. As more potent drugs like retatrutide enter the market, discussions around drug pricing and healthcare system sustainability will intensify.
Furthermore, while the 40-week data is highly encouraging, long-term safety and efficacy beyond this period will be crucial for regulatory approval and widespread adoption. The ongoing studies will provide vital information on the sustained benefits and any potential rare or long-term adverse events. Adherence to chronic injectable therapies also remains a consideration for patients, though the weekly or bi-weekly injection schedules of modern incretin mimetics have significantly improved convenience compared to daily regimens.
In conclusion, Eli Lilly’s retatrutide represents a significant step forward in the battle against type 2 diabetes and obesity. The impressive weight loss and A1C reductions, coupled with improvements in cardiovascular risk factors and the unique triple agonist mechanism, highlight its potential to offer a new level of therapeutic efficacy. As the comprehensive Phase 3 program continues, the medical community and patients alike will eagerly await further data that could solidify retatrutide’s role as a transformative agent in cardiometabolic health.














Leave a Reply