The global transition toward renewable energy has long been anchored by silicon-based photovoltaic technology, a medium that demands extreme structural purity and energy-intensive manufacturing to function. However, a disruptive class of materials known as lead-halide perovskites has challenged this status quo, demonstrating remarkable energy-conversion efficiency despite being riddled with structural defects and impurities. For over a decade, the scientific community remained divided over how such "imperfect" materials could rival the performance of ultra-pure silicon. A definitive answer has finally emerged from the Institute of Science and Technology Austria (ISTA), where researchers have identified a hidden network of internal "highways" that allow electrical charges to bypass defects and travel vast distances. This discovery, published in Nature Communications, provides a comprehensive physical framework for understanding perovskites and paves the way for a new era of low-cost, high-efficiency solar energy.
The Silicon Standard and the Perovskite Disruption
For more than half a century, the solar industry has been defined by the Bell Labs-era legacy of crystalline silicon. To produce an efficient silicon solar cell, manufacturers must refine silicon to a "solar-grade" purity of 99.9999%. This process requires temperatures exceeding 1,400 degrees Celsius and complex vacuum environments to ensure that the resulting crystal lattice is nearly perfect. Any significant defect in a silicon wafer acts as a "trap" for electrical charges, causing them to recombine and vanish before they can be converted into usable electricity.
In stark contrast, lead-halide perovskites can be manufactured at room temperature using relatively simple solution-based methods, such as spin-coating or inkjet printing. Despite these "dirty" production methods, which inevitably introduce structural flaws, perovskite solar cells have seen an unprecedented rise in power conversion efficiency (PCE). In 2009, the first perovskite cells yielded a meager 3.8% efficiency. By 2024, laboratory-scale cells have surpassed 26%, effectively matching the performance of high-end commercial silicon cells that took six decades to perfect.
The central mystery facing physicists was the "recombination paradox." In most semiconductors, the presence of defects leads to rapid charge recombination. In perovskites, however, charges—specifically electrons and their positive counterparts, "holes"—seem to ignore these obstacles, remaining separated and mobile for much longer than theoretical models predicted.
Chronology of a Scientific Mystery: From 1970 to 2024
The journey of lead-halide perovskites began in the late 1970s, when they were first synthesized and characterized for their unique crystal structure, which mimics the mineral calcium titanate (the original "perovskite"). For decades, they were viewed as a chemical curiosity with little practical application. The timeline of their ascent to the forefront of materials science is marked by several key milestones:
- 1978: Researchers first describe the synthesis of methylammonium lead halide crystals, noting their semiconducting properties but focusing primarily on their basic chemical structure.
- 2009: Professor Tsutomu Miyasaka and his team at Toin University of Yokohama report the first use of perovskites in a liquid-electrolyte solar cell, achieving 3.8% efficiency.
- 2012: A breakthrough occurs as researchers replace the liquid electrolyte with a solid-state hole transporter, boosting efficiency to over 10% and improving stability. This sparks a global "gold rush" in perovskite research.
- 2015–2020: Efficiency rates climb rapidly, surpassing 20%. Simultaneously, perovskites are found to be highly effective for LEDs, X-ray detectors, and tandem solar cells (stacking perovskite on top of silicon).
- 2024: The ISTA study provides the first unified physical explanation for the material’s efficiency, identifying the role of domain walls in charge transport.
The ISTA Breakthrough: Identifying the Internal "Highways"
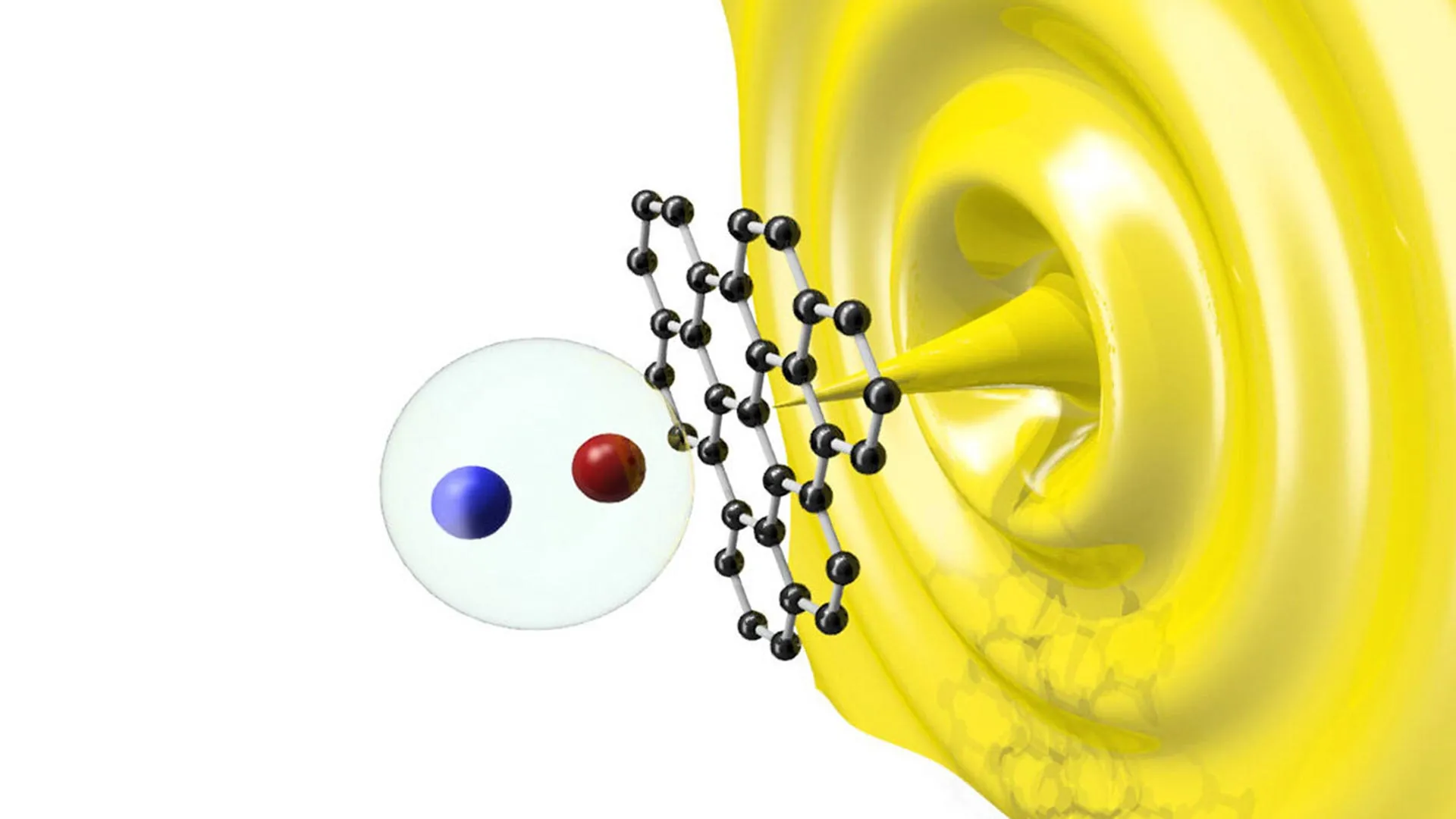
The research conducted by Dmytro Rak and Zhanybek Alpichshev at ISTA focused on resolving why charges in perovskites do not recombine immediately. When a photon hits a solar cell, it creates an electron and a hole. In most materials, these two are attracted to each other and form a bound state called an "exciton." If they recombine, the energy is lost as heat or light rather than being captured as electricity.
To investigate this, the ISTA team utilized advanced nonlinear optical techniques. By injecting charges deep into the bulk of a perovskite crystal using ultra-fast laser pulses, they observed a consistent, spontaneous electrical current. This current occurred without any external voltage, suggesting that the material possessed internal electric fields strong enough to pull electrons and holes apart.
"Our observations clearly indicated that even deep inside single crystals of unmodified, as-grown perovskites, there are internal forces that separate opposite charges," explains Alpichshev. The researchers hypothesized that these forces were concentrated at "domain walls"—interfaces where the orientation of the crystal lattice shifts slightly.
Methodology: "Angiography" for Crystals
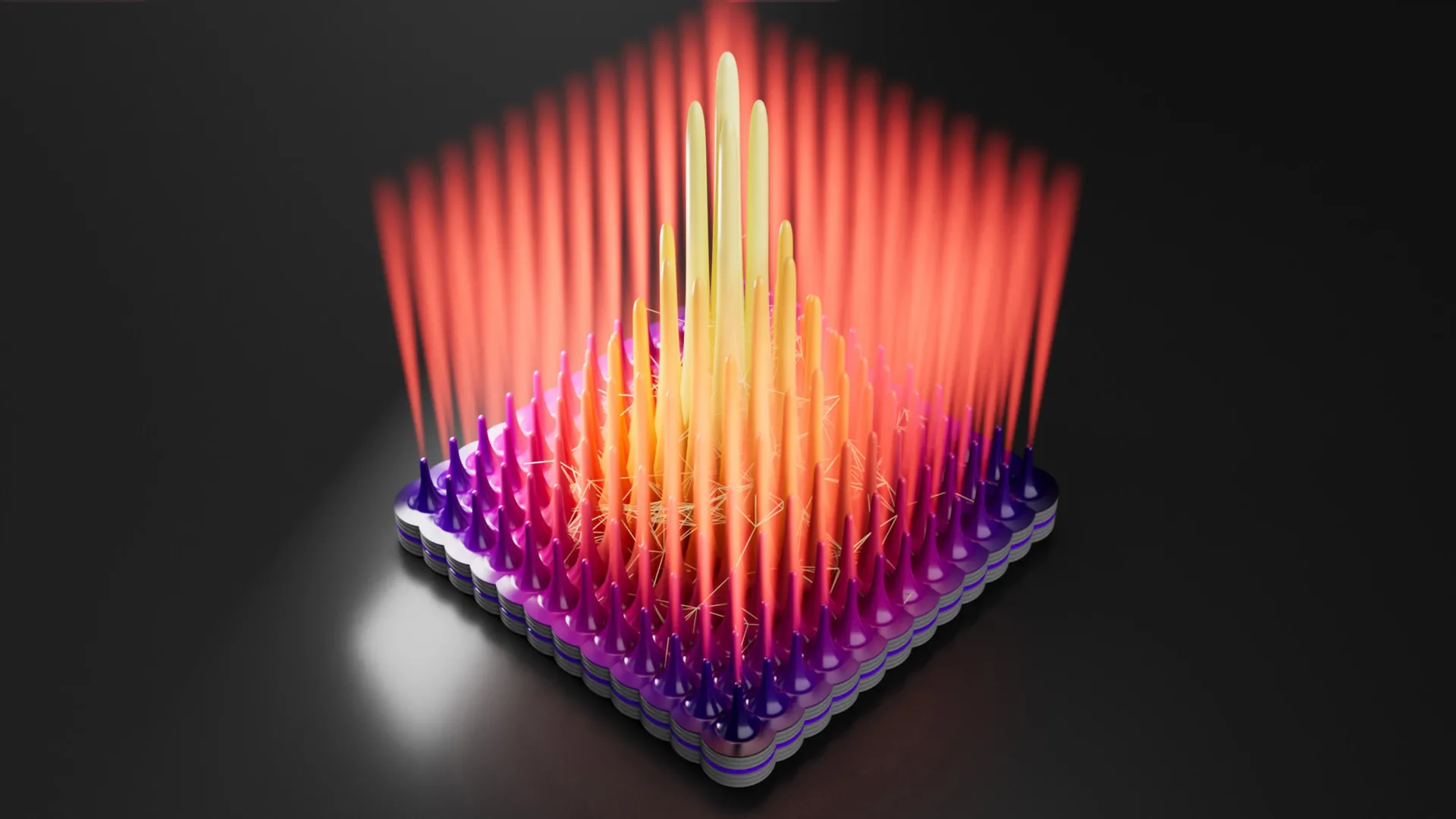
One of the primary obstacles in studying domain walls is that they are buried deep within the three-dimensional structure of the crystal. Standard surface-imaging tools, such as Atomic Force Microscopy (AFM), cannot see into the material’s interior. To solve this, Dmytro Rak developed an innovative visualization technique inspired by medical imaging.
Exploiting the fact that perovskites are ionic conductors, Rak introduced silver ions into the crystal. These ions naturally migrated through the material, seeking out the areas of least resistance and highest reactivity: the domain walls. Once the ions had accumulated along these hidden structures, they were chemically converted into metallic silver. This created a visible, three-dimensional "map" of the internal network, much like how contrast agents allow doctors to see blood vessels in human tissue.
The resulting images revealed a dense, interconnected network of domain walls permeating the entire crystal. These walls act as "highways" for charge carriers. When a photon creates an electron-hole pair near a domain wall, the local electric field at the wall pulls them in opposite directions. Once separated by the wall, the charges are shielded from one another, preventing recombination and allowing them to travel long distances—up to hundreds of microns—to reach the electrodes.
Supporting Data: The Scale of Efficiency
The significance of this discovery is highlighted by the scale of charge transport in perovskites compared to other thin-film materials. In organic solar cells, charges typically travel only a few nanometers before recombining. In lead-halide perovskites, the ISTA team confirmed that charges could traverse distances equivalent to the thickness of a human hair.
Data from the study indicates that:
- Charge Separation Velocity: The internal fields at domain walls are sufficient to separate excitons into free carriers within picoseconds.
- Diffusion Lengths: The effective diffusion length of charges in these "defective" crystals is comparable to that of high-purity, monocrystalline silicon.
- Ubiquity of Networks: The silver-ion staining technique confirmed that these domain-wall networks are a natural byproduct of the material’s crystallization process, explaining why even "crude" manufacturing methods yield efficient cells.
Industry Implications and Expert Reactions
The revelation that structural defects are a feature, rather than a bug, of perovskites has profound implications for the solar industry. Until now, much of the research has focused on trying to eliminate defects to make perovskites more like silicon. The ISTA findings suggest that a more effective strategy would be to engineer the domain-wall networks themselves.
"With this comprehensive picture, we are finally able to reconcile many previously conflicting observations," says Rak. "This insight could move perovskite solar cells closer to widespread real-world use by shifting our focus from chemical purity to structural engineering."
While the ISTA team’s work has been met with enthusiasm, industry analysts note that one significant hurdle remains: stability. Lead-halide perovskites are sensitive to moisture, oxygen, and heat, often degrading within months, whereas silicon cells are warranted for 25 years. However, by understanding the role of domain walls, engineers may be able to design "encapsulated" structures that protect these charge highways while maintaining their efficiency.
Analysis: The Future of the Energy Landscape
The ISTA discovery arrives at a critical juncture for the global energy sector. As nations strive to meet net-zero carbon targets, the demand for photovoltaics is projected to grow exponentially. Silicon, while reliable, is energy-intensive to produce and has a high "energy payback time"—the period a cell must operate to generate the energy used in its manufacture.
Perovskites offer a path to a significantly lower carbon footprint for the solar industry itself. Their ability to be manufactured at low temperatures and their high efficiency make them ideal candidates for "tandem cells." By layering a perovskite film onto a standard silicon cell, manufacturers can capture a broader spectrum of sunlight, pushing the combined efficiency toward 30% or higher.
The ISTA research provides the theoretical foundation needed to optimize these tandem structures. By manipulating the density and orientation of domain walls, scientists can potentially create "super-highways" for charges, further boosting the efficiency of the next generation of solar panels.
Conclusion: A Unified Theory of Perovskite Physics
The study by Rak and Alpichshev marks the end of a long-standing debate in materials science. By proving that the very imperfections of lead-halide perovskites are the source of their strength, the ISTA team has provided a roadmap for future development. The "highway" mechanism explains why these materials are so forgiving of impurities and why they have been able to challenge silicon’s dominance in such a short period.
As research transitions from understanding the fundamental physics to commercial-scale engineering, the insights gained from this "angiography" of crystals will likely be remembered as the turning point that brought perovskite solar technology out of the lab and into the light of day. The mystery of the flawed material’s perfection has been solved, and the future of solar energy looks brighter—and more efficient—than ever.














Leave a Reply